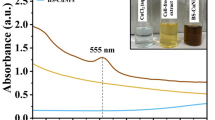
Abstract
Pseudomonas includes ubiquitous, opportunistic, pathogenic bacteria known to produce a variety of bioactive compounds. Objective of present study was to evaluate bioactive secondary metabolites produced by novel Pseudomonas aeruginosa BS25 strain. The bacterium isolated from patient’s wound was identified via 16S rRNA sequencing. To prepare extracts, nonpolar to polar solvents, i.e., hexane [H], ethyl acetate [E], acetone–ethyl acetate [AE] and water [W], were used. Cytotoxicity screening was performed against three cancer cell lines: HeLa, HepG2 and SHSY5Y. The effect on cell proliferation and mitochondrial activity was measured in HepG2 and Leishmania major. Furthermore, induction of apoptosis, necrosis, singlet oxygen release, lipid peroxidation and DNA binding were evaluated. For chemical composition colorimetric methods, TLC, HPLC and GC–MS analysis were performed. Extract [H] was effective against all cancer cell lines (relative viability: HeLa = 42.97 ± 3.46%, HepG2 = 41.54 ± 4.26%, SHSY5Y = 49.18 ± 2.98%). It strongly inhibited cell proliferation and mitochondrial activity (HepG2 IC50: 168.93 µg/ml), inducing apoptosis (apoptotic cells: 150 µg/ml = 24.89 ± 1.7%, 200 µg/ml = 43.83 ± 2.92%). Furthermore, [H] induced oxidative stress (ΦΔ = 0.5 ± 0.17) and generated TBARs (150 µg/ml = 1.81 ± 0.02 and 200 µg/ml = 2.1 ± 0.2) and DNA degradation. On the other hand, IC50 for L. major was 94.2 µg/ml. Chemical composition analysis indicated the presence of flavonoids, whereas TLC and HPLC revealed two major fractions, and GC–MS showed similarity with l-(+)-ascorbic acid 2,6-dihexadecanoate and 7,9-di-tertbutyl-1-oxaspiro (4,5) deca-6,9-diene-8-one. [H] of BS25 strain exhibits strong biological activity including ROS production, DNA damage and induction of apoptosis. Further investigations on normal cells and in vivo effects are required to fully understand its therapeutic potential.









Similar content being viewed by others
References
Baig, N.F.; Dunham, S.J.B.; Morales-Soto, N.; et al.: HHS Public Access. Analyst 140, 6544–6552 (2016)
Drees, S.L.; Fetzner, S.: PqsE of Pseudomonas aeruginosa acts as pathway-specific thioesterase in the biosynthesis of alkylquinolone signaling molecules. Chem. Biol. 22, 611–618 (2015)
Barkal, L.J.; Theberge, A.B.; Guo, C.-J.; et al.: Microbial metabolomics in open microscale platforms. Nat. Commun. 7, 10610 (2016)
Singla, R.K.; Dubey, H.D.; Dubey, A.K.: Therapeutic spectrum of bacterial metabolites. Indo Glob. J. Pharm. 2, 52–64 (2014)
Isnansetyo, A.; Kamei, Y.: Bioactive substances produced by marine isolates of Pseudomonas. J. Ind. Microbiol. Biotechnol. 36, 1239–1248 (2009)
Osman, Y.A.; El-Deep, D.; Younis, S.A.: Azurin: a powerful anticancer from “A” local Pseudomonas aeruginosa isolate. J. Am. Sci. 9, 755–764 (2013)
Leite, G.G.F.; Figueirôa, J.V.; Almeida, T.C.M.; et al.: Production of rhamnolipids and diesel oil degradation by bacteria isolated from soil contaminated by petroleum. Biotechnol. Progr. 32, 262–270 (2016)
Compant, S.; Brader, G.; Muzammil, S.; et al.: Use of beneficial bacteria and their secondary metabolites to control grapevine pathogen diseases. Biocontrol 58, 1–21 (2013)
Buzid, A.; Muimhneacháin, E.Ó.; Reen, F.J.; et al.: Synthesis and electrochemical detection of a thiazolyl-indole natural product isolated from the nosocomial pathogen Pseudomonas aeruginosa. Anal. Bioanal. Chem. 408, 6361–6367 (2016)
Meisel, J.D.; Panda, O.; Mahanti, P.; et al.: Chemosensation of bacterial secondary metabolites modulates neuroendocrine signaling and behavior of C. elegans. Cell. 159, 267–280 (2014)
Spago, F.R.; Mauro, C.I.; Oliveira, A.G.; et al.: Pseudomonas aeruginosa produces secondary metabolites that have biological activity against plant pathogenic Xanthomonas species. Crop Prot. 62, 46–54 (2014)
Hernández-Padilla, L.; Vázquez-Rivera, D.; Sánchez-Briones, L.A.; et al.: The antiproliferative effect of cyclodipeptides from Pseudomonas aeruginosa PAO1 on HeLa cells involves inhibition of phosphorylation of Akt and S6k kinases. Molecules 22, 1024 (2017)
