Abstract
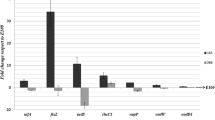
Rhizobitoxine-producing (RT+) strains of Bradyrhizobium japonicum, differing in their abilities to induce foliar chlorosis with ‘Forrest’ soybean (Glycine max [L.] Merr.), were evaluated for effects on short term shoot productivity, nodulation, N2 fixation, and nodule protein production under greenhouse conditions. Soybeans were singly inoculated with washed suspensions of (Group II) USDA strains 31, 46, 76, 94, 110, 123 or 130. Strains USDA 110 and USDA 123 (Group I/Ia) were included as RT-controls. The plants were cultured in the absence of combined N in horticultural-grade vermiculite for 49 days. Beginning 21 days after planting, plants were evaluated weekly for chlorophyll, leaf protein and biomass accumulation, nodular contents of leghemoglobin, soluble protein and RT, and total shoot N content. Rhizobitoxine was detected in nodules of all RT+ strains with the exception of USDA 31. However, only USDA 76 and USDA 94 produced both quantifiable concentrations of RT and symptoms of RT-induced chlorosis. Coincident with moderate to severe chlorosis were reductions in chlorophyll concentrations, shoot and nodule dry weight, leaf protein and total N2 fixation. During extended periods of severe chlorosis, reductions in Lb and soluble nodular protein were observed. Based on carbon accumulation, all non-chlorotic treatments were statistically more productive than the chlorotic treatments. Similarly, non-chlorotic Group II treatments tended to fix less carbon relative to the RT-Group I/Ia controls, although these differences were not statistically significant. The results of this study suggest that, in the absence of discernable foliar chlorosis, the effect of RT+ (Group II) nodulation on short term soybean productivity is minimal.
Similar content being viewed by others
References
Ahmed S and Evans H J 1960 Cobalt: A micronutrient element for the growth of soybean plants under symbiotic conditions. Soil Sci. 90, 205–210.
Allen E K and Allen O N 1958 Biological aspects of symbiotic nitrogen fixation. In Encyclopedia of Plant Physiology. Ed. W Ruhland, Vol 3. pp 48–118. Springer-Verlag, Berlin.
Arnon D I 1949 Copper enzymes in isolated chloroplasts Polyphenoloxidase in Beta vulgaris. Plant Physiol. 24, 1–15.
Bergersen F J, Turner G L and Appleby C A 1973 Studies of the physiological role of leghaemoglobin in soybean root nodules. Biochim. Biophys. Acta 292, 271–282.
Box G E P and Cox D R 1964 An analysis of transformations. J. Roy. Statist. Soc. B 26, 211–252.
Bremner J M 1965 Total nitrogen. In Methods of Soil Analysis, Part 2: Chemical and Microbiological Properties. Ed. C A Black. pp 1149–1178. American Society of Agronomy, Madison, WI.
Devine T E, Kuykendall L D and O'Neill J J 1988 DNA homology group and the identity of bradyrhizobial strains producing rhizobitoxine-induced foliar chlorosis on soybean. Crop Sci. 28, 939–941.
Erdman L W, Johnson H W and Clark F 1956 A bacterial-induced chlorosis in the Lee soybean. Plant Dis. Rep. 40, 646.
Erdman L W, Johnson H W and Clark F 1957 Varietal responses of soybeans to a bacterial-induced chlorosis. Agron. J. 49, 267–271.
Farquhar G D, vonCaemmerer S and Berry J A 1980 A biochemical model of photosynthetic CO2: Assimilation in leaves of C3 species. Planta 149, 78–90.
Fuchsman W H and Appleby C A 1979 Separation and determination of the relative concentrations of the homogeneous components of soybean leghemoglobin by isoelectric focusing. Biochim. Biophys. Acta 597, 314–324.
Fuhrmann J 1990 Symbiotic effectiveness of indigenous soybean bradyrhizobia as related to serological, morphological, rhizobitoxine, and hydrogenase phenotypes. Appl. Environ. Microbiol. 56, 224–229.
Fuhrmann J J and Wollum A G 1989 Symbiotic interactions between soybean and competing strains of Bradyrhizobium japonicum. Plant and Soil 119, 139–145.
Giovanelli J, Owens L D and Mudd S H 1972 β-cystathionase: In vitro inactivation by rhizobitoxine and role of the enzyme in methionine biosynthesis in corn. Plant Physiol. 51, 492–503.
Hartwig V, Boller B and Nosberger H P 1987 Oxygen supply limits nitrogenase activity of clover nodules after defoliation. Ann. Bot. 59, 285–291.
Hollis A B, Kloos W E and Elkan G H 1981 DNA:DNA hybridization studies of Rhizobium japonicum and related Rhizobiaceae. J. Gen. Microbiol. 123, 215–222.
Hunt S, King B J, Canvin D T and Layzell D B 1987 Steady and non-steady-state gas exchange characteristics of soybean nodules in relation to the oxygen diffusion barrier. Plant Physiol. 84, 164–172.
Johnson H W, Means U M and Clark F E 1958 Factors affecting the expression of bacterial-induced chlorosis of soybeans. Agron. J. 50, 571–574.
Johnson H W, Means U M and Clark F E 1959 Response of seedlings to extracts of soybean nodules bearing select strains of Rhizobium japonicum. Nature 163, 308–309.
Keith D D, DeBernardo S and Weigele M 1975 The absolute configuration of rhizobitoxine. Tetrahedron 31, 2629–2632.
