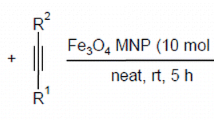Abstract
An atom-economical, efficient and mild protocol is described for the synthesis of 2-amino-3-cyanopyridine and 2-amino-3-cyano-4H-pyran derivatives in the presence of high surface area Fe3O4 as a highly effective heterogeneous catalyst via one-pot multicomponent cyclo-condensation reaction.




Similar content being viewed by others
References
P.L. Barili, G. Biagi, O. Livi, L. Mucciand, V.J. Scartoni, Heterocycl. Chem. 24, 997 (1987)
M.T. Cocco, C. Congiu, V. Lilliu, V. Onnis, Eur. J. Med. Chem. 40, 1365 (2005)
T.R. Reddy, R. Mutter, W. Heal, K. Guo, V.J. Gillet, S. Pratt, B. Chen, J. Med. Chem. 49, 607 (2006)
G. Jones, A.R. Katritzky, C.W. Rees, E.F.V. Scriven, A. McKillop, Compr Heterocycl Chem II 5, 167 (1996)
J.P. Michael, Nat. Prod. Rep. 22, 627 (2005)
X.S. Wang, D.Q. Shi, S.T. Tu, C.S. Yao, Synth. Commun. 33, 119 (2003)
S. Hatakeyama, N. Ochi, H. Numata, S. Takano, J. Chem. Soc., Chem. Commun. 17, 1202 (1988)
C.J. Shishoo, M.B. Devani, V.S. Bhadti, S. Ananthan, G.V. Ullas, Tetrahedron Lett. 24, 4611 (1983)
A.H. Adbel-Fattah, A.M. Hesien, S.A. Metwally, M.H. Elnagdi, Ann. Chem. 21, 585 (1989)
M. Mantri, O. De Graaf, J. Van Veldhoven, T. Mulder-Krieger, R. Link, H. De Vries, M.W. Beukers, J. Brussee, A.P. Ijzerman, J. Med. Chem. 51, 4449 (2008)
J.L. Wang, D. Liu, Z.J. Zhang, S. Shan, X. Han, S.M. Srinivasula, C.M. Croce, E.S. Alnemri, Z. Huang, Proc. Natl. Acad. Sci. U.S.A. 97, 7124 (2000)
L.L. Andreani, E. Lapi, Bull. Chim. Farm. 99, 583 (1960)
R. Gupta, A. Jain, M. Jain, R. Joshi, Bull. Korean Chem. Soc. 31, 3180 (2010)
F. Zhang, Y. Zhao, L. Sun, L. Ding, Y. Gu, P. Gong, Eur. J. Med. Chem. 46, 3149 (2011)
D. Fang, H.B. Zhang, Z.L. Liu, J. Heterocycl. Chem. 47, 63 (2010)
M. Seifi, H. Sheibani, Catal. Lett. 126, 275 (2008)
M.M. Heravi, B.A. Jani, F. Derikvand, F.F. Bamoharram, H.A. Oskooie, Catal. Commun. 10, 272 (2008)
M.M. Heravi, Kh Bakhtiari, A. Fatehi, F. Bamoharram, Catal. Commun. 9, 289 (2008)
M.M. Heravi, S. Sadjadi, J. Iran. Chem. Soc. 6, 1 (2009)
T. Cheng, D. Zhang, H. Li, G. Liu, Green Chem. 16, 3401 (2014)
R. Mr´owczy´nski, A. Nanb, J. Liebscher, RSC Adv. 4, 5927 (2014)
M.M. Heravi, F. Mousavizadeh, N. Ghobadi, M. Tajbakhsh, Tetrahedron Lett. 55, 1226 (2014)
H.A. Oskoole, M.M. Heravi, Kh Bakhtiari, V. Zadsirjan, F. Bamoharram, Synlett 11, 1768 (2007)
J.Y. Lek, L. Xi, B.E. Kardynal, L.H. Wong, Y.M. Lam, ACS Appl. Mater. 3, 287 (2011)
M.M. Heravi, S. Moghimi, Curr. Org. Chem. 17, 504 (2013)
F. Iskandar, Adv. Powder Technol. 20, 283 (2009)
V.K. Sharma, R.A. Yngard, Y. Lin, Adv. Colloid Interface Sci. 145, 83 (2009)
G.J. Hutchings, Catal. Today 100, 55 (2005)
M.M. Heravi, E. Hashemi, S.Y. Shirazibeheshtiha, Kh Kamjou, M. Toolabi, N. Hosseintash, J. Mol. Catal. A: Chem. 392, 173 (2014)
F. Bamoharram, M.M. Heravi, M. Roshani, J. Mol. Catal. A: Chem. 271, 126 (2007)
M.M. Heravi, Gh Rajabzadeh, F. Bamoharram, J. Mol. Catal. A: Chem. 256, 238 (2006)
