Abstract
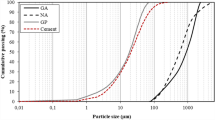
Porous calcium hexa-aluminate (CA6) ceramics were synthesized by a molten salt-mediated technique using CaCO3 and α-Al2O3 powders as the main raw materials. The effects of salt type and content, along with firing temperature on phase formation, microstructural evolution, pore generation and size distribution, and mechanical strength of fired samples were investigated and compared. Among the three tested salts (KCl, NaCl, and CaCl2), KCl showed the best whereas CaCl2 showed the worst accelerating effect on CA6 formation. In the case of the former, phase-pure CA6 was formed after 3 h at 1500 °C, whereas in the case of the latter, CA2 was formed instead as a main phase. The salt not only acted as a reaction medium, accelerating CA6 formation, but also acted as a pore former, creating high levels of porosity in the fired samples. The apparent porosity of the fired samples clearly increased with an increase in KCl content. But the majority of pores were still micron sized and the pore size distribution was relatively narrow. This, along with the possible toughening effect from the well-developed plate-like CA6, conferred relatively high mechanical strength on the final highly porous CA6 ceramics.








Similar content being viewed by others
References
Chen, J.H., Chen, H.-y., Yan, M.W., Cao, Z., Mi, W.J.: Formation mechanism of calcium hexaluminate. Int. J. Min. Metall. & Mater. 23 (2016) [10] 1225–1230
de la Iglesia, P.G., García-Moreno, O. Torrecillas,,R., Menéndez, J.L.: Influence of different parameters on calcium hexaluminate reaction sintering by spark plasma. Ceram. Int. 38 (2012) [7] 5325–5332
An, L., Chan, H.M., Soni, K.K.: Control of calcium hexaluminate grain morphology in in-situ toughened ceramic composites. J. Mater. Sci. 31 (1996) [12] 3223–3229
Domínguez, C., Chevalier, J., Torrecillas, R., Fantozzi, G.: Microstructure development in calcium hexaluminate. J. Eur. Ceram. Soc. 21 (2001) [3] 381–387
Domínguez, C., Chevalier, J., Torrecillas, R., Gremillard, L., Fantozzi, G.: Thermomechanical properties and fracture mechanisms of calcium hexaluminate. J. Eur. Ceram. Soc. 21 (2001) [7] 907–917
An, L., Chan, H.M.: R-curve behavior of in-situ-toughened Al2O3:CaAl12O19 ceramic composites. J. Am. Ceram. Soc. 79 (1996) [12] 3142–3148
Vishista, K., Gnanam, F.D., Awaji, H.: Sol-gel synthesis and characterization of alumina-calcium hexaaluminate composites. J. Am. Ceram. Soc. 88 (2005) [5] 1175–1179
Buhr A., Büchel, G., Aroni, J.M., Racher, R.P.: Bonite - a new raw material alternative for refractory innovations. Proc. 47th Int. Colloquium on Refractories (Aachen, Germany) 40 (2006) 205–210
Pieta, A., Bucko, M.M., Janus, M. Lis, J., Jonas, S.: Calcium hexaaluminate synthesis and its influence on the properties of CA2-Al2O3-based refractories. J. Eur. Ceram. Soc. 35 (2015) 4567–4571
Ianoş, R., Lazău, I., Păcurariu, C., Barvinschi, P.: Peculiarities of CaO·6Al2O3 formation by using low-temperature combustion synthesis. Eur. J. Inorg. Chem. 6 (2008) 925–930
Chandradass, J., Bae, D.S., Kim, K.H.: Synthesis of calcium hexaaluminate (CaAl12O19) via reverse micelle process. J. Non-Crystall. Sol. 355 (2009) [48–49] 2429–2432
Tulliani, J.M., Pagès, G., Fantozzi, G., Montanaro, L.: Dilatometry as a tool to study a new synthesis for calcium hexaluminate. J. Therm. Analysis & Calorimetry 72 (2003) 1135–1140
Altay, A., Carter, C.B., Arslan, I., Gülgün, M.A.: Crystallization of CaAl4O7 and CaAl12O19 powders. Philos. Mag. 89 (2009) [7] 605–621
