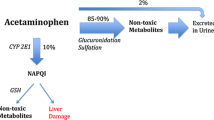
Abstract
Introduction and Aim
Previous studies and systematic reviews have not provided conclusive evidence on the effect of N-acetylcysteine (NAC) in non-acetaminophen-induced acute liver failure (NAI-ALF). We aimed to study the value of intravenous NAC in reducing liver transplantation and mortality in NAI-ALF.
Patients and Methods
In a prospective, multicenter, observational study, acute liver failure patients without clinical or historical evidence of acetaminophen overdose were enrolled. NAC infusion (in empirical dose) was given as 150 mg/kg in 100 ml dextrose 5% over half an hour, then 70 mg/kg in 500 ml dextrose 5% over 4 h, then 70 mg/kg in 500 ml dextrose 5% over 16 h. Thereafter continuous infusion was administered over 24 h of 150 mg/kg in 500 ml dextrose 5% until up to two consecutive normal international normalized ratios (INRs) were obtained. Our endpoints were recovery, transplantation, or death. The primary outcome of the study was to assess reduction in mortality or liver transplantation. The secondary outcome was the evaluation of other clinical outcomes (length of ICU and hospital stays, organ system failure, and hepatic encephalopathy).
Results
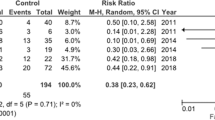
The study included a total of 155 adults; the NAC group (n = 85) were given NAC between January 2011 to December 2013 and the control group (n = 70) were not given NAC and were included from files dating between 2010 and 2011. Both groups (before NAC) were comparable with regard to etiology, age, sex, smoking, presence of co-morbidities, encephalopathy, liver profile, and INR. The success rate (transplant-free survival) in the NAC group was 96.4%. While in the control group, 17 patients (23.3%) recovered and 53 (76.6%) did not recover, of these 37 (53.3%) had liver transplantation and 16 (23.3%) died (p < 0.01). The NAC group had significantly shorter hospital stays (p < 0.001), less encephalopathy (p = 0.02), and less bleeding (p < 0.01) than the control group. The control group reported a higher ICU admission (p = 0.01) rate and abnormal creatinine and electrolytes (p = 0.002, p < 0.01, respectively). Liver profile and INR (after NAC infusion) differed significantly between the two groups with regard to bilirubin (increased in controls, p = 0.02), AST and INR (decreased in NAC group, p < 0.001 for both), but the ALT decrease showed no statistical significance between the two groups.
Conclusions
When administered on admission, intravenous NAC caused a reduction in NAI-ALF mortality and need for transplantation. NAC decreased encephalopathy, hospital stay, ICU admission, and failure of other organs.
Similar content being viewed by others
References
Mumtaz K, Azam Z, Hamid S, Abid S, Memon S, Ali Shah H, Jafri W. Role of N-acetylcysteine in adults with non-acetaminophen induced acute liver failure in a center without the facility of liver transplantation. Hepatol Int. 2009;3:563–70. doi:10.1007/s12072-009-9151-0.
O’Grady JG, Schalm SW, Williams R. Acute liver failure: redefining the syndromes. Lancet. 1993;342(8866):273–5.
O’Grady JG, Williams R. Classification of acute liver failure. Lancet. 1993;342(8873):743.
Smilkstein MJ, Bronstein AC, Linden C, Augenstein WL, Kulig KW, Rumack BH. Acetaminophen overdose: a 48-h intravenous N-acetylcysteine treatment protocol. Ann Emerg Med. 1991;20(10):1058–63.
Smilkstein MJ, Knapp GL, Kulig KW, Rumack BH. Efficacy of oral N-acetylcysteine in the treatment of acetaminophen overdose. Analysis of the national multicenter study (1976–1985). N Engl J Med. 1988;319(24):1557–62.
Burgunder JM, Varriale A, Lauterburg BH. Effect of N-acetylcysteine on plasma cysteine and glutathione following paracetamol administration. Eur J Clin Pharmacol. 1989;36(2):127–31.
Zafarullah M, Li WQ, Sylvester J, Ahmad M. Molecular mechanisms of N-acetylcysteine actions. Cell Mol Life Sci. 2003;60(1):6–20.
Harrison P, Wendon J, Williams R. Evidence of increased guanylate cyclase activation by acetylcysteine in fulminant hepatic failure. Hepatology. 1996;23(5):1067–72.
Harrison PM, Wendon JA, Gimson AE, Alexander GJ, Williams R. Improvement by acetylcysteine of hemodynamics and oxygen transport in fulminant hepatic failure. N Engl J Med. 1991;324(26):1852–7.
Ytrebo LM, Korvald C, Nedredal GI, Elvenes OP, Nielsen Grymyr OJ, Revhaug A. N-Acetylcysteine increases cerebral perfusion pressure in pigs with fulminant hepatic failure. Crit Care Med. 2001;29(10):1989–95.
Peristeris P, Clark BD, Gatti S, Faggioni R, Mantovani A, Mengozzi M, et al. N-acetylcysteine and glutathione as inhibitors of tumor necrosis factor production. Cell Immunol. 1992;140:390–9.
Ben-Ari Z, Vaknin H, Tur-Kaspa R. N-Acetylcysteine in acute hepatic failure (non-paracetamol induced). Hepatogastroenterology. 2000;47(33):786–9.
