Abstract
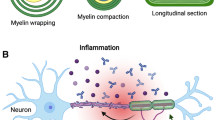
Microglia, the major resident immune cells in the central nervous system (CNS) are considered as the key cellular mediators of neuroinflammatory processes. In the past few years, microglial research has become a main focus in cellular neuroimmunology and neuroinflammation. Chronic/remitting neurological disease such as multiple sclerosis (MS) has long been considered an inflammatory autoimmune disease with the infiltration of peripheral myelin-specific T cells into the CNS. With the rapid advancement in the field of microglia and astrocytic neurobiology, the term neuroinflammation progressively started to denote chronic CNS cell-specific inflammation in MS. The direct glial responses in MS are different from conventional peripheral immune responses. This review attempts to summarize current findings of neuroinflammatory responses within the CNS by direct infection of neural cells by mouse hepatitis virus (MHV) and the mechanisms by which glial cell responses ultimately contribute to the neuropathology on demyelination. Microglia can be persistently infected by MHV. Microglial activation and phagocytosis are recognized to be critically important in the pathogenesis of demyelination. Emerging evidence for the pathogenic role of microglia and the activation of inflammatory pathways in these cells in MHV infection supports the concept that microglia induced neuroinflammation is an amplifier of virus-induced neuropathology.



Similar content being viewed by others
References
Aarum J, Sandberg K, Haeberlein SL, Persson MA (2003) Migration and differentiation of neural precursor cells can be directed by microglia. Proc Natl Acad Sci U S A 100(26):15983–15988
An SF, Giometto B, Groves M, Miller RF, Beckett AA, Gray F, Tavolato B, Scaravilli F (1997) Axonal damage revealed by accumulation of beta-APP in HIV-positive individuals without AIDS. J Neuropathol Exp Neurol 56(11):1262–1268
Baker CA, Martin D, Manuelidis L (2002) Microglia from Creutzfeldt-Jakob disease-infected brains are infectious and show specific mRNA activation profiles. J Virol 76(21):10905–10913
Betmouni S, Perry VH, Gordon JL (1996) Evidence for an early inflammatory response in the central nervous system of mice with scrapie. Neuroscience 74(1):1–5
Chan WY, Kohsaka S, Rezaie P (2007) The origin and cell lineage of microglia: new concepts. Brain Res Rev 53(2):344–354
Chen DS, Asanaka M, Chen FS, Shively JE, Lai MM (1997) Human carcinoembryonic antigen and biliary glycoprotein can serve as mouse hepatitis virus receptors. J Virol 71(2):1688–1691
Dandekar AA, Wu GF, Pewe L, Perlman S (2001) Axonal damage is T cell-mediated and occurs concomitantly with demyelination in mice infected with a neurotropic coronavirus. J Virol 75(13):6115–6120
Das Sarma J (2010) A mechanism of virus-induced demyelination. Interdisc Perspect Infect Dis 2010:109239
Das Sarma J, Fu L, Hingley ST, Lavi E (2001) Mouse hepatitis virus type-2 infection in mice: an experimental model system of acute meningitis and hepatitis. Exp Mol Pathol 71(1):1–12
Das Sarma J, Fu L, Tsai JC, Weiss SR, Lavi E (2000) Demyelination determinants map to the spike glycoprotein gene of coronavirus mouse hepatitis virus. J Virol 74(19):9206–9213
Das Sarma J, Iacono K, Gard L, Marek R, Kenyon LC, Koval M, Weiss SR (2008) Demyelinating and nondemyelinating strains of mouse hepatitis virus differ in their neural cell tropism. J Virol 82(11):5519–5526
Das Sarma J, Kenyon LC, Hingley ST, Shindler KS (2009) Mechanisms of primary axonal damage in a viral model of multiple sclerosis. J Neurosci 29(33):10272–10280
Das Sarma J, Scheen E, Seo SH, Koval M, Weiss SR (2002) Enhanced green fluorescent protein expression may be used to monitor murine coronavirus spread in vitro and in the mouse central nervous system. J Neurovirol 8(5):381–391