Moayedi, A.; Nowroozi, J.; Akhavan Sepahi, A.: Cytotoxic effect of pyocyanin on human pancreatic cancer cell line (Panc-1). Iran J. Basic Med. Sci. 2018(21), 794–799 (2018)
Fonseca, A.P.; Correia, P.; Sousa, J.C.; et al.: Association patterns of Pseudomonas aeruginosa clinical isolates as revealed by virulence traits, antibiotic resistance, serotype and genotype. FEMS Immunol. Med. Microbiol. 51, 505–516 (2007)
Gill, M.M.; Usman, J.; Kaleem, F.; et al.: Frequency and antibiogram of multi-drug resistant Pseudomonas aeruginosa. J. Coll. Phys. Surg. Pak. 21, 531–534 (2011)
Lister, P.D.; Wolter, D.J.; Hanson, N.D.: Antibacterial-resistant Pseudomonas aeruginosa: clinical impact and complex regulation of chromosomally encoded resistance mechanisms. Clin. Microbiol. Rev. 22, 582–610 (2009)
Vital-Lopez, F.G.; Reifman, J.; Wallqvist, A.: Biofilm formation mechanisms of Pseudomonas aeruginosa predicted via genome-scale kinetic models of bacterial metabolism. PLoS Comput. Biol. 11, 1–24 (2015)
Fujii, A.; Seki, M.; Higashiguchi, M.; et al.: Respiratory medicine case reports pneumonia caused by Pseudomonas aeruginosa. Respir. Med. Case Rep. 12, 30–33 (2014)
Seki, M.; Machida, N.; Yamagishi, Y.; et al.: Nosocomial outbreak of multidrug-resistant Pseudomonas aeruginosa caused by damaged transesophageal echocardiogram probe used in cardiovascular surgical operations. J. Infect. Chemother. 19, 677–681 (2013)
Quick, J.; Cumley, N.; Wearn, C.M.; et al.: Seeking the source of Pseudomonas aeruginosa infections in a recently opened hospital: an observational study using whole-genome sequencing. BMJ Open 4, e006278 (2014)
Shruti, G.; Tanuja, K.; Shweta, Y.: Exposure to ZnO-NPs enhanced gut-associated microbial activity in Eisenia fetida. J. Toxicol. Environ. Health Sci. 7, 9–17 (2015)
Mithun, S.L.V.; Rao, C.S.V.R.: Isolation and molecular characterization of anti-cancerous compound producing marine bacteria by using 16S rRNA sequencing and GC–MS techniques. IJMER 2, 4510–4515 (2012)
Chen, W.; Kuo, T.: A simple and rapid method for the preparation of gram-negative bacterial genomic DNA. Nucl. Acids Res. 21, 2260 (1993)
Null, A.P.; George, L.T.; Muddiman, D.C.: Evaluation of sample preparation techniques for mass measurements of PCR products using ESI-FT-ICR mass spectrometry. J. Am. Soc. Mass. Spectr. 13, 338–344 (2002)
Thomas, A.T.; Rao, J.V.; Subrahmanyam, V.M.; et al.: In vitro anticancer activity of microbial isolates from diverse habitats. Braz. J. Pharm. Sci. 47, 279–287 (2011)
Jafri, L.; Saleem, S.; Mirza, B.: In vitro assessment of antioxidant potential and determination of polyphenolic compounds of Hedera nepalensis K. Koch. Arab. J. Chem. 10, S3699–S3706 (2014)
Velioglu, Y.S.; Mazza, G.; Gao, L.; et al.: Antioxidant activity and total phenolics in selected fruits, vegetables, and grain products. J. Agric. Food Chem. 46, 4113–4117 (1998)
Wagner, H.; Bladt, S.; Zgainski, E.: Plant Drug Analysis, pp. 225–230. Springer, Berlin (1984)
Anantha, P.S.; Deventhiran, M.; Saravanan, P.; et al.: A comparative GC–MS analysis of bacterial secondary metabolites of Pseudomonas species. J. Pharm. Innov. 5, 84–89 (2016)
Arooj, S.; Nazir, S.; Nadhman, A.; et al.: Novel ZnO: Ag nanocomposites induce significant oxidative stress in human fibroblast malignant melanoma (Ht144) cells. Beilstein J. Nanotechnol. 6, 570–582 (2015)
Mahmoudvand, H.; Sharififar, F.; Rahmat, M.S.; et al.: Evaluation of antileishmanial activity and cytotoxicity of the extracts of Berberis vulgaris and Nigella sativa against Leishmania tropica. J. Vector Borne Dis. 51, 294–299 (2014)
Mascotti, K.; Mccullough, J.; Burger, S.R.: HPC viability measurement: trypan blue versus acridine orange and propidium iodide. Transfusion 40, 693–696 (2000)
Tada, D.B.; Vono, L.L.R.; Duarte, E.L.; et al.: Methylene blue-containing silica-coated magnetic particles: a potential magnetic carrier for photodynamic therapy. Langmuir 23, 8194–8199 (2007)