Katoonizadeh A, Decaestecker J, Wilmer A, Aerts R, Verslype C, Vansteenbergen W, et al. MELD score to predict outcome in adult patients with non-acetaminophen-induced acute liver failure. Liver Int. 2007;27(3):329–34.
Escorsell A, Mas A, de la Mata M. Acute liver failure in Spain: analysis of 267 cases. Liver Transpl. 2007;13(10):1389–95.
Sklar GE, Subramaniam M. Acetylcysteine treatment for non-acetaminophen-induced acute liver failure. Ann Pharmacother. 2004;38(3):498–500 Epub 2004 Jan 12.
Lee WM, Larson AM, Stravitz RT. AASLD Position Paper: The Management of Acute Liver Failure: Update 2011. http://www.aasld.org/practiceguidelines/ Documents/AcuteLiverFailureUpdate2011.pdf. Accessed Jan 2016.
Lee WM, Hynan LS, Rossaro L, Fontana RJ, Stravitz RT, Larson AM, et al. Acute Liver Failure Study Group. Intravenous N-acetylcysteine improves transplant-free survival in early stage non-acetaminophen acute liver failure. Gastroenterology. 2009;137:856–86.
Sales I, Dzierba AL, Smithburger PL, Rowe D, Kane-Gill SL. Use of acetylcysteine for non-acetaminophen-induced acute liver failure. Ann Hepatol. 2013;12(1):6–10.
Bass S, Zook N. Intravenous acetylcysteine for indications other than acetaminophen overdose. Am J Health Syst Pharm. 2013;70(17):1496–501. doi:10.2146/ajhp120645.
Hu J, Zhang Q, Ren X, Sun Z, Quan Q. Efficacy and safety of acetylcysteine in “non-acetaminophen” acute liver failure: A meta-analysis of prospective clinical trials. Clin Res Hepatol Gastroenterol. 2015;39(5):594–9. doi:10.1016/j.clinre.2015.01.003 Epub 2015 Feb 26.
Chughlay MF, Kramer N, Spearman CW, Werfalli M, Cohen K. N-acetylcysteine for non-paracetamol drug-induced liver injury: a systematic review. Br J Clin Pharmacol. 2016;. doi:10.1111/bcp.12880 [Epub ahead of print].
Koch A, Trautwein C. N-acetylcysteine on its way to a broader application in patients with acute liver failure. Hepatology. 2010;51(1):338–40. doi:10.1002/hep.23484.
Trey C, Davidson CS. The management of fulminant hepatic failure. Prog Liver Dis. 1970;3:282–98.
Zwingmann C, Bilodeau M. Metabolic insights into the hepato-protective role of N-acetylcysteine in mouse liver. Hepatology. 2006;43:454–63.
Pitari G, Dupre S, Spirito A, Antonini G, Amicarelli F. Hypotaurine protection on cell damage by singlet oxygen. Adv Exp Med Biol. 2000;483:157–62.
Jones AL. Mechanism of action and value of N-acetylcysteine in the treatment of early and late acetaminophen poisoning: a critical review. J Toxicol Clin Toxicol. 1998;36:277–85.
Kortsalioudaki C, Taylor RM, Cheeseman P, Bansal S, Mieli-Vergani G, Dhawan A. Safety and efficacy of N-acetylcysteine in children with non-acetaminophen-induced acute liver failure. Liver Transpl. 2008;14(1):25–30.
Gunduz H, Karabay O, Tamer A, Özaras R, Mert A, Tabak ÖF. N-acetylcysteine therapy in acute viral hepatitis. World J Gastroenterol. 2003;9(12):2698–700.
Squires RH, Dhawan A, Alonso E, Narkewicz MR, Shneider BL, Rodriguez-Baez N, et al. Intravenous N-acetylcysteine in pediatric patients with non-acetaminophen acute liver failure: a Placebo-Controlled Clinical Trial. Hepatology. 2013;57(4):1542–9. doi:10.1002/hep.26001.
Nguyen-Khac E, Thevenot T, Piquet MA, Benferhat S, Goria O, Chatelain D, Tramier B. Glucocorticoids plus N-acetylcysteine in severe alcoholic hepatitis. N Engl J Med. 2011;365:1781–9.
Singh S, Hynan LS, Lee WM. Improvements in hepatic serological biomarkers are associated with clinical benefit of intravenous N-acetylcysteine in early stage non-acetaminophen acute liver failure. Dig Dis Sci. 2013;58(5):1397–402. doi:10.1007/s10620-012-2512-x.
Acknowledgements
The authors thank Prof. Dr. Wafaa Al-Akel who provided the statistics for this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This research did not receive any specific grant from any funding agency in the public, commercial, or not-for-profit sector.
Conflicts of interest
The authors declare no conflicts of interest.
Ethical approval
All procedures in this study were in accordance with the 1964 Declaration of Helsinki (and its amendments), and the requirements of the ethics committee or institutional review board that approved the study.
Informed consent
Written informed consent obtained from patients, parents, or care givers.
Data sharing statement
No additional data are available.
Rights and permissions
About this article
Cite this article
Darweesh, S.K., Ibrahim, M.F. & El-Tahawy, M.A. Effect of N-Acetylcysteine on Mortality and Liver Transplantation Rate in Non-Acetaminophen-Induced Acute Liver Failure: A Multicenter Study. Clin Drug Investig 37, 473–482 (2017). https://doi.org/10.1007/s40261-017-0505-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40261-017-0505-4