Dong, B., Yuan, B., Wang, G., Chen, K., Han, J., Li, H.: Fabrication of porous SiC/Calcium hexaluminate composites. J. Eur. Ceram. Soc. 36 (2016) 3889–3893
Colombo, P.: Conventional and novel processing methods for cellular ceramics. Philos. Trans. Royal Soc. A: Math., Phys. and Eng. Sci. 364 (2006) [1838] 109–124
Peng, H.X., Fan, Z., Evans, J.R.G., Busfield, J.J.C.: Microstructure of ceramic foams. J. Eur. Ceram. Soc. 20 (2000) [7] 807–813
Singh, V., Chakradhar, R.P.S., Rao, J.L., Kim, D.-K.: Photoluminescence and EPR studies of Cr-doped hibonite (CaAl12O19) phosphors. Solid State Sci. 10 (2008) [11] 1525–1532
Nagaoka, T., Tsugoshi, T., Hotta, Y., Yasuoka, M., Watari, K.: Forming and sintering of porous calcium-hexaaluminate ceramics with hydraulic alumina. J. Materials Science 41 (2006) [22] 7401–7405
Salomão, R., Ferreira, V.L., de Oliveira, I.R., Souza, A.D.V., Correr, W.R.: Mechanism of pore generation in calcium hexaluminate (CA6) ceramics formed in situ from calcined alumina and calcium carbonate aggregates. J. Eur. Ceram. Soc. 36 (2016) 4225–4235
Salomão, R., Ferreira, V.L., Costa, L.M.M., de Oliveira, I.R.: Effects of the initial CaO-Al2O3 ratio on the microstructure development and mechanical properties of porous calcium hexaluminate. Ceram. Int. 44 (2018) 2626–2631
Kawaguchia, K., Suzukia, Y., Gotoc, T., Choc, S.H., Sekinoc, T.: Homogeneously bulk porous calcium hexaaluminate (CaAl12O19): Reactive sintering and microstructure development. Ceram. Int. 44 (2018) 4462–4466
Li, Y., Xiang, R., Xu, N., Wang, Q., Li, S., Wu, M., Yang, C.: Fabrication of calcium hexaluminate-based porous ceramic with microsilica addition. Int J Appl Ceram Technol. 15 (2018) 1054–1059
Feng, Y., Wang, K., Yao, J., Webley, P.A., Smart, S., Wang, H.: Effect of the addition of polyvinylpyrrolidone as a pore-former on microstructure and mechanical strength of porous alumina ceramics. Ceram. Int. 39 (2013) [7] 7551–7556
Gregorová, E., Pabst, W.: Process control and optimized preparation of porous alumina ceramics by starch consolidation casting. J. Eur. Ceram. Soc. 31 (2011) [12] 2073–2081
Li, Z., Zhang, S., Lee, W.E.: Molten salt synthesis of LaAlO3 powder at low temperatures. J. Eur. Ceram. Soc. 27 (2007) [10] 3201–3205
Ye, J., Zhang, S., Lee, W.E.: Molten salt synthesis and characterization of SiC coated carbon black particles for refractory castable applications. J. Eur. Ceram. Soc. 33 (2013) [10] 2023–2029
Zhang, S., Jayaseelan, D.D., Bhattacharya, G., Lee, W.E.: Molten Salt Synthesis of magnesium aluminate (MgAl2O4) spinel powder. J. Am. Ceram. Soc. 89 (2006) [5] 1724–1726
Liu, X., Yang, D., Huang, Z., Yi, S., Ding, H., Fang, M., Zhang, S., Liu, Y.-g.: Novel synthesis method and characterization of porous calcium hexaaluminate ceramics. J. Am. Ceram. Soc. 97 (2014) [9] 2702–2704
Wang, S., Zhang, F., Liu, X., Zhang, L.: CaO solubility and activity coefficient in molten salts CaCl2-x (x = 0, NaCl, KCl, SrCl2, BaCl2 and LiCl). Thermochimica Acta 470 (2008) 105–107
Acknowledgment
This work was financially supported by the National Natural Science Foundation of China (Grant no. 51032007), the Fundamental Research Funds for the Central Universities (Grant no. 2012067), and the Doctoral Scientific Research Foundation of Quzhou University (Grant no. BSYJ201707).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, X., Liu, Yg., Huang, Z. et al. Preparation of High-Quality Porous Calcium Hexa-Aluminate Ceramics using Salt as Both Reaction Medium and Pore-Former. Interceram. - Int. Ceram. Rev. 67 (Suppl 1), 50–57 (2018). https://doi.org/10.1007/s42411-018-0045-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42411-018-0045-9