Abstract
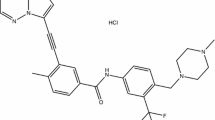
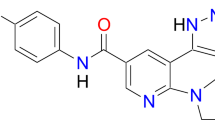
Asciminib (Scemblix®) is an orally administered, small molecule, selective allosteric inhibitor that targets the myristoyl pocket of the BCR-ABL1 tyrosine kinase and is being developed by Novartis for the treatment of haematological malignancies, including Philadelphia chromosome-positive (Ph+) chronic myeloid leukaemia (CML). The drug is active against a number of the single catalytic-site mutations, such as T315I, that confer resistance to conventional tyrosine kinase inhibitors (TKIs) that bind to the ATP-binding site of BCR-ABL1. In October 2021, asciminib monotherapy was granted accelerated approval for the treatment of adults with Ph+ CML in chronic phase (CML-CP), previously treated with ≥ 2 TKIs, and full approval for the treatment of adults with Ph+ CML-CP with the T315I mutation. The drug is under regulatory review for use as monotherapy in CML in the EU, and is in phase 1–3 development exploring its potential in first-line, later-line and paediatric patients with CML. This article summarizes the milestones in the development of asciminib leading to this first approval for the treatment of adults with Ph+ CML-CP, previously treated with ≥ 2 TKIs, and Ph+ CML-CP with the T315I mutation.
Similar content being viewed by others
References
Jabbour E, Kantarjian H. Chronic myeloid leukemia: 2020 update on diagnosis, therapy and monitoring. Am J Hematol. 2020;95(6):691–709.
Hochhaus A, Saussele S, Rosti G, et al. Chronic myeloid leukaemia: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2017;28(Suppl 4):iv41–51.
Quintas-Cardama A, Cortes J. Molecular biology of bcr-abl1-positive chronic myeloid leukemia. Blood. 2009;113(8):1619–30.
Wylie AA, Schoepfer J, Jahnke W, et al. The allosteric inhibitor ABL001 enables dual targeting of BCR-ABL1. Nature. 2017;543(7647):733–7.
Schoepfer J, Jahnke W, Berellini G, et al. Discovery of asciminib (ABL001), an allosteric inhibitor of the tyrosine kinase activity of BCR-ABL1. J Med Chem. 2018;61(18):8120–35.
Novartis. SCEMBLIX (asciminib) tablets, for oral use: US Prescribing information. 2021. https://www.accessdata.fda. Accessed 10 Nov 2021.
Novartis. FDA approves Novartis Scemblix® (asciminib), with novel mechanism of action for the treatment of chronic myeloid leukemia [media release]. 29 Oct 2021. https://www.novartis.com.
Schuld P, Grzesiek S, Schlotte J, et al. Structural and biochemical studies confirming the mechanism of action of asciminib, an agent specifically targeting the ABL myristoyl pocket (STAMP) [abstract]. Blood. 2020;136(Suppl 1):34–5.
Cortes J. Asciminib for the treatment of patients with chronic myeloid leukemia. Clin Adv Hematol Oncol. 2021;19(4):207–8.
Eide CA, Zabriskie MS, Savage Stevens SL, et al. Combining the allosteric inhibitor asciminib with ponatinib suppresses emergence of and restores efficacy against highly resistant BCR-ABL1 mutants. Cancer Cell. 2019;36(4):431-43.e5.
Qiang W, Antelope O, Zabriskie MS, et al. Mechanisms of resistance to the BCR-ABL1 allosteric inhibitor asciminib. Leukemia. 2017;31(12):2844–7.
Eadie LN, Saunders VA, Branford S, et al. The new allosteric inhibitor asciminib is susceptible to resistance mediated by ABCB1 and ABCG2 overexpression in vitro. Oncotarget. 2018;9(17):13423–37.
Gleixner KV, Filik Y, Berger D, et al. Asciminib and ponatinib exert synergistic anti-neoplastic effects on CML cells expressing BCR-ABL1 (T315I)-compound mutations. Am J Cancer Res. 2021;11(9):4470–84.
Malyukova A, Ujvari D, Yektaei-Karin E, et al. Combination of tyrosine kinase inhibitors and the MCL1 inhibitor S63845 exerts synergistic antitumorigenic effects on CML cells. Cell Death Dis. 2021;12(10):875.
Yurttas NO, Eskazan AE. Tyrosine kinase inhibitor-associated platelet dysfunction: does this need to have a significant clinical impact? Clin Appl Thromb Hemost. 2019. https://doi.org/10.1177/1076029619866925.
Nesr G, Laffan M, Claudiani S, et al. Platelet function in patients with chronic myeloid leukemia treated with asciminib. Leuk Lymphoma. 2020;61(12):3021–3.
