Abstract
Background and Objectives
A new once-daily formulation of tacrolimus (Envarsus®) has recently been developed, with alleged different pharmacokinetics from previous tacrolimus formulations. The objectives of this study were to develop population pharmacokinetic models and Bayesian estimators based on limited sampling strategies for Envarsus® in kidney and liver transplant recipients.
Materials and Methods
Full tacrolimus concentration-time profiles (13 samples) were drawn from 57 liver (113 profiles) and 49 kidney (97 profiles) graft recipients transplanted for at least 6 months and switched from Prograf® to Envarsus®. The two databases were split into a development (75%) and a validation (25%) dataset. Pharmacokinetic models characterised by a single compartment with first-order elimination and absorption in two phases described by a sum of two gamma distributions were developed using non-parametric (Pmetrics) and parametric (ITSIM) approaches in parallel. The best limited sampling strategy for each patient group was determined using the multiple model optimal algorithm. The performance of the models and derived Bayesian estimators was evaluated in the validation set.
Results
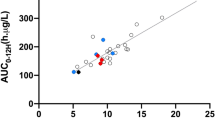
The best limited sampling strategy was 0, 8 and 12 h post-dose, leading to a relative bias ± standard deviation (root-mean-square error) between observed and modelled inter-dose area under the curve in the validation dataset of: 0.32 ± 6.86% (6.87%) for ITSIM and 3.4 ± 13.4% (13.2%) for Pmetrics in kidney transplantation; and 0.89 ± 7.32% (7.38%) for ITSIM and −2.62 ± 8.65% (8.89%) for Pmetrics in liver transplantation.
Conclusion
Population pharmacokinetic models and Bayesian estimators for Envarsus® in kidney and liver transplantation were developed and are now available online for area under the curve-based tacrolimus dose adjustment.




Similar content being viewed by others
References
Venkataramanan R, Swaminathan A, Prasad T, et al. Clinical pharmacokinetics of tacrolimus. Clin Pharmacokinet. 1995;29:404–30.
Laskow DA, Vincenti F, Neylan JF, et al. An open-label, concentration-ranging trial of FK506 in primary kidney transplantation: a report of the United States Multicenter FK506 Kidney Transplant Group. Transplantation. 1996;62:900–5.
Wallemacq P, Armstrong VW, Brunet M, et al. Opportunities to optimize tacrolimus therapy in solid organ transplantation: report of the European consensus conference. Ther Drug Monit. 2009;31:139–52.
Neely MN, van Guilder MG, Yamada WM, et al. Accurate detection of outliers and subpopulations with Pmetrics, a nonparametric and parametric pharmacometric modeling and simulation package for R. Ther Drug Monit. 2012;34:467–76.
Staatz CE, Willis C, Taylor PJ, Tett SE. Population pharmacokinetics of tacrolimus in adult kidney transplant recipients. Clin Pharmacol Ther. 2002;72:660–9.
Staatz CE, Tett SE. Clinical pharmacokinetics of once-daily tacrolimus in solid-organ transplant patients. Clin Pharmacokinet. 2015;54:993–1025.
Gaber AO, Alloway RR, Bodziak K, et al. Conversion from twice-daily tacrolimus capsules to once-daily extended-release tacrolimus (LCPT): a phase 2 trial of stable renal transplant recipients. Transplantation. 2013;96:191–7.
Alloway RR, Eckhoff DE, Washburn WK, Teperman LW. Conversion from twice daily tacrolimus capsules to once daily extended-release tacrolimus (LCP-Tacro): phase 2 trial of stable liver transplant recipients. Liver Transpl. 2014;20:564–75.
Tremblay S, Nigro V, Weinberg J, et al. A Steady-State Head-to-Head Pharmacokinetic Comparison of All FK-506 (Tacrolimus) Formulations (ASTCOFF): an open-label, prospective, randomized, two-arm, three-period crossover study. Am J Transplant. 2017;17(2):432–42.
Saint-Marcoux F, Woillard J-B, Jurado C, Marquet P. Lessons from routine dose adjustment of tacrolimus in renal transplant patients based on global exposure. Ther Drug Monit. 2013;35:322–7.
Prémaud A, Debord J, Rousseau A, et al. A double absorption-phase model adequately describes mycophenolic acid plasma profiles in de novo renal transplant recipients given oral mycophenolate mofetil. Clin Pharmacokinet. 2005;44:837–47.
Saint-Marcoux F, Knoop C, Debord J, et al. Pharmacokinetic study of tacrolimus in cystic fibrosis and non-cystic fibrosis lung transplant patients and design of Bayesian estimators using limited sampling strategies. Clin Pharmacokinet. 2005;44:1317–28.
Saint-Marcoux F, Debord J, Undre N, et al. Pharmacokinetic modeling and development of Bayesian estimators in kidney transplant patients receiving the tacrolimus once-daily formulation. Ther Drug Monit. 2010;32:129–35.
Bayard DS, Neely M. Experiment design for nonparametric models based on minimizing Bayes Risk: application to voriconazole. J Pharmacokinet Pharmacodyn. 2016. doi:10.1007/s10928-016-9498-5
Bustad A, Terziivanov D, Leary R, et al. Parametric and nonparametric population methods: their comparative performance in analysing a clinical dataset and two Monte Carlo simulation studies. Clin Pharmacokinet. 2006;45:365–83.
Prémaud A, Weber LT, Tönshoff B, et al. Population pharmacokinetics of mycophenolic acid in pediatric renal transplant patients using parametric and nonparametric approaches. Pharmacol Res. 2011;63:216–24.
Woillard J-B, Saint-Marcoux F, Monchaud C, et al. Mycophenolic mofetil optimized pharmacokinetic modelling, and exposure-effect associations in adult heart transplant recipients. Pharmacol Res. 2015;99:308–15.
Woillard J-B, Lebreton V, Neely M, et al. Pharmacokinetic tools for the dose adjustment of ciclosporin in haematopoietic stem cell transplant patients. Br J Clin Pharmacol. 2014;78:836–46.
Koop DR, Bleyle LA, Munar M, et al. Analysis of tacrolimus and creatinine from a single dried blood spot using liquid chromatography tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci. 2013;926:54–61.
Hesselink DA, Bouamar R, Elens L, et al. The role of pharmacogenetics in the disposition of and response to tacrolimus in solid organ transplantation. Clin Pharmacokinet. 2014;53:123–39.
Acknowledgements
The authors gratefully thank Mrs. Karen Poole for manuscript editing, and Veloxis and Chiesi for providing the phase II pharmacokinetic data and for funding this pharmacokinetic analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
J.-B. Woillard have no conflict of interest. J. Debord have no conflict of interest. C. Monchaud have no conflict of interest. F. S. Marcoux have no conflict of interest. Pierre Marquet has been a member of scientific boards for, and received funding for this study from, Chiesi France.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Woillard, JB., Debord, J., Monchaud, C. et al. Population Pharmacokinetics and Bayesian Estimators for Refined Dose Adjustment of a New Tacrolimus Formulation in Kidney and Liver Transplant Patients. Clin Pharmacokinet 56, 1491–1498 (2017). https://doi.org/10.1007/s40262-017-0533-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40262-017-0533-5