Abstract
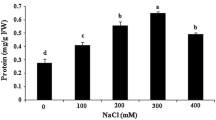
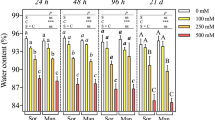
Callus and suspension cultures adapted to various concentrations of NaCl or mannitol were developed from the cultivated potato Solanum tuberosum cv. Desire. Growth of the calli was less inhibited by mannitol than by iso-osmotic concentrations of NaCl. Reduction of growth by both NaCl and mannitol was considerably lower in osmotically adapted calli than in non-adapted ones. Salt-adapted suspension cultures that grew in the medium to which they had been originally adapted had a shorter lag in growth as well as a shorter time required to achieve the maximum growth, as compared with non-adapted cells. Suspension cultures adapted to NaCl concentrations higher than 150 mM were obtained only after preadaptation to osmotic stress. Adaptation of these cells was found to be stable. Accumulation of Na+ was lower and level of K+ was more stable in osmotically adapted than in non-adapted calli, when both were exposed to salt. Potassium level in NaCl-adapted calli exposed to saline medium was lower than that in non-adapted calli in standard medium. The maximum of Cl− and Na+ accumulation was reached at higher external salt concentration in salt-adapted than in non-adapted suspension cultures. In both callus and suspension cultures, Cl− accumulated more than Na+. Potassium level decreased more in non-adapted than in NaCl-adapted suspension cultures. The decrease of osmotic potential in osmotically adapted calli exposed to mannitol and in salt-adapted calli and suspension cultures exposed to salt was correlated to the increase of the external concentration. Such a correlation was not found in osmotically adapted calli exposed to salt. Non-electrolytes were found to be the main contributors to the decrease is osmotic potential in both callus and suspension cultures.
Similar content being viewed by others
References
Ben-Hayyim G (1987) Relationship between salt tolerance and resistance to polyethylene glycol-induced water stress in cultured citrus cells. Plant Physiol 85: 430–433
Ben-Hayyim G, Kochba J (1983) Aspects of salt tolerance in a NaCl-selected stable cell line of Citrus sinensis. Plant Physiol 72: 685–690
Ben-Hayyim G, Spiegel-Roy P, Neumann H (1985) Relation between ion accumulation of salt-sensitive and isolated stable salt-tolerant cell lines of Citrus aurantium. Plant Physiol 78: 144–148
Binzel ML, Hasegawa PM, Handa AK, Bressan RA (1985) Adaptation of tobacco cells to NaCl. Plant Physiol 78: 118–125
Binzel ML, Hasegawa PM, Rhodes D, Handa S, Handa AK, Bressan RA (1987) Solute accumulation in tobacco cells adapted to NaCl. Plant Physiol 84: 1408–1415.
Bressan RA, Hasegawa PM, Handa AK (1981) Resistance of cultured higher plant cells to polyethylene glycol-induced water stress. Plant Sci Lett 21: 23–30
Croughan TP, Stavarek SJ, Rains DW (1978) Selection of a NaCl-tolerant line of cultivated alfalfa cells. Crop Sci 18: 959–963
Dracup M, Gibbs J, Greenway H (1986) Melibiose, a suitable nonpermeating osmoticum for suspensioncultured tobacco cells. J Exp Bot 37: 1079–1089
Gibbs J, Dracup M, Greenway H, McComb JA (1989) Effects of high NaCl on growth, turgor and internal solutes of tobacco callus. J Plant Physiol 134: 61–69
Handa AK, Bressan RA, Handa S, Hasegawa PM (1982) Tolerance to water and salt stress in cultured cells. In: Fujiwara A (Ed) Proc 5th Int Congr Plant Tissue and Cell Culture (pp 471–474) Abe Photo Printing Co, Tokyo
Harms CT, Oertli JJ (1985) The use of osmotically adapted cell cultures to study salt tolerance in vitro. J Plant Physiol 120: 29–38
Hasegawa PM, Bressan RA, Handa AK (1980) Growth characteristics of NaCl-selected and nonselected cells of Nicotiana tabacum L. Plant Cell Physiol 21: 1347–1355
Hasegawa PM, Bressan RA, Handa AK (1986) Cellular mechanism of salinity tolerance. HortScience 21: 1356–1324
McCoy TJ (1987) Characterization of alfalfa (Medicago sativa L.) plants regenerated from selected NaCl-tolerant cell lines. Tissue Culture Assoc Meeting, Washington DC (pp 92–96)
McHughen A, Swartz M (1984) A tissue culture derived salt-tolerant line of flax (Linum usitatissimum). J Plant Physiol 117: 109–117
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol Plant 15: 473–497
Nabors MW, Gibbs GE, Bernstein CS, Meis ME (1980) NaCl-tolerant tobacco plants from cultured cells. Z Pflanzenphysiol 97: 13–17
Pua EC, Thorpe TA (1986) Differential Na2SO4-grown callus. Plant Cell Environ 9: 9–16
Richards LA (Ed) (1954) Diagnosis and Improvement of Saline and Alkali Soils. US Dept of Agriculture Handbook No 60
Shimshi D, Livne A (1967) The estimation of the osmotic potential of plant by refractometry and conductimeter: a field method. Ann Bot 31: 505–511
Tal M (1989) Somoclonal variation for salt resistance. In: Bajaj YPS (Ed) Biotechnology in Agriculture and Forestry. Springer-Verlag, Berlin (in press)
Taylor JA, West DW (1980) The use of Evan's blue stain to test the survival of plant cells after exposure to high salt and high osmotic pressure. J Exp Bot 31: 571–576
Thompson MR, Douglas TJ, Obata-Sasamoto H, Thorpe TA (1986) Mannitol metabolism in cultured plant cells. Physiol Plant 67: 365–369
Van Swaaij N, Jacobsen E, Kiel JAKW, Feenstra WJ (1986) Selection, characterization and regeneration of hydroxyproline-resistant cell lines of Solanum tuberosum: tolerance to NaCl and freezing stress. Physiol Plant 68: 359–366
Watad AA, Reinhold L, Lerner HR (1983) Comparison between a stable NaCl-selected Nicotiana cell line and the wild type K+, N+ and proline pools as a function of salinity. Plant Physiol 73: 624–629
Watad AA, Lerner HR, Reinhold L (1985) Stability of the salt-resistance character in Nicotiana cell lines adapted to grow in high NaCl concentrations. Physiol Veg 23: 887–894
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sabbah, S., Tal, M. Development of callus and suspension cultures of potato resistant to NaCl and mannitol and their response to stress. Plant Cell Tiss Organ Cult 21, 119–128 (1990). https://doi.org/10.1007/BF00033430
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00033430