Abstract
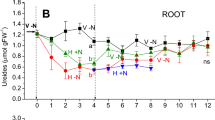
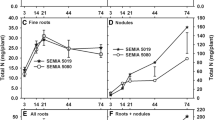
Perennial legume such as alfalfa have the capacity to sustain shoot regrowth and some nodule N2-fixation after removal (‘cutting’) of shoots which contain practically all of the plant's photosynthetic capacity. The role of the roots in supporting these processes has not been fully described. Measurements were made of the nodules' responses to removal of shoots from 8-week-old seedlings in terms of N2-fixation, as nitrogenase activity (NA) measured as acetylene reduction, dark CO2 fixation, measured as in vitro phosphoenolpyruvate carboxylase (PEPC) activity, and total non-structural carbohydrate (NSC) content. These properties decreased and recovered in that sequence, which suggests that nodule NSC supported the substrate requirements of NA and PEPC immediately after cutting. The utilization and redistribution or root carbon and nitrogen, prelabeled with 14C and 15N, were also followed after cutting 8-week-old alfalfa seedlings. In the first 2 weeks of regrowth 12% of root C and 25% of root N were transferred for incorporation into new shoots. Up to 40% of the root C was used for plant respiration to support 28 days of shoot regrowth and N2-fixation. The decline of N2-fixation was slower after cutting and its minimum activity rose up 45% of pre-cut activity as root reserves were built up with plant age. Therefore, the stored reserves of nodulated roots play an important role in support of N2-fixation after cutting.
Similar content being viewed by others
References
Boller B C and Heichel G H 1983 Photosynthate partitioning in relation to N2 fixation capacity of alfalfa. Crop Sci. 23, 655–659.
Cralle H T and Heichel G H 1981 Nitrogen fixation and vegatative regrowth of alfalfa and birdsfoot trefoil after successive harvests or floral debudding. Plant Physiol. 67, 898–905.
Fiedler R and Proksch G 1975 The determination of nitrogen-15 by emission and mass spectrometry in biochemical analysis. Anal. Chem. Acta. 78, 1–62.
Hardy R W F, Holsten R F P, Jackson E K and Burns R C 1968 The acetylene-ethylene assay for nitrogen fixation: Laboratory and field evaluation. Plant Physiol. 43, 1185–1207.
Henson C A and Collins M 1984 Carbon metabolism in alfalfa root nodules: Developmental patterns of host plant enzymes before and after shoot removal. Crop Sci. 24, 727–732.
Hodgkinson K C 1969 The utilization of root organic compounds during regeneration of lucerne. Aust. J. Biol. Sci. 22, 1113–1123.
Macdowall F D H 1983 Kinetis of first-cutting regrowth of alfalfa plants and nitrogenase activity in a controlled environment with and without added nitrate. Can. J. Bot. 61, 2405–2409.
Macdowall F D H and Kristjansson G T 1989 Acetylene-induced decline in acetylene reduction by nodulated roots of alfalfa. Can. J. Bot. 67, 360–364.
Mahin D T and Lofberg R T 1966 A simplified method of sample preparation for determination of tritium, carbon-14, or sulfur-35 in blood or tissue by liquid scintillation counting. Anal. Biochem. 16, 500–509.
Minchin F R, Witty J F, Sheehy J E and Miller M 1983 A major error in the acetylene reduction assay: Decreases in nodular nitrogenase activity under assay conditions. J. Exp. Bot. 34, 641–649.
Pearce R B, Fissel G and Carlson G E 1969 Carbon uptake and distribution before and after defoliation of alfalfa. Crop Sci. 9, 756–759.
Phillips D A, Center D M and Jones M B 1983 Nitrogen turnover and assimilation during regrowth in Trifolium subterraneum L. and Bromus mollis L. Plant Physiol. 71, 472–476.
Reibach P H and Steeter J G 1983 Metabolism of 14C-labeled photosynthate and distribution of enzymes of glucose metabolism in soybean nodules. Plant Physiol. 72, 634–640.
Ryle G J A, Powell C E and Gordon A J 1981 Patterns of 14C-labeled assimilate partitioning in red and white clover during vegetative growth. Ann. Bot. 47, 505–514.
Ryle G J A, Powel C E and Gordon A J 1986 Defoliation in white clover: Nodule metaboism, nodule growth and maintenance, and nitrogenase functioning during growth and regrowth. Ann. Bot. 57, 263–271.
Smith D 1962 Carbohydrate root reserves in alfalfa, red clover and birdsfoot trefoil under several management schedules. Crop Sci. 2, 75–78.
Smith D and Silva J P 1969 Use of carbohydrate and nitrogen root reserves in the regrowth of alfalfa from greenhouse experiments under light and dark conditions. Crop Sci. 9, 464–467.
Smith J H and Marten G C 1970 Foliar regrowth of alfalfa utilizing 14C-labelled carbohydrates stored in roots. Crop Sci. 10, 146–150.
Ta T C, Madowall F D H and Faris M A 1987 Utilization of carbon from photosynthesis and nodule CO2 fixation in the fixation and assimilation of nitrogen by alfalfa root nodules. Can. J. Bot. 65, 2537–2541.
Ta T C and Faris M A 1986 Effects of alfalfa proportions and clipping frequencies on timothy-alfalfa mixtures. II. Nitrogen fixation and transfer. Agron. J. 79, 820–824.
Vance C P, Heichel G H, Barnes D K, Bryan J W and Johnson L E 1979 Nitrogen fixation, nodule development and vegetative growth of alfalfa (Medicago sativa L.) following harvest. Plant Physiol. 64, 1–8.
Vance C P, Stade S and Maxell C A 1983 Alfalfa root nodule carbon dioxide fixation: I. Association with nitrogen fixation and incorporation into amino acids. Plant Physiol. 72, 468–473.
Author information
Authors and Affiliations
Additional information
Contribution No. 1265 from Plant Research Center.
Contribution No. 1265 from Plant Research Center.
Rights and permissions
About this article
Cite this article
Ta, T.C., Macdowall, F.D.H. & Faris, M.A. Utilization of carbon and nitrogen reserves of alfalfa roots in supporting N2-fixation and shoot regrowth. Plant Soil 127, 231–236 (1990). https://doi.org/10.1007/BF00014430
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00014430