Davis R, Robertson D (1997) Textbook of neuropathology. Williams and Wilkins, Baltimore
Dveksler GS, Dieffenbach CW, Cardellichio CB, McCuaig K, Pensiero MN, Jiang GS, Beauchemin N, Holmes KV (1993) Several members of the mouse carcinoembryonic antigen-related glycoprotein family are functional receptors for the coronavirus mouse hepatitis virus-A59. J Virol 67(1):1–8
Eikelenboom P, Bate C, Van Gool WA, Hoozemans JJ, Rozemuller JM, Veerhuis R, Williams A (2002) Neuroinflammation in Alzheimer's disease and prion disease. GLIA 40(2):232–239
Frohman EM, Racke MK, Raine CS (2006) Multiple sclerosis—the plaque and its pathogenesis. N Engl J Med 354(9):942–955
Fujinami RS, Oldstone MB (1985) Amino acid homology between the encephalitogenic site of myelin basic protein and virus: mechanism for autoimmunity. Science 230(4729):1043–1045
Gallagher TM, Buchmeier MJ, Perlman S (1992) Cell receptor-independent infection by a neurotropic murine coronavirus. Virology 191(1):517–522
Garden GA (2002) Microglia in human immunodeficiency virus-associated neurodegeneration. GLIA 40(2):240–251
Gilden DH (2005) Infectious causes of multiple sclerosis. Lancet Neurol 4(3):195–202
Gordon S (2007) Macrophage heterogeneity and tissue lipids. J Clin Invest 117(1):89–93
Goverman J, Brabb T (1996) Rodent models of experimental allergic encephalomyelitis applied to the study of multiple sclerosis. Lab Anim Sci 46(5):482–492
Hosking MP, Lane TE (2009) The biology of persistent infection: inflammation and demyelination following murine coronavirus infection of the central nervous system. Curr Immunol Rev 5(4):267–276
Jung S, Schwartz M (2012) Non-identical twins—microglia and monocyte-derived macrophages in acute injury and autoimmune inflammation. Front Immunol 3:89
Kaur C, Hao AJ, Wu CH, Ling EA (2001) Origin of microglia. Microsc Res Tech 54(1):2–9
Kigerl KA, Gensel JC, Ankeny DP, Alexander JK, Donnelly DJ, Popovich PG (2009) Identification of two distinct macrophage subsets with divergent effects causing either neurotoxicity or regeneration in the injured mouse spinal cord. J Neurosci 29(43):13435–13444
Kishore A, Kanaujia A, Nag S, Rostami, AR, Kenyuon, LK, Shindler, KS, Das Sarma J (2013) Different mechanisms of inflammation induced in virus and autoimmune-mediated models of multiple sclerosis in C57BL6 mice. BioMed Res Int. doi:10.1155/2013/589048
Knobler RL, Dubois-Dalcq M, Haspel MV, Claysmith AP, Lampert PW, Oldstone MB (1981) Selective localization of wild type and mutant mouse hepatitis virus (JHM strain) antigens in CNS tissue by fluorescence, light, and electron microscopy. J Neuroimmunol 1(1):81–92
Kuerten S, Gruppe TL, Laurentius LM, Kirch C, Tary-Lehmann M, Lehmann PV, Addicks K (2011) Differential patterns of spinal cord pathology induced by MP4, MOG peptide 35-55, and PLP peptide 178-191 in C57BL/6 mice. APMIS 119(6):336–346
Ladeby R, Wirenfeldt M, Dalmau I, Gregersen R, Garcia-Ovejero D, Babcock A, Owens T, Finsen B (2005) Proliferating resident microglia express the stem cell antigen CD34 in response to acute neural injury. GLIA 50(2):121–131
Lang HL, Jacobsen H, Ikemizu S, Andersson C, Harlos K, Madsen L, Hjorth P, Sondergaard L, Svejgaard A, Wucherpfennig K, Stuart DI, Bell JI, Jones EY, Fugger L (2002) A functional and structural basis for TCR cross-reactivity in multiple sclerosis. Nat Immunol 3(10):940–943
Lassmann H, Bruck W, Lucchinetti C (2001) Heterogeneity of multiple sclerosis pathogenesis: implications for diagnosis and therapy. Trends Mol Med 7(3):115–121
Lavi E, Fishman PS, Highkin MK, Weiss SR (1988) Limbic encephalitis after inhalation of a murine coronavirus. Lab Invest 58(1):31–36