Kollman G E, Streeter J G, Jeffers D L and Curry R D 1974 Accumulation and distribution of mineral nutrients, carbohydrates, and dry matter in soybean plants as influenced by reproductive sink size. Agron. J. 66, 549–554.
LaFavre J S, LaFavre A K and Eaglesham A R J 1988 Rhizobitoxine production by and nodulation characteristics of colony-type derivatives of Bradyrhizobium japonicum USDA 76. Can. J. Microbiol. 34, 1017–1022.
Lawn R J and Brun W A 1974 Symbiotic nitrogen fixation in soybeans. I. Effect of photosynthetic source-sink manipulations. Crop Sci. 14, 11–16.
McClure P R and Israel D W 1979 Transport of nitrogen in the xylem of soybean plants. Plant Physiol. 64, 411–416.
Minamisawa K 1989 Comparison of extracellular polysaccharide composition, rhizobitoxine production, and hydrogenase phenotype among various strains of Bradyrhizobium japonicum. Plant Cell Physiol. 30, 877–884.
Minamisawa K 1990 Division of rhizobitoxine-producing and hydrogen-uptake positive strains of Bradyrhizobium japonicum by nifDKE sequence divergence. Plant Cell Physiol. 31, 81–89.
Minamisawa K and Fukai K 1991 Production of indole-3-acetic acid by Bradyrhizobium japonicum: A correlation with genotype grouping and rhizobitoxine production. Plant Cell Physiol, 32, 1–9.
Minamisawa K and Kume N 1987 Determination of rhizobitoxine and dihydrorhizobitoxine in soybean plants by amino acid analyzer. Soil Sci. Plant Nutr. 33, 645–649.
Nash D T and Schulman H M 1976 Leghemoglobins and nitrogenase activity during soybean root nodule development. Can. J. Bot. 54, 2790–2797.
Owens L D and Wright D A 1965 Rhizobial-induced chlorosis in soybeans: Isolation, production in the nodule, and varietal specificity of the toxin. Plant Physiol. 40, 927–930.
Owens L D, Guggenheim S and Hilton J L 1968 Rhizobium-synthesized phytotoxin: An inhibitor of B-cystathionase in Salmonella typhimurium. Biochim. Biophys. Acta 158, 219–225.
Owens L D, Lieberman M and Kunishi A 1971 Inhibition of ethylene production by rhizobitoxine. Plant Physiol. 48, 1–4.
Owens L D, Thompson J F and Williams R G 1972 Structure of rhizobitoxine: An antimetabolic enol-ether amino acid from Rhizobium japonicum. J. C. S. Chem. Com. p. 714.
Paau A S and Cowles J R 1979 Effect of induced nodule senescence on parameters related to dinitrogen-fixation, bacterioid size and nucleic acid content. J. Gen. Microbiol. 111, 101–107.
Peterson G L 1977 A simplification of the protein assay method of Lowery et al. which is more generally applicable. Anal. Biochem. 83, 346–356.
Pfeiffer N E, Torres C M and Wagner F W 1983 Proteolytic activity in soybean root nodules. Plant Physiol. 71, 797–802.
Stanley J, Brown G G and Verma D P S 1985 Slow-growing Rhizobium japonicum comprises two highly divergent symbiotic types. J. Bacteriol. 163, 148–154.
Sutton W D 1983 Nodule development and senescence. In Nitrogen Fixation, Vol. 3: Legumes. Ed. W J Broughton. pp 144–212. Oxford University Press, New York.
Vessey J K, Walsh K B, Layzell D B 1988 Oxygen limitation of N2 fixation in stem-girdled and nitrate treated soybean. Physiol. Plant. 73, 113–121.
Virtanen A I, Erkama J and Linkola H 1947 On the relation between nitrogen fixation and leghaemoglobin content of leguminous root nodules. II. Acta Chem. Scand. 1, 861–870.
VonCaemmerer S and Farquhar G D 1981 Some relationships between the biochemistry of photosynthesis and the gas exchange of leaves. Planta 153, 376–387.
Walsh K B, Canny M J and Layzell D B 1989 Vascular transport and soybean nodule function. II. A role for phloem supply in product export. Plant Cell Environ. 12, 713–723.
Weaver R W and Frederick L R 1982 Rhizobium. In Methods of Soil Analysis, Part 2: Chemical and Microbiological Properties. 2nd ed. Eds. A L Page, D E Baker and R Ellis. pp 1043–1070. American Society of Agronomy, Soil Science Society of America, Madison, WI.
Weber D F, Keyser H H, and Uratsu S L 1989 Serological distribution of Bradyrhizobium japonicum from U.S. soybean production areas. Agron. J. 81, 786–798.
Wittenbach V A, Ackerson R C, Giaquinta R T, and Herbert R R 1980 Changes in photosynthesis, ribulose bisphosphate carboxylase, proteolytic activity and ultrastructure of soybean leaves during senescence. Crop Sci. 20, 225–231.
Author information
Authors and Affiliations
Additional information
Published as Miscellaneous Paper No. 1439 of the Delaware Agricultural Experiment Station.
Published as Miscellaneous Paper No. 1439 of the Delaware Agricultural Experiment Station.
Rights and permissions
About this article
Cite this article
Teaney, G.B., Fuhrmann, J.J. Soybean response to nodulation by bradyrhizobia differing in rhizobitoxine phenotype. Plant Soil 145, 275–285 (1992). https://doi.org/10.1007/BF00010356
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00010356