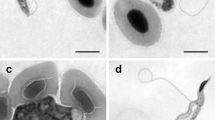
Abstract
Size profiles and antigenic comparisons were made of polypeptides from cultured fish trypanosomes. Twenty, 19 and 18 polypeptides (15–106 kD) were resolved from Trypanosoma phaleri, T. catostomi and Trypanosoma sp., respectively using SDS-PAGE and gel densitometry. Differences between species were also observed in the relative amount of a polypeptide (according to its molecular weight). Most polypeptides in the homologous (PS1 & PS2) and heterologous (LO1 & LO2) clones of T. phaleri were antigenically similar as demonstrated by Western blotting. However antigenicity of 70–75 kD polypeptides differed. In contrast, only a 100 kD polypeptide in Trypanosoma sp. and 85 kD polypeptides in both T. catostomi and Trypanosoma sp. appeared to be antigenically similar to those of T. phaleri. Examination of T. phaleri using the microscopic immuno-substrate-enzyme technique (MISET) suggested that antigenic differences were probably associated with surface antigens. Some limitations of SDS-PAGE and Western blotting as tools in systematics are discussed.
Similar content being viewed by others
References
Anderson, N.L., Parish, N.M., Richardson, J.P. & Pearson T.W. (1985) Comparison of African trypanosomes of different antigenic phenotypes, sub-species and life cycle stages by two dimensional gel electrophoresis. Molecular and Biochemical Parasitology, 16, 299–314.
Bardsley, J.E. & Harmsen, R. (1973) The trypanosomes of Anura. Advances in Parasitology, 11, 1–73.
Birkelund, S. & Andersen, H. (1988) Comparative studies of Mycoplasma antigens and corresponding antibodies. In: Bjerrum, O.J. & Heegaard, N.H.H. (Eds.) Handbook of immunoblotting proteins. Boca Raton, Florida: C.R.C. Press, Vol. 2, pp. 25–33.
Bower, S.M. & Woo, P.T.K. (1982) Immunological comparison of four Trypanosoma spp. (subgenus Schizotrypanum) from bats. Parasitology, 85, 111–114.
Bradford, M.M. (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, 72, 248–254.
Cohen, C. (1962) Blood groups in rabbits. Annals of the New York Academy of Science, 97, 26–36.
Diamond, L.S. (1965) A study of the morphology, biology and taxonomy of the trypanosomes of Anura. Wildlife Diseases, 44, 1–77.
Dwyer, D.M. (1980) Isolation and partial characterization of surface membranes from Leishmania donovani promastigotes. Journal of Protozoology, 27, 176–182.
Dwyer, D.M. & D'Alesandro, P.A. (1980) Isolation and characterization of pellicular membranes from Trypanosoma lewisi bloodstream forms. Journal of Parasitology, 66, 377–389.
Fong, D. & Cheng, K.-P. (1981) Tubulin biosynthesis in the developmental cycle of a parasitic protozoan, Leishmania mexicana, changes during differentiation of motile and nonmotile stages. Proceedings of the National Academy of Sciences of the United States of America, 78, 7624–7628.
Godfrey, D.G. (1978) Identification of economically important parasites. Nature, 273, 600–604.
Johansson, K.-E. (1988) Separation of antigens by analytical gel electrophoresis. In: Bjerrum, O.J. & Heegaard, N.H.H. (Eds.) Handbook of immunoblotting protein. Boca Raton, Florida: C.R.C. Press, Vol. 1, pp. 31–50.
Jones, S.R.M. & Woo, P.T.K. (1990) The biology of Trypanosoma phaleri n. sp. from bowfin, Amia calva L. in Canada and the United States. Canadian Journal of Zoology, 68, 1956–1961.
Jones, S.R.M. & Woo, P.T.K. (1991a) Development and infectivity of Trypanosoma phaleri in leech and fish hosts. Canadian Journal of Zoology, 69, 1522–1529.
Jones, S.R.M. & Woo, P.T.K. (1991b) Culture characteristics of Trypanosoma catostomi and Trypanosoma phaleri from North American freshwater fishes. Parasitology, 103, 237–243.
Jones, S.R.M. & Woo, P.T.K. (1992) Vector specificity of Trypanosoma catostomi and its infectivity to freshwater fishes. Journal of Parasitology, 78, 87–92.
Laemmli, U.K. (1970) Cleavage of structural proteins during the assembly of the lead of bacteriophage T4. Nature, 227, 680–685.
Letch, C.A. (1979) Host restriction, morphology and isoenzymes among trypanosomes of some British freshwater fishes. Parasitology, 79, 107–117.
Lom, J. (1979) Biology of the trypanosomes and trypanoplasms of fish. In: Lumsden, W.H.R. & Evans, D.A. (Eds) Biology of the Kinetoplastida, London: Academic Press, Vol. 2, pp. 269–337.
Majiwa, P.A.O., Hamers, R., vanMeirvenne, N. & Matthyssens G. (1986) Evidence for genetic diversity in Trypanosoma (Nannomonas) congolense. Parasitology, 93, 291–304.
Ramlau, J. (1988) Polyclonal antibodies as primary reagent in immunoblotting-uses and limitations. In: Bjerrum, O.J. & Heegaard, N.H.H. (Eds) Handbook of immunoblotting proteins, Boca Raton, Florida: C.R.C. Press, Vol. 1, pp. 151–158.
Seebeck, T., Hemphill, A. & Lawson, D. (1990) The cytoskeleton of trypanosomes. Parasitology Today, 6, 49–52.
Taylor, A.E.R., Edwards, Y.H., Smith, V., Baker, J.R., Woo, P.T.K., Lanham, S.M. & Pennick, N.C. (1982) Trypanosoma (Schizotrypanum) species from insectivorous bats (Microchiroptera), characterization by polypeptide profiles. Systematic Parasitology, 4, 155–168.
Towbin, H., Staehelin, T. & Bordon, J. (1979) Electrophoretic transfer of protein from polyacrylamide gels to nitrocellulose sheets, procedure and some applications. Proceedings of the National Academy of Sciences of the United States of America, 76, 4350–4354.
Woo, P.T.K. (1969a) The haematocrit centrifuge for the detection of trypanosomes. Canadian Journal of Zoology, 47, 921–923.
Woo, P.T.K. (1969b) The life cycle of Trypanosoma chrysemydis. Canadian Journal of Zoology, 49, 1139–1151.
Woo, P.T.K. (1979) Trypanoplasma salmositica: experimental infections in rainbow trout, Salmo gairdneri. Experimental Parasitology, 47, 36–48.
Woo, P.T.K. (1990) MISET: An immunological technique for the serodiagnosis of Cryptobia salmositica (Sarcomastigophora: Kinetoplastida) infection in Oncorhynchus mykiss. Journal of Parasitology, 76, 389–393.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jones, S.R.M., Woo, P.T.K. Antigenic characterisation of cultured trypanosomes isolated from three species of fishes. Syst Parasitol 23, 43–50 (1992). https://doi.org/10.1007/BF00008008
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00008008