Lavi E, Gilden DH, Highkin MK, Weiss SR (1984a) Persistence of mouse hepatitis virus A59 RNA in a slow virus demyelinating infection in mice as detected by in situ hybridization. J Virol 51(2):563–566
Lavi E, Gilden DH, Wroblewska Z, Rorke LB, Weiss SR (1984b) Experimental demyelination produced by the A59 strain of mouse hepatitis virus. Neurology 34(5):597–603
Lewicki H, Tishon A, Homann D, Mazarguil H, Laval F, Asensio VC, Campbell IL, DeArmond S, Coon B, Teng C, Gairin JE, Oldstone MB (2003) T cells infiltrate the brain in murine and human transmissible spongiform encephalopathies. J Virol 77(6):3799–3808
London A, Cohen M, Schwartz M (2013) Microglia and monocyte-derived macrophages: functionally distinct populations that act in concert in CNS plasticity and repair. Front Cell Neurosci 7:34
Lucchinetti C, Bruck W, Parisi J, Scheithauer B, Rodriguez M, Lassmann H (2000) Heterogeneity of multiple sclerosis lesions: implications for the pathogenesis of demyelination. Ann Neurol 47(6):707–717
Lucchinetti CF, Bruck W, Rodriguez M, Lassmann H (1996) Distinct patterns of multiple sclerosis pathology indicates heterogeneity on pathogenesis. Brain Pathol 6(3):259–274
Mantovani A, Sica A, Sozzani S, Allavena P, Vecchi A, Locati M (2004) The chemokine system in diverse forms of macrophage activation and polarization. Trends Immunol 25(12):677–686
Matthews AE, Lavi E, Weiss SR, Paterson Y (2002) Neither B cells nor T cells are required for CNS demyelination in mice persistently infected with MHV-A59. J Neurovirol 8(3):257–264
Medzhitov R, Janeway C Jr (2000) Innate immunity. N Engl J Med 343(5):338–344
Miller SD, Vanderlugt CL, Begolka WS, Pao W, Yauch RL, Neville KL, Katz-Levy Y, Carrizosa A, Kim BS (1997) Persistent infection with Theiler's virus leads to CNS autoimmunity via epitope spreading. Nat Med 3(10):1133–1136
Mix E, Meyer-Rienecker H, Zettl UK (2008) Animal models of multiple sclerosis for the development and validation of novel therapies—potential and limitations. J Neurol 255(Suppl 6):7–14
Muller FJ, Snyder EY, Loring JF (2006) Gene therapy: can neural stem cells deliver? Nat Rev Neurosci 7(1):75–84
Murray PJ, Wynn TA (2011) Protective and pathogenic functions of macrophage subsets. Nat Rev Immunol 11(11):723–737
Nakamichi K, Saiki M, Sawada M, Takayama-Ito M, Yamamuro Y, Morimoto K, Kurane I (2005) Rabies virus-induced activation of mitogen-activated protein kinase and NF-kappaB signaling pathways regulates expression of CXC and CC chemokine ligands in microglia. J Virol 79(18):11801–11812
Olson JK, Girvin AM, Miller SD (2001) Direct activation of innate and antigen-presenting functions of microglia following infection with Theiler's virus. J Virol 75(20):9780–9789
Parihar A, Eubank TD, Doseff AI (2010) Monocytes and macrophages regulate immunity through dynamic networks of survival and cell death. J Innate Immunol 2(3):204–215
Perry VH, Cunningham C, Boche D (2002) Atypical inflammation in the central nervous system in prion disease. Curr Opin Neurol 15(3):349–354
Roussarie JP, Ruffie C, Edgar JM, Griffiths I, Brahic M (2007) Axon myelin transfer of a non-enveloped virus. PLoS One 2(12):e1331
Saijo K, Winner B, Carson CT, Collier JG, Boyer L, Rosenfeld MG, Gage FH, Glass CK (2009) A Nurr1/CoREST pathway in microglia and astrocytes protects dopaminergic neurons from inflammation-induced death. Cell 137(1):47–59
Schmitz G, Leuthauser-Jaschinski K, Orso E (2009) Are circulating monocytes as microglia orthologues appropriate biomarker targets for neuronal diseases? Cent Nerv Syst Agents Med Chem 9(4):307–330
Shalhoub, J., M. A. Falck-Hansen, A. H. Davies and C. Monaco (2011). "Innate immunity and monocyte-macrophage activation in atherosclerosis." J Inflamm (Lond) 8: 9.
