Abstract
Background
Sarcopenia is a major health issue especially in patients on maintenance hemodialysis. Low skeletal muscle mass is included in the diagnostic criteria for sarcopenia. The skeletal muscle mass is usually evaluated by modalities such as bioimpedance analysis (BIA) or dual-energy X-ray absorptiometry, however the assessment of skeletal muscle mass using computed tomography (CT) images has not been established. The purpose of the study was to investigate the feasibility of the assessment of skeletal muscle mass using CT images in hemodialysis patients.
Methods
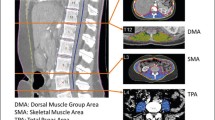
Skeletal muscle mass index (SMI) was measured by BIA and psoas muscle index (PMI) was measured by cross-sectional CT images in 131 patients. The relationship between SMI and PMI and the diagnostic ability of PMI for low muscle mass were evaluated. Furthermore, the patients were followed up and long-term survival in patients with low and high PMI were compared.
Results
PMI measured at the L3 vertebral level was strongly correlated with SMI (r = 0.597, p < 0.001). Age, sex, and SMI were the influencing factors for PMI. Patients with low PMI showed higher incidence rates of mortality during the follow up.
Conclusions
PMI assessed by CT image can be an alternative to BIA in patients on hemodialysis.




Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Harris R, Chang Y, Beavers K et al (2017) Risk of fracture in women with sarcopenia, low bone mass, or both. J Am Geriatr Soc 65:2673–2678. https://doi.org/10.1111/jgs.15050
Tolea MI, Galvin JE (2015) Sarcopenia and impairment in cognitive and physical performance. Clin Interv Aging 10:663–671. https://doi.org/10.2147/CIA.S76275
Atkins JL, Whincup PH, Morris RW et al (2014) Sarcopenic obesity and risk of cardiovascular disease and mortality: a population-based cohort study of older men. J Am Geriatr Soc 62:253–260. https://doi.org/10.1111/jgs.12652
Yoshida D, Suzuki T, Shimada H et al (2014) Using two different algorithms to determine the prevalence of sarcopenia. Geriatr Gerontol Int 14:46–51. https://doi.org/10.1111/ggi.12210
Yoshimura N, Muraki S, Oka H et al (2017) Is osteoporosis a predictor for future sarcopenia or vice versa? Four-year observations between the second and third ROAD study surveys. Osteoporos Int 28:189–199. https://doi.org/10.1007/s00198-016-3823-0
Moon SJ, Kim TH, Yoon SY et al (2015) Relationship between stage of chronic kidney disease and sarcopenia in Korean aged 40 years and older using the Korea National Health and Nutrition Examination Surveys (KNHANES IV-2, 3, and V-1, 2), 2008–2011. PLoS ONE 10:1–11. https://doi.org/10.1371/journal.pone.0130740
Ren H, Gong D, Jia F et al (2016) Sarcopenia in patients undergoing maintenance hemodialysis: Incidence rate, risk factors and its effect on survival risk. Ren Fail 38:364–371. https://doi.org/10.3109/0886022X.2015.1132173
Isoyama N, Qureshi AR, Avesani CM et al (2014) Comparative associations of muscle mass and muscle strength with mortality in dialysis patients. Clin J Am Soc Nephrol 9:1720–1728. https://doi.org/10.2215/CJN.10261013
Chen L, Liu L, Woo J et al (2014) Sarcopenia in Asia: Consensus Report of the Asian Working Group for Sarcopenia. J Am Med Dir Assoc 15:95–101
Cruz-Jentoft AJ, Bahat G, Bauer J et al (2019) Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 48:16–31. https://doi.org/10.1093/ageing/afy169
Durand F, Buyse S, Francoz C et al (2014) Prognostic value of muscle atrophy in cirrhosis using psoas muscle thickness on computed tomography. J Hepatol 60:1151–1157. https://doi.org/10.1016/j.jhep.2014.02.026
Hamaguchi Y, Kaido T, Okumura S et al (2016) Proposal for new diagnostic criteria for low skeletal muscle mass based on computed tomography imaging in Asian adults. Nutrition 32:1200–1205. https://doi.org/10.1016/j.nut.2016.04.003