Bonacin, J.A.; Engelmann, F.M.; Severino, D.; et al.: Singlet oxygen quantum yields (Φd) in water using beetroot extract and an array of LEDs. J. Braz. Chem. Soc. 20, 31–36 (2009)
Ohkawa, H.; Ohishi, N.; Yagi, K.: Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 95, 351–358 (1979)
Arslantas, A.; Devrim, A.K.; Necefoglu, H.: The interaction of sheep genomic DNA with a cobalt (ii) complex containing p-nitrobenzoate and n,n′-diethylnicotinamide ligands. Int. J. Mol. Sci. 8, 1225–1233 (2007)
Khan, S.Z.; Amir, M.K.; Abbasi, R.; et al.: New 3D and 2D supramolecular heteroleptic palladium(II) dithiocarbamates as potent anticancer agents. J. Coord. Chem. 69(20), 2999–3009 (2016)
d’Angelo, F.; Santillo, A.; Sevi, A.; Albenzio, M.: A simple salting-out method for DNA extraction from milk somatic cells: investigation into the goat CSN1S1 gene. JDS 90, 3550–3552 (2007)
Zohra, M.; Fawzia, A.: Hemolytic activity of different herbal extracts used in Algeria. IJPSR 5, 495–500 (2014)
Abbasi, R.; Efferth, T.; Kuhmann, C.; et al.: The endoperoxide ascaridol shows strong differential cytotoxicity in nucleotide excision repair-deficient cells. Toxicol. Appl. Pharmacol. 259, 302–310 (2012)
Chauhan, M.; Banerjee, K.; Arjmand, F.: DNA binding studies of novel copper(II) complexes containing l-Tryptophan as chiral auxiliary: in vitro antitumor activity of Cu–Sn2 complex in human neuroblastoma cells. Inorg. Chem. 46, 3072–3082 (2007)
McGaw, L.J.; Bagla, V.P.; Steenkamp, P.A.; et al.: Antifungal and antibacterial activity and chemical composition of polar and non-polar extracts of Athrixia phylicoides determined using bioautography and HPLC. BMC Complement. Altern. Med. 13, 356 (2013)
Brandhagen, B.N.; Tieszen, C.R.; Ulmer, T.M.; et al.: Cytostasis and morphological changes induced by mifepristone in human metastatic cancer cells involve cytoskeletal filamentous actin reorganization and impairment of cell adhesion dynamics. BMC Cancer 13, 35 (2013)
Du, K.; Ramachandran, A.; Jaeschke, H.: Oxidative stress during acetaminophen hepatotoxicity: sources, pathophysiological role and therapeutic potential. Redox Biol. 1, 148–156 (2016)
Kryston, T.B.; Georgiev, A.B.; Pissis, P.; et al.: Mutation research/fundamental and molecular mechanisms of mutagenesis role of oxidative stress and dna damage in human carcinogenesis. Mutat. Res. Fund. Mol. Mech. 711, 193–201 (2011)
Cherrak, S.A.; Mokhtari-Soulimane, N.; Berroukeche, F.; et al.: In vitro antioxidant versus metal ion chelating properties of flavonoids: a structure-activity investigation. PLoS ONE 11, e0165575 (2016)
Semwal, D.K.; Semwal, R.B.; Combrinck, S.; et al.: Myricetin: a dietary molecule with diverse biological activities. Nutrients 8, 1–31 (2016)
Ta, C.A.K.; Arnason, J.T.: Mini review of phytochemicals and plant taxa with activity as microbial biofilm and quorum sensing inhibitors. Molecules (Basel, Switzerland) 21, E29 (2015)
Gibellini, L.; Pinti, M.; Nasi, M.; et al.: Interfering with ROS metabolism in cancer cells: the potential role of quercetin. Cancers 2, 1288–1311 (2010)
Zhang, L.; Li, J.; Zong, L.; et al.: Reactive oxygen species and targeted therapy for pancreatic cancer. Oxid. Med. Cell. Longev. 2016, 1616781 (2016)
Acknowledgements
The study was supported by the Higher Education Commission (HEC), Pakistan (Project ID: PMIPFP/HRD/HEC/2010/1815), and the Institute of Biomedical and Genetic Engineering, Islamabad, Pakistan. The authors wish to thank Prof. Dr. Z. K. Shinwari (Molecular Systematics and Applied Ethnobotany lab, Department of Biotechnology, Quaid-i-Azam University) for his advice and support, and N. Khan (Institute of Biomedical and Genetic Engineering, Islamabad), H. Shakeel (International Islamic University, Islamabad) and A. Anwar (PMAS-University of Arid Agriculture, Rawalpindi) for their excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mushtaq, S., Uzair, B., Hameed, A. et al. In Vitro Cytotoxicity of Secondary Metabolites Extracted from Pseudomonas aeruginosa BS25 Strain. Arab J Sci Eng 45, 81–94 (2020). https://doi.org/10.1007/s13369-019-04092-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13369-019-04092-2