Shindler KS, Chatterjee D, Biswas K, Goyal A, Dutt M, Nassrallah M, Khan RS, Das Sarma J (2011) Macrophage-mediated optic neuritis induced by retrograde axonal transport of spike gene recombinant mouse hepatitis virus. J Neuropathol Exp Neurol 70(6):470–480
Shindler KS, Kenyon LC, Dutt M, Hingley ST, Das Sarma J (2008) Experimental optic neuritis induced by a demyelinating strain of mouse hepatitis virus. J Virol 82(17):8882–8886
Simard AR, Soulet D, Gowing G, Julien JP, Rivest S (2006) Bone marrow-derived microglia play a critical role in restricting senile plaque formation in Alzheimer's disease. Neuron 49(4):489–502
Sospedra M, Martin R (2005) Immunology of multiple sclerosis. Annu Rev Immunol 23:683–747
Sroga JM, Jones TB, Kigerl KA, McGaughy VM, Popovich PG (2003) Rats and mice exhibit distinct inflammatory reactions after spinal cord injury. J Comp Neurol 462(2):223–240
Streit WJ (2002) Microglia as neuroprotective immunocompetent cells of the CNS. GLIA 40(2):133–139
Streit WJ (2004) Microglia and Alzheimer's disease pathogenesis. J Neurosci Res 77(1):1–8
Streit WJ, Kincaid-Colton CA (1995) The brain's immune system. Sci Am 273(5):54–55, 58-61
Streit WJ, Semple-Rowland SL, Hurley SD, Miller RC, Popovich PG, Stokes BT (1998) Cytokine mRNA profiles in contused spinal cord and axotomized facial nucleus suggest a beneficial role for inflammation and gliosis. Exp Neurol 152(1):74–87
Tan K, Zelus BD, Meijers R, Liu JH, Bergelson JM, Duke N, Zhang R, Joachimiak A, Holmes KV, Wang JH (2002) Crystal structure of murine sCEACAM1a[1,4]: a coronavirus receptor in the CEA family. EMBO J 21(9):2076–2086
Trapp BD, Nave KA (2008) Multiple sclerosis: an immune or neurodegenerative disorder? Annu Rev Neurosci 31:247–269
Tsunoda I, Fujinami RS (2002) Inside-out versus outside-in models for virus-induced demyelination: axonal damage triggering demyelination. Springer Semin Immunopathol 24(2):105–125
Upender MB, Naegele JR (1999) Activation of microglia during developmentally regulated cell death in the cerebral cortex. Dev Neurosci 21(6):491–505
Vanderlugt CL, Miller SD (2002) Epitope spreading in immune-mediated diseases: implications for immunotherapy. Nat Rev Immunol 2(2):85–95
Walton MR, Gibbons H, MacGibbon GA, Sirimanne E, Saura J, Gluckman PD, Dragunow M (2000) PU.1 expression in microglia. J Neuroimmunol 104(2):109–115
Weiner LP, Johnson RT, Herndon RM (1973) Viral infections and demyelinating diseases. N Engl J Med 288(21):1103–1110
Wu GF, Dandekar AA, Pewe L, Perlman S (2000) CD4 and CD8 T cells have redundant but not identical roles in virus-induced demyelination. J Immunol 165(4):2278–2286
Acknowledgments
Virus-induced experimental animal model for studying to understand the neuroinflammatory mechanism of demyelination research is supported by the Department of Biotechnology (DBT), Council for Scientific and Industrial Research (CSIR), and IISER-K, India, National Multiple Sclerosis Society, USA, M. E. Groff Surgical Medical Research and Education Charitable Trust, and Lindback Foundation Career Enhancement Award, USA. The success of this work has largely depended on the contribution and devotion of my PhD students, project students, and many collaborators whose names are listed in the references.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 30 kb)
Rights and permissions
About this article
Cite this article
Das Sarma, J. Microglia-mediated neuroinflammation is an amplifier of virus-induced neuropathology. J. Neurovirol. 20, 122–136 (2014). https://doi.org/10.1007/s13365-013-0188-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13365-013-0188-4