Abstract
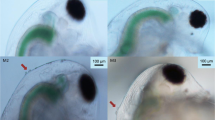
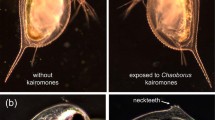
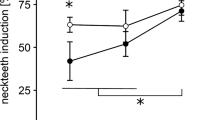
In ponds, a chemical produced by predaceous Chaoborus (Insecta, Diptera) larvae changes the development of juvenile Daphnia pulex (Crustacea, Branchiopoda) so the juveniles grow spines (neckteeth) on the back of their head. It is generally assumed that the spined phenotype is (or is an indicator of) a morphological predator defense. The research reported here tests the hypothesis that the induced neckteeth do in fact increase Daphnia survivorship, over a range of temperatures. Predation experiments were conducted over a range of temperatures from 6 to 22 °C using fourth instar Chaoborus americanus larvae as the predator. The prey were a mixture of spined (induced “necktooth” phenotype) and unspined (uninduced) juvenile Daphnia pulex. At 6 and 11 °C, Chaoborus selected the unspined phenotype over the spined phenotype, as expected. However, at 22 °C, the selectivity was reversed: significantly fewer on the spined survived compared to the unspined phenotype. These results suggest that the spined phenotype may either increase or decrease Daphnia pulex survival, depending on temperature and clone.
Similar content being viewed by others
References
Bengtsson, J., 1987. Competitive dominance among Cladocera: Are single-factor explanations enough? Hydrobiologia 145: 19–28.
Black, A. R., 1991. Predator-induced phenotypic plasticity in Daphnia pulex. Ph.D. Thesis, University of Wisconsin, Madison, Wisconsin.
Black, A. R., 1993. Predator-induced phenotypic plasticity in Daphnia pulex: Life history and morphological responses to Notonecta and Chaoborus. Limnol. Oceanogr. 38: 986–989.
Black, A. R. & S. I. Dodson, 1990. Demographic costs of Chaoborus-induced phenotypic plasticity in Daphnia pulex. Oecologia 83: 117–122.
De Bernardi, R. & R. H. Peters, 1987. Daphnia. Memorie Dell'Instituto Italiano di Idrobiologia-Verbania Pallanza Vol. 45. Consiglo Nationale dell Richere, Pallanza, Italy: 1–9.
DeMott, W. R., 1989. The role of competition in zooplankton succession. In U. Sommer (ed.), Plankton Ecology: Succession in Plankton Communities. Springer-Verlag, New York: 195–252.
Dini, M. L., J. O'Donnell, S. R. Carpenter, M. M. Elser, J. J. Elser & A. M. Berquist, 1987. Daphnia size structure, vertical migration, and phosphorus redistribution. Hydrobiologia 150: 185–191.
Dodson, S. I., 1974. Adaptive change in plankton morphology in response to size-selective predation: A new hypothesis of cyclomorphosis. Limnol. Oceanogr. 19: 721–729.
Dodson, S. I., 1989. Predator-induced reaction norms. Bioscience 39: 447–452.
Elser, M. M., C. N. Von Ende, P. Soranno & S. R. Carpenter, 1987. Chaoborus populatins: Response to food web manipulation and potential effects on zooplankton communities. Can. J. Zool. 65: 2846–2852.
Havel, J. E. & S. I. Dodson, 1984. Chaoborus predation on typical and spined morphs of Daphnia pulex: Behavioral observations. Limnol. Oceanogr. 29: 487–494.
Krueger, D. A. & S. I. Dodson, 1981. Embryological induction and predation ecology in Daphnia pulex. Limnol. Oceanogr. 26: 219–223.
Krylov, P.I., 1992. Density-dependent predation of Chaoborus flavicans on Daphnia longispina in a small lake: The effect of prey size. Hydrobiologia 239: 131–140.
Larsson, P. & S. Dodson, 1993. Invited review: Chemical communication in planktonic animals. Arch. Hydrobiologia 129: 129–155.
Lehman, J. T., 1980. Nutrient recycling as an interface between algae and grazers in freshwater communities. Am. Soc. Limnol. Oceanogr. Spec. Symp. 3: 251–263. New England.
Lewis, W. M. Jr., 1980. Evidence for stable zooplankton community structure gradients maintained by predation. Am. Soc. Limnol. Oceanogr. Spec. Symp. 3: 625–634. New England.
Nichols, H. W., 1973. Growth Media — Freshwater. In J. R. Stein (ed.), Handbook of Phycological Methods: Culture Methods and Growth Measurements. Cambridge University Press, Cambridge: 7–24.
Parejko, K. S., 1992. Embryology of Chaoborus-induced spines in Daphnia pulex. Hydrobiologia 231: 77–84.
Parejko, K. S. & S. I. Dodson, 1991. Progress towards characterization of a predator/prey kairomone: Daphnia pulex and Chaoborus americanus. Hydrobiologia 198: 51–59.
Pastorok, R. A., 1981. Prey vulnerability and size selection by Chaoborus larvae. Ecology 62: 1311–1324.
Riessen, H. P., 1992. Cost-benefit model for the induction of an anti-predator defense. Am. Nat. 140: 349–362.
Seale, D. B. & F. P. Binkowski, 1988. The vulnerability of early life intervals of Coregonus hoyi to predation by a freshwater mysid, Mysis relicta. Envir. Biol. Fishes 21: 117–126.
Siegel, S., 1956. Nonparametric statistics for the behavioral sciences. McGraw-Hill, New York.
Spitze, K., 1992. Predator-mediated plasticity of prey life history and morphology: Chaoborus americanus predation on Daphnia pulex. Am.Nat. 139: 229–247.
Threlkeld, S. T., 1986. Differential temperature sensitivity of two cladoceran species to resource variation during a blue-green algal bloom. Can. J. Zool. 64: 1739–1744.
Walls, M. & M. Ketola, 1989. Effects of predator-induced spines on individual fitness in Daphnia pulex. Limnol. Oceanogr. 34: 390–396.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dodson, S.I., Wagner, A.E. Temperature affects selectivity of Chaoborus larvae-eating Daphnia . Hydrobiologia 325, 157–161 (1996). https://doi.org/10.1007/BF00028276
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00028276