Abstract
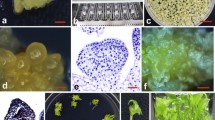
Genetically transformed alfalfa (Medicago sativa L., cv. Zaječarska 83) plantlets were obtained by inoculating somatic embryos with Agrobacterium tumefaciens strains A281/pGA472 and LBA4404/pBI121. Single somatic embryos, 5–7 mm long, were released from a repetitively embryogenic culture, wounded, and cocultivated with the bacteria. The agar-solidified culture medium contained mineral salts, vitamins, 40 g l−1 sucrose, 1 g l−1 yeast extract and 0.05 mg l−1 BA. Five clones, transformed with A281/pGA472, and 4 clones transformed with LBA4404/pBI121, were selected for proliferation by repetitive somatic embryogenesis, on media containing 100 mg l−1 of kanamycin. The transformation of kanamycin-resistant clones was confirmed by assaying the activity of neomycin phosphotransferase II and/or β-glucuronidase enzymes, and by the Southern blot analysis. It is suggested that the transformation/regeneration system based on somatic embryogenesis may be suitable for establishing transgenic alfalfa lines. The relatively low frequency of embryo transformation is compensated for by abundant proliferation in secondary somatic embryogenesis.
Similar content being viewed by others
Abbreviations
- BA:
-
6-benzyladenine
- GUS:
-
β-glucuronidase
- Km:
-
kanamycin
- NPTII:
-
neomycin phosphotransferase II
- X-gluc:
-
5-bromo-4-chloro-3-indolyl-β-glucuronic acid
- BM:
-
basal medium
References
An G, Watson B D, Stachel S, Gordon MP & Nester E W (1985) New cloning vehicles for transformation of higher plants. EMBO J. 4: 277–284
Brown D C W & Atanassov A (1985) Role of genetic background in somatic embryogenesis in Medicago. Plant Cell Tiss. Org. Cult. 4: 111–122
Chabaud M, Passiatore J E, Cannon F & Buchanan-Wollaston V (1988) Parameters affecting the frequency of kanamycin resistant alfalfa obtained by Agrobacterium tumefaciens mediated transformation. Plant Cell Rep. 7: 512–516
Deak M, Kiss G B, Koncz C & Dudits D (1986) Transformation of Medicago by Agrobacterium mediated gene transfer. Plant Cell Rep. 5: 97–100
DeKathen A & Jacobsen H J (1990) Agrobacterium tumefaciens mediated transformation of Pisum sativum L. using binary and cointegrate vectors. Plant Cell Rep. 9: 276–279
Dellaporta,S L, Wood,J & Hicks,J B (1985) Maize DNA Miniprep. In: Malmberg,R, Messing,J & Sussex,I (Eds.) Molecular Biology of Plants: A Laboratory Course Manual (pp 36–37). Cold Spring Harbor, New York
D'Halluin K, Botterman J & DeGreff W (1990) Engineering of herbicide-resistant alfalfa and evaluation under field conditions. Crop Sci. 30: 866–871
Dos Santos A V P, Cutter E G & Davey M R (1983) Origin and development of somatic embryos in Medicago sativa (Alfalfa). Protoplasma 117: 107–115
Du S, Erickson L & Bowley S (1994) Effect of plant genotype on the transformation of cultivated alfalfa (Medicago sativa) by Agrobacterium tumefaciens. Plant Cell Rep. 13: 330–334
Horsch R B, Fry J E, Hoffmann N L, Eicholz D, Rogers S G & Fraley R T (1985) A simple and general method for transferring genes into plants. Science 227: 1229–1231
Jefferson R A, Kavanagh T A & Bevan M W (1987) GUS fusions: β-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J. 6: 3901–3907
Kao K N & Michayluk M R (1981) Embryoid formation in alfalfa cell suspension cultures from different plants. In Vitro 17: 645–648
Kutchuk N, Komarnitski I, Shakhovsky A & Gleba Y (1990) Genetic transformation of Medicago species by Agrobacterium tumefaciens and electroporation of protoplasts. Plant Cell Rep. 8: 660–663
Maheshwaran G & Williams E G (1984) Direct somatic embryoid formation on immature embryos of Trifolium repens, T. pratense and Medicago sativa, and rapid clonal propagation of T. repens. Ann. Bot. 54: 201–211