Hoch M, Zack J, Quinlan M, et al. Pharmacokinetics of asciminib when taken with imatinib or with food. Clin Pharmacol Drug Dev. 2021. https://doi.org/10.1002/cpdd.1019.
Tran P, Hanna I, Eggimann FK, et al. Disposition of asciminib, a potent BCR-ABL1 tyrosine kinase inhibitor, in healthy male subjects. Xenobiotica. 2020;50(2):150–69.
Hoch M, Sato M, Zack J, et al. Pharmacokinetics of asciminib in individuals with hepatic or renal impairment. J Clin Pharmacol. 2021;61(11):1454–65.
Combes FP, Li YF, Sy SKB, et al. Dose justification for asciminibin patients with Ph+ chronic myeloid leukemia in chronic phase with or without the T315I mutation previously treated with two or more tyrosine kinase inhibitors [abstract no. T6107V]. In: American Association of Pharmaceutical Scientists PHARMSCI 360. 2021.
Rea D, Mauro MJ, Boquimpani C, et al. A phase 3, open-label, randomized study of asciminib, a STAMP inhibitor, vs bosutinib in CML after 2 or more prior TKIs. Blood. 2021;21:2031–41.
Mauro M, Minami H, Rea D, et al. Efficacy and safety results from ASCEMBL, a multicenter, open-label, phase 3 study of asciminib, a first-in-class STAMP inhibitor, vs bosutinib in patients with chronic myeloid leukemia in chronic phase after ≥2 prior tyrosine kinase inhibitors: update after 48 weeks [abstract no. 310]. In: American Society of Hematology Annual Meeting & Exhibition. 2021.
Mauro MJ, Boquimpani C, Rea D, et al. ASCEMBL phase 3 health-related quality of life (HRQL) comparison of asciminib and bosutinib for patients with chronic myelogenous leukemia in chronic phase (CML-CP), previously treated with 2 or more tyrosine kinase inhibitors (TKI) [abstract no. CML-417]. Clin Lymphoma Myeloma Leuk. 2021;21(Suppl 1):S336.
Hughes TP, Mauro MJ, Cortes JE, et al. Asciminib in chronic myeloid leukemia after ABL kinase inhibitor failure. N Engl J Med. 2019;381(24):2315–26.
Hughes T, Mauro MJ, Kim DW, et al. Asciminib in heavily pretreated patients (PTS) with philadelphia chromosome-positive (PH+) chronic myeloid leukemia in chronic phase (CML-CP) sensitive to tyrosine kinase inhibitor (TKI) therapy [abstract no. S170]. HemaSphere. 2020;4(Suppl 1):39–40.
Cortes JE, Hughes TP, Mauro MJ, et al. Asciminib, a first-in-class STAMP inhibitor, provides durable molecular response in patients (pts) with chronic myeloid leukemia (CML) harboring the T315I mutation: primary efficacy and safety results from a phase 1 trial [abstract]. Blood. 2020;136(Suppl 1):47–50.
Talpaz M, Cortes J, Lang F, et al. Combination of asciminib, a novel and specific BCR-ABL1 inhibitor, plus imatinib in previously treated chronic myeloid leukemia (CML) patients: phase 1 study results [abstract no. CML-041]. Clin Lymphoma Myeloma Leuk. 2019;19(Suppl 1):S287–8.
DeAngelo DJ, Mauro MJ, Kim DW, et al. Combination of asciminib+nilotinib or asciminib+dasatinib in previously treated chronic myeloid leukemia (CML) patients: phase 1 study results [abstract no. CML-102]. Clin Lymphoma Myeloma Leuk. 2019;19(Suppl 1):S290–1.
Luskin M, Murakami MA, Stevenson KE, et al. A phase I study of asciminib (ABL001) in combination with dasatinib and prednisone for untreated BCR-ABL1-positive ALL in older adults [abstract]. Blood. 2019;134(Suppl 1):3879.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The preparation of this review was not supported by any external funding.
Authorship and Conflict of interest
During the peer review process the manufacturer of the agent under review was offered an opportunity to comment on the article. Changes resulting from any comments received were made by the authors on the basis of scientific completeness and accuracy. Emma Deeks is a contracted employee of Adis International Ltd/Springer Nature, and declares no relevant conflicts of interest. All authors contributed to the review and are responsible for the article content.
Ethics approval, Consent to participate, Consent to publish, Availability of data and material, Code availability
Not applicable.
Additional information
This profile has been extracted and modified from the AdisInsight database. AdisInsight tracks drug development worldwide through the entire development process, from discovery, through pre-clinical and clinical studies to market launch and beyond.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Deeks, E.D. Asciminib: First Approval. Drugs 82, 219–226 (2022). https://doi.org/10.1007/s40265-021-01662-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40265-021-01662-3