Abstract
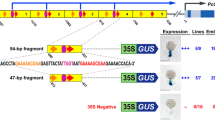
Type I element (CCACGTCACCGATCCGCG) is a well-conserved regulatory element found in proximal promoter region of a certain class of plant histone genes, that is composed of two independent cis-acting elements of the hexamer (ACGTCA) and the reverse-oriented octamer (GATCCGCG) motifs. To investigate functional role(s) of the type I element in regulation of a wheat histone H3 gene (TH012) promoter activity in vivo, base substitution mutations were introduced into the element and activities of the mutated promoters were examined in cultured rice cells, and in regenerated roots and anther walls of transgenic rice plants by employing a GUS reporter system. Mutations of each or both of the hexamer and the octamer motifs caused a reduction in the promoter activity in protoplasts transfected transiently or stably transformed calli. The mutation of the octamer motif with or without the mutation of the hexamer motif caused a marked reduction of the promoter activity in the root meristem of transgenic rice although the mutation of the hexamer motif alone caused a weak reduction. In contrast to these results, no effect of the mutations of either the hexamer or the octamer motif was found in the anther wall in which replication-independent activity of the H3 promoter was observed. Our results suggested that the hexamer and the octamer motifs may play important role(s) in regulation of replication-dependent but not of replication-independent expression of the wheat histone H3 gene.
Similar content being viewed by others
References
Atanassova R, Chaubet N, Gigot C: A 126 bp fragement of a plant histone gene promoter confers preferential expression in meristem of transgenic Arabidopsis. Plant J 2: 291–300 (1992).
Brignon P, Chaubet N: Constitutive and cell-division-inducible protein-DNA interactions in two maize histone gene promoters. Plant J 4: 445–457 (1993).
Chaubet N, Phillips G, Chaboute ME, Ehling M, Gigot C: Nucleotide sequences of two corn histone H3 genes. Genomic organization of the corn histone H3 and H4 genes. Plant Mol Biol 6: 253–263 (1986).
Chaubet N, Clement B, Philipps G, Gigot G: Organ-specific expression of different histone H3 and H4 gene subfamilies in developing and adult maize. Plant Mol Biol 17: 935–940 (1991).
Chaubet N, Clement B, Gigot G: Genes encoding a histone H3.3-like variant in Arabidopsis contain intervening sequences. J Mol Biol 225: 569–574 (1992).
Heinz N: The regulation of histone gene expression during the cell cycle. Biochim Biophys Acta 1088: 327–339 (1991).
Jefferson RA: Assaying chimeric genes in plants: the GUS gene fusion system. Plant Mol Biol Rep 5: 387–405 (1987).
Kapros T, Bogre L, Nemeth K, Bako L, Gyorgyey J, Wu SC, Dudits D: Differential expression of histone H3 gene variant during cell cycle and somatic embryogenesis in alfalfa. Plant Physiol 98: 621–625 (1992).
Kapros T, Stefanov I, Magyar Z, Ocsovszky I, Dudits D: A short histone H3 promoter from alfalfa specifies expression in S-phase cells and meristems. In Vitro Cell Dev Biol 29P: 27–32 (1993).
Kawata T, Nakayama T, Mikami K, Tabata T, Takase H, Iwabuchi M: DNA binding protein(s) interact with a conserved nonameric sequence in the upstream regions of wheat histone genes. FEBS Lett 239: 319–323 (1988).
Konig AJ, Tanimoto EY, Kiehne K, Rost T, Comai L: Cell-specific expression of plant histone H2A genes. Plant Cell 3: 657–665 (1991).
Kosugi S, Ohashi Y, Nakajima K, Arai Y: An improved assay for β-glucuronidase in transformed cells: Methanol almost completely suppresses a putative endogenous β-glucuronidase activity. Plant Sci 70: 133–140 (1990).
Kyozuka J, Shimamoto K: Transformation and regeneration of rice protoplasts. In: Lindsey K (eds) Plant Tissue Culture Manual, pp. B1: 1–16. Kluwer Academic Publishers, Dordrecht (1991).
Lepetit M, Ehling M, Chaubet N, Gigot C: A plant histone gene promoter can direct both replication-dependent and- independent gene expression in transgenic plants. Mol Gen Genet 231: 276–285 (1992).