Maniatis, T, Fritsch, F F & Sambrook, J (1982) Molecular Cloning: A Laboratory Manual. Cold Spring Harbor, New York
Mariotti D, Davey M R, Draper J, Freeman J P & Cocking E C (1984) Crown gall tumorigenesis in the forage legume Medicago sativa L. Plant Cell Physiol. 25: 473–482
Mathews H, Litz R E, Wilde H D, Merkle S A & Wetzstein H Y (1992) Stable integration and expression of β-glucuronidase and NPTII genes in mango somatic embryos. In Vitro Cell. Dev. Biol. 28P: 172–178
McCoy T & Walker K (1984) Alfalfa. In: Ammirato P V, Evans D A, Sharp W R & Yamada Y (Eds.) Handbook of Plant Cell Culture. Vol 3. Crop Species (pp 171–192). Macmillan Publ. Co., New York and London
McGranahan G H, Leslie C A, Uratsu S L, Martin L A & Dandekar A M (1988) Agrobacterium-mediated transformation of walnut somatic embryos and regeneration of transgenic plants. Bio/Technol. 6: 800–804
McGranahan G H, Leslie C A, Uratsu S L & Dandekar A M (1990) Improved efficiency of the walnut somatic embryo gene transfer system. Plant Cell Rep. 8: 512–516
McGranahan G H, Leslie C A, Dandekar A M, Uratsu S L & Yates I E (1993) Transformation of pecan and regeneration of transgenic plants. Plant Cell Rep. 12: 634–638
McHughen A & Jordan M C (1989) Recovery of transgenic plants from “escape” shoots. Plant Cell Rep. 7: 611–614
McKersie B D, Chen Y, deBeus M, Bowley S R, Bowler C, Inze D, D'Halluin K & Botterman J (1993) Superoxide dismutase enhances tolerance of freezing stress in transgenic alfalfa (Medicago sativa L.). Plant Physiol. 103: 1155–1163
Mijatović M, Milijić S & Mitrović S (1985) Biological characteristics and productivity of the new alfalfa cultivar “Zaječarska 83”. V Yug. Symp. on Forage Crops (pp 15–18). Banja Luka (In Serbian)
Nagata T, Okada K, Kawazu T & Takebe I (1987) Cauliflower mosaic virus 35S promoter directs S phase specific expression in plant cells. Mol. Gen. Genet. 207: 242–244
Ninković S, Miljuš-Djukić J & Nešković M (1988) In vitro performance of Medicago sativa L. cv. Zaječarska 83: Direct somatic embryogenesis, cell and protoplast culture. Bul. Inst. Jard. Bot. Univ. Beograd 22: 27–34
Parrott W A, Hoffmann L M, Hilderbrandt D F, Williams E G & Collins G B (1989) Recovery of primary transformants of soybean. Plant Cell Rep. 7: 615–617
Polito V S, McGranahan G, Pinney K, Leslie C (1989) Origin of somatic embryos from repetitively embryogenic cultures of walnut (Juglans regia L.): implications for Agrobacterium-mediated transformation. Plant Cell Rep. 8: 219–221
Saunders J W & Bingham E T (1972) Production of alfalfa plants from callus tissue. Crop Sci. 12: 804–808
Schrammeijer B, Sijmons P C, van denElzen P J M & Hoekama A (1990) Meristem transformation of sunflower via Agrobacterium. Plant Cell Rep. 9: 55–60
Shahin E A, Spielmann A, Sukhapinda K, Simpson R B & Yashar M (1986) Transformation of cultivated alfalfa using disarmed Agrobacterium tumefaciens. Crop Sci. 26: 1235–1239
Thomas M R, Rose R J & Nolan K E (1992) Genetic transformation of Medicago trunculata using Agrobacterium with genetically modified Ri and disarmed Ti plasmids. Plant Cell Rep. 11: 113–117
VanLarebeke N, Genetello C H, Hernalsteens J P, DePicker A, Zaenen I, Messens E, VanMontagu M & Schell J (1977) Transfer of Ti plasmids between Agrobacterium strains by mobilization with the conjugative plasmid RP4. Molec. Gen. Genet. 152: 119–124
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ninković, S., Miljuš-Djukić, J. & Nešković, M. Genetic transformation of alfalfa somatic embryos and their clonal propagation through repetitive somatic embryogenesis. Plant Cell Tiss Organ Cult 42, 255–260 (1995). https://doi.org/10.1007/BF00029996
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00029996