E. Hashemi, S.Y. Shirazi Beheshtiha, S. Ahmadi, M.M. Heravi, Transit. Met. Chem. 39, 593 (2014)
W.S. Chiu, S. Radiman, M.H. Abdullah, P.S. Khiew, N.M. Huang, R. Abd-Shukor, Mater. Chem. Phys. 106, 231 (2007)
M.M. Heravi, M. Tajbakhsh, A.N. Ahmadi, B. Mohajerani, Monatsh. Chem. 137, 175 (2006)
M.M. Heravi, Kh Bakhtiari, Z. Daroogheha, F. Bamoharram, Catal. Commun. 8, 1991 (2007)
M.M. Heravi, M. Khorasani, F. Derikvand, H.A. Oskoole, F. Bamoharram, Catal. Commun. 8, 1886 (2007)
M.M. Heravi, V. Zadsirjan, Kh Bakhtiari, Catal. Commun. 8, 315 (2007)
M.M. Heravi, Kh Bakhtiari, V. Zadsirjan, Bioorg. Med. Chem. Lett. 17, 4262 (2007)
M.M. Heravi, Kh Bakhtiari, Z. Daroogheha, F. Bamoharram, J. Mol. Catal. A: Chem. 273, 99 (2007)
D. Astruc, F. Lu, J.R. Aranzaes, Angew. Chem. Int. Ed. 44, 7852 (2005)
M.M. Heravi, T. Alishiri, Heterocycles 85, 545 (2012)
G. Evanom, N. Blanchard, M. Toumi, Chem. Rev. 108, 3054 (2008)
M.M. Heravi, Sh Asadi, B.M. Lashkariani, Mol. Divers. 17, 389 (2013)
M.M. Heravi, F.K. Behbahani, M. Darai, H.A. Oskooie, Mol. Divers. 13, 375 (2009)
M.M. Heravi, S. Moghimi, J. Iran. Chem. Soc. 8, 306 (2011)
M. Negwar, Organic-chemical Drugs and Their Synonyms (Akademie, Berlin, 1994)
M.G. Dekamin, Z. Mokhtari, Z. Karimi, Sci. Iran. Trans. 18, 1356 (2011)
M.M. Heravi, Z. Faghihi, J. Iran. Chem. Soc. 11, 209 (2014)
F. Nemati, M.M. Heravi, R. Saeedi Rad, Chin. J. Catal. 33, 1825 (2012)
F. Janati, M.M. Heravi, A.M. Shokraie, Synth. React. Org. Chem. 45, 1 (2015)
T. Alishiri, H.A. Oskooei, M.M. Heravi, Synth. Commun. 43, 3357 (2013)
S. Bakhshayesh, H. Dehghani, J. Iran. Chem. Soc. 11, 769 (2014)
R. Rahimi, H. Kerdari, M. Rabbani, M. Shafiee, Desalination 280, 412 (2011)
R. Rahimi, A. Tadjarodi, M. Rabbani, H. Kerdari, J. Supercond. Nov. Magn. 26, 219 (2013)
J.B. Mamani, A.J. Costa-Filho, D.R. Cornejo, E.D. Vieira, L.F. Gamarra, Mater. Charact. 81, 28 (2013)
M.G. Assy, S.H.A. Youssif, N.H. Ouf, Pol. J. Chem. 6, 896 (1995)
L. Zhi, C.M. Tegley, B. Pio, J.P. Edward, M. Motamedi, T.D. Jones, K.B. Marschke, D.E. Mais, B. Risek, W.T. Schrader, J. Med. Chem. 46, 4104 (2003)
S. Khaksar, M. Yaghoobi, J. Fluor. Chem. 142, 41 (2012)
Y.S. Beheshtia, M. Khorshidi, M.M. Heravi, B. Baghernejad, Eur. J. Chem. 3, 232 (2010)
J. Tang, L. Wang, Y. Yao, L. Zhang, W. Wanga, Tetrahedron Lett. 52, 509 (2011)
G.M. Nazeruddin, Y.I. Shaikh, A.A. Shaikh, RJPBCS 5, 1773 (2014)
Acknowledgments
M. M. H is thankful to Iran National Science Foundation (INSF) for partial financial assistance allocated for project 93043105.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Heravi, M.M., Yahya Shirazi Beheshtiha, S., Dehghani, M. et al. Using magnetic nanoparticles Fe3O4 as a reusable catalyst for the synthesis of pyran and pyridine derivatives via one-pot multicomponent reaction. J IRAN CHEM SOC 12, 2075–2081 (2015). https://doi.org/10.1007/s13738-015-0684-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-015-0684-y