Lepetit M, Ehling M, Atanassova R, Chaubet N, Gigot C: Replication-independent cis-acting element of a maize histone gene promoter. Plant Sci 89: 177–184 (1993).
Mikami K, Tabata T, Kawata T, Nakayama T, Iwabuchi M: Nuclear protein(s) binding to the conserved DNA hexameric sequence postulated to regulate transcription of wheat histone genes. FEBS Lett 223: 273–278 (1987).
Mikami K, Iwabuchi M: Regulation of cell cycle-dependent gene expression. In: Verma DPS (eds) Control of Plant Gene Expression, pp. C4: 51–68. CRC Press, Boca Raton (1993).
Minami M, Huh G-H, Yang P, Iwabuch M: Coordinate gene expression of five subclass histones and the putative transcription factors, HBP-1a and HBP-1b, of histone genes in wheat. Plant Mol Biol 23: 429–434 (1993).
Nakayama T, Iwabuchi M: Regulation of wheat histone gene expression. Crit Rev Plant Sci 12: 97–110 (1993).
Nakayama T, Ohtsubo N, Mikami K, Kawata T, Tabata T, Kanazawa M, Iwabuchi M: cis-acting sequences that modulate transcription of wheat histone H3 gene and 3′ processing of H3 premature mRNA. Plant Cell Physiol 30: 825–832 (1989).
Nakayama T, Sakamoto A, Yang P, Minami M, Fujimoto Y, Ito T, Iwabuchi M: Highly conserved hexamer, octamer and nonamer motifs are positive cis-regulatory elements of wheat histone H3 gene. FEBS Lett 300: 167–170 (1992).
Ohtsubo N, Nakayama T, Terada R, Shimamoto K, Iwabuchi M: Proximal promoter region of the wheat histone H3 gene confers S phase-specific transcription in transformed rice cells. Plant Mol Biol 23: 553–565 (1993).
Osley MA: The regulation of histone synthesis in the cell cycle. Annu Rev Biochem 60: 827–861 (1991).
Raghavan V: mRNAs and a cloned histone gene are differentially expressed during anther and pollen development in rice (Oryza sativa L.) J. Cell Sci 92: 217–229 (1989).
Razafimahatratra P, Chaubet N, Philipps G, Gigot C: Nucleotide sequence and expression of a maize H1 histone cDNA. Nucl Acids Res 19: 1491–1476 (1991).
Sakamoto A, Minami M, Huh G-H, Iwabuchi M: The putative zinc-finger protein WZF1 interacts with a cis-acting element of wheat histone genes. Eur J Biochem 217: 1049–1056 (1993).
Shimamoto K, Terada R, Izawa T, Fujimoto H: Fertile transgenic rice plants regenerated from transformed protoplasts. Nature 338: 274–276 (1989).
Tabata T, Takase H, Takayama S, Mikami K, Nakatsuka A, Kawata T, Nakayama T, Iwabuchi M: A protein that binds to a cis-acting element of wheat histone genes has a leucine zipper motif. Science 245: 965–967 (1989).
Tabata T, Nakayama T, Mikami K, Iwabuchi M: HBP-1a and HBP-1b: leucine zipper-type transcription factors of wheat. EMBO J 6: 1459–1467 (1991).
Terada R, Nakayama T, Iwabuchi M, Shimamoto K: A wheat histone H3 promoter confers cell division-dependent and- independent expression of the gusA gene in transgenic rice plants. Plant J 3: 241–252 (1993).
Wu S-C, Borge L, Vincze E, Kiss GB, Dudits D: Isolation of an alfalfa histone H3 gene: structure and expression. Plant Mol Biol 11: 641–649 (1988).
Wu S-C, Gyorgyey J, Dudits D: Polyadenylated H3 histone transcripts and H3 histone variants in alfalfa. Nucl Acids Res 17: 3057–3063 (1989).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Terada, R., Nakayama, T., Iwabuchi, M. et al. A type I element composed of the hexamer (ACGTCA) and octamer (CGCGGATC) motifs plays a role(s) in meristematic expression of a wheat histone H3 gene in transgenic rice plants. Plant Mol Biol 27, 17–26 (1995). https://doi.org/10.1007/BF00019175
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00019175