Caan BJ, Cespedes Feliciano EM, Prado CM et al (2018) Association of muscle and adiposity measured by computed tomography with survival in patients with nonmetastatic breast cancer. JAMA Oncol 4:798–804. https://doi.org/10.1001/jamaoncol.2018.0137
Masuda T, Shirabe K, Ikegami T et al (2014) Sarcopenia is a prognostic factor in living donor liver transplantation. Liver Transplant 20:401–407. https://doi.org/10.1002/lt.23811
Linder N, Schaudinn A, Langenhan K et al (2019) Power of computed-tomography-defined sarcopenia for prediction of morbidity after pancreaticoduodenectomy. BMC Med Imaging 19:1–10. https://doi.org/10.1186/s12880-019-0332-6
Giglio J, Kamimura MA, Souza NC et al (2019) Muscle mass assessment by computed tomography in chronic kidney disease patients: agreement with surrogate methods. Eur J Clin Nutr 73:46–53. https://doi.org/10.1038/s41430-018-0130-1
Schweitzer L, Geisler C, Pourhassan M et al (2015) What is the best reference site for a single MRI slice to assess whole body skeletal muscle and adipose tissue volumes in healthy adults? Am J Clin Nutr 102:58–65. https://doi.org/10.3945/ajcn.115.111203
Morley JE, Anker SD, von Haehling S (2014) Prevalence, incidence, and clinical impact of sarcopenia: facts, numbers, and epidemiology—update 2014. J Cachexia Sarcopenia Muscle 5:253–259. https://doi.org/10.1007/s13539-014-0161-y
Tanimoto Y, Watanabe M, Kono R et al (2010) Changes in muscle mass with aging in Japanese. Nihon Ronen Igakkai Zasshi (in Japanese) 47:52–57
Tandon P, Ney M, Irwin I et al (2012) Severe muscle depletion in patients on the liver transplant wait list: prevalence and independent prognostic value. Liver Transpl 18:1209–1216. https://doi.org/10.1002/lt
Carrero JJ, Johansen KL, Lindholm B et al (2016) Screening for muscle wasting and dysfunction in patients with chronic kidney disease. Kidney Int 90:53–66. https://doi.org/10.1016/j.kint.2016.02.025
Liakopoulos V, Eleftheriadis T, Mertens PR (2018) Screening for renal cell carcinoma in dialysis patients. Kidney Int 94:1238. https://doi.org/10.1016/j.kint.2018.08.032
Sabatino A, Regolisti G, di Mario F et al (2020) Validation by CT scan of quadriceps muscle thickness measurement by ultrasound in acute kidney injury. J Nephrol 33:109–117. https://doi.org/10.1007/s40620-019-00659-2
Chang KV, Wu WT, Huang KC, Han DS (2019) Segmental body composition transitions in stroke patients: trunks are different from extremities and strokes are as important as hemiparesis. Clin Nutr 39:1968–1973. https://doi.org/10.1016/j.clnu.2019.08.024
Author information
Authors and Affiliations
Contributions
Conceptualization: TT, KY, AI, MO, SH, MY, YM, and TI; data acquisition; TT, AM, and KT; formal analysis and investigation; TT, ST: writing—original draft preparation; TT: writing—review and editing: MT, AN: supervision; HI.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study was conducted retrospectively from data obtained for clinical purposes. This study was approved by the ethical committee of the Tottori University Hospital (approval number: 19A222), and was conducted according to the declaration of Helsinki.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
40620_2020_871_MOESM1_ESM.tif
Supplementary file1 Scheme of the assessment of muscle mass. Representative computed tomography images from patients with high and low PMI are shown (TIF 509 kb)
Rights and permissions
About this article
Cite this article
Takata, T., Motoe, A., Tanida, K. et al. Feasibility of computed tomography-based assessment of skeletal muscle mass in hemodialysis patients. J Nephrol 34, 465–471 (2021). https://doi.org/10.1007/s40620-020-00871-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40620-020-00871-5