Abstract
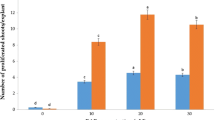
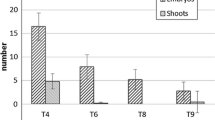
A method for micropropagation of Campanula isophylla Moretti is described. The method is based on division of the basal parts of shoot clusters into sections, each with four 3 mm stem stubs. Shoots from the shoot clusters are easy to root and give plants without apparent phenotypic aberrations. It is thus possible to propagate the stock and produce rooted plantlets in the same process. Basal sections of shoot clusters formed more shoots than shoot tips or single nodes. The medium used for propagation was MS with 4.4 μM benzyladenine (BA). Addition of naphthaleneacetic acid or raising the concentration of BA did not improve the results significantly. As primary explants 2 mm stem segments with an axillary or apical bud were used; smaller explants often failed to grow. For rooting the concentration of macronutrients was reduced to one-half, and BA was omitted. The cultures received an irradiance of 20 μmol m-2 s-1 fluorescent light; dry weight of shoots decreased if the irradiance was reduced. The method was used for propagation of 113 genotypes; shoot numbers and days to first root differed significantly among genotypes.
Similar content being viewed by others
References
BentzSE, ParlimanBJ, TalbottH-J & AckermanWL (1988) Factors affecting in vitro propagation of Yucca glauca. Plant Cell Tiss. Org. Cult. 14: 111–120
ColemanGD & ErnstSG (1990) Axillary shoot proliferation and growth of Populus deltoides shoot cultures. Plant Cell Rep. 9: 165–167
deFossardRA (1986) Principles of plant tissue culture. In: ZimmermanRH, GriesbachRJ, HammerschlagFA & LawsonRH (Eds) Tissue Culture as a Plant Production System for Horticultural Crops (pp 1–13). Martinus Nijhoff, Dordrecht
EvaldssonIE & WelanderNT (1985) The effects of medium composition on in vitro propagation and in vivo growth of Cordyline terminalis cv Atoom. J. Hort. Sci. 60: 525–530
FahrenkrogPS (1983) Untersuchungen zur in vitro Vermehrung und Organogenese bei Campanula pyramidalis. Thesis, Hannover University, FRG
HutchinsonJF (1984) Factors affecting shoot proliferation and root initiation in organ cultures of the apple ‘Northern Spy’. Scientia Hort. 22: 347–358
IkedaLR & MJTanabe (1989) In vitro subculture applications for ginger. HortScience 24: 142–143
JamesDJ & NewtonB (1977) Auxin:cytokinin interactions in the in vitro micropropation of strawberry plants. Acta Hort. 78: 321–331
LauzerD & ViethJ (1990) Micropropagation of seedderived plants of Cynara scolymus L., cv. ‘Green Globe’. Plant Cell Tiss. Org. Cult. 21: 237–244
McCownDD & McCownBH (1987) North American hardwoods. In: BongaJM & DurzanDJ (Eds) Cell and Tissue Culture in Forestry, Vol 3 (pp 247–260). Martinus Nijhoff, Boston
MurashigeT & SkoogF (1962) A revised medium for rapid growth and bio-assays with tobacco tissue cultures. Physiol. Plant. 15: 473–497
ParfittDE & AlmehdiAA (1986) In vitro propagation of peach: II. A medium for in vitro multiplication of 56 peach cultivars. Fruit Var. J. 40: 46–47
ReedBM (1990) Multiplication of Rubus germplasm in vitro: A screen of 256 accessions. Fruit Var. J. 44: 141–148
SAS Institute Inc. (1989) SAS/STAT User's Guide, Version 6, Fourth Edition, SAS Institute Inc, Cary NC
SauerA, WaltherF & PreilW (1985) Different suitability for in vitro propagation of rose cultivars. Gartenbauwissenschaft 50: 133–138
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Brandt, K. Micropropagation of Campanula isophylla Moretti. Plant Cell Tiss Organ Cult 29, 31–36 (1992). https://doi.org/10.1007/BF00036143
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00036143