Summary
Anthers of male fertile, cytosterile and restored male fertile clones of Petunia hybrida were compared for esterase activity and composition in subsequent stages of microsporogenesis. Three methods were applied (i) ultra-thin layer isoelectric focussing on polyacryl amide gels, (ii) quantitative spectrophotometrical assay, (iii) histochemical determination of total esterase activity associated to the azo-coupling method (Pearse, 1972).
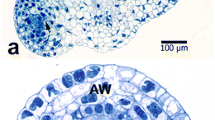
In male fertile and restorer idiotypes the isozyme patterns were quite similar. Both the number and intensity of bands increased gradually till the tetrad stage. In contrast, esterase activity in cytosterile anthers remained at a low level and hardy any new bands showed up in later stages. This unvariable, low activity level in cytosterile anthers was also found in the spectrophotometric assay. Histochemical determinations revealed that in male fertile anthers esterase activity is concentrated in the outer tapetal layer at late prophase and that it accumulates there till the early microspore stage. In male sterile anthers esterase accumulation in the tapetal cells stops at the moment that tapetal breakdown becomes visible. This suggests that differences in esterase activity and composition are an effect rather than a cause of the failing pollen formation.
Similar content being viewed by others
References
Abbott, A. G., C. C., Ainsworth & R. B., Flavell, 1984. Characterization of anther differentiation in cytoplasmic male sterile maize using a specific isozyme system (esterase). Theor. Appl. Genet. 67: 469–473.
Ahokas, H., 1976. Evidence of a pollen esterase capable of hydrolyzing sporopollenin. Experimentia 32: 175–177.
Bassiri, A., 1976. Barley cultivar identification by use of isoenzyme electrophoretic pattern. Canad. J. Plant Sci. 56: 1–6.
Bennett, M. D., 1976. The cell in sporogenesis and spore development. In: Yeoman, M. M. (Ed.), Cell division in higher plants. Academic Press, London, pp. 161–198.
Bino, R. J., 1985a. Ultrastructural aspects of cytoplasmic male sterility in Petunia hybrida. Protoplasma 127: 230–240.
Bino, R. J., 1985b. Histological aspects of microsporogenesis in fertile, male sterile and restored fertile Petunia hybrida. Theor. Appl. Genet. 69: 423–428.
Frankel, R. & E., Galun, 1977. Pollination mechanisms, reproduction and plant breeding. Springer Verlag, Berlin etc, pp. 196–234.
Frankel, R., S., Izhar & J., Nitsan, 1969. Timing of callase activity and cytoplasmic male sterility in Petunia. Bioch. Genet. 3: 451–455.
Gahan, P. B. & J., McLean, 1969. Subcellular localization and possible functions of acid glycerophosphatases and naphtol esterases in plant cells. Planta 89: 126–135.
Höhler, B. & Th., Börner, 1980. Studies on isoenzymes of anther tissues of fertile and cytoplasmic male sterile wheat plants. Biochem. Physiol. Pflanzen 175: 562–569.
Holt, S. J. & R. M., Hicks, 1966. The importance of osmiophilia in the production of stable azoindoxyl complexes of high contrast for combined enzyme cytochemistry and electron microscopy. J. Cell Biol. 29: 316–366.
Izhar, S. & R., Frankel, 1971. Mechanisms of male sterility in Petunia. The relationship between pH, callase activity in the anthers and the breakdown of the microsporogenesis. Theor. Appl. Genet. 41: 104–108.
Jensen, W. A., 1985. The effect of a chemical hybridiying agent on the development of wheat pollen. In: Willemse, M. T. M. & J. L.van, Went (Eds), Sexual reproduction in seed plants, ferns and mosses. PUDOC, Wageningen, p. 134.
Karim, M. A., S. L., Metha & P. M., Singh, 1984. Studies on esterase isoenzyme patterns in anthers and seeds of male sterile wheats. Z.Pflanzenzüchtg. 93: 309–319.
Knox, R. B., B. J., Howlett, J., Heslop-Harrison & Y., Heslop-Harrison, 1973. Pollen-wall proteins: gametophytic and sporophytic fractions: their origin, localization and emission. Incompatibility Newsletter 3: 77–78.
Laser, K. D. & N. R., Lersten, 1972. Anatomy and cytology of microsporogenesis in cytoplasmic male sterile angiosperms. Bot. Rev. 38: 425–454.
Marrewijk, G. A. M.van, 1969. Cytoplasmic male sterility in petunia. I. Restoration of fertility with special reference to the influence of environment. Euphytica 18: 1–20.
Payne, R. C. & T. T., Kaszykovski, 1978. Esterase isoenzyme differences in seed extracts among soybean cultivars. Crop Sci. 18: 557–559.
Pearse, A. G. E., 1972. Histochemistry, theoretical and applied. Vol. 2. Churchill Livingstone, Edinburgh etc., pp. 761–807.
Radola, B. J., 1980. Ultrathin-layer isoelectric focussing in 50–100 μm polyacrylamide gels on silanized glass plates or polyester films. Electrophoresis 1: 43–56.
Tripathi, D. P., A. B., Dongre, S. L., Metha & N. G. P., Rao, 1982. Soluble protein and esterase isoenzyme pattern on isoelectric focussing from seeds and anthers of diverse cytoplasmic genic male sterile sorghums (Sorghum bicolor L. Moench). Z. Pflanzenzüchtg. 88: 69–78.
Vithanage, H. I. M. V. & R. B., Knox, 1976. Pollen-wall proteins: quantitative cytochemistry of the origins of intine and exine enzymes in Brassica oleracea. J. Cell Sci. 21: 423–435.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Van Marrewijk, G.A.M., Bino, R.J. & Suurs, L.C.J.M. Characterization of cytoplasmic male sterility in Petunia hybrida. I. Localization, composition and activity of esterases. Euphytica 35, 77–88 (1986). https://doi.org/10.1007/BF00028544
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00028544