Abstract
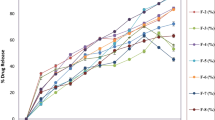
The purpose of this investigation was to prepare and characterize acyclovir loaded floating microspheres by emulsification solvent evaporation method. Piperine was added to investigate its effect on acyclovir bioavailability. The microspheres were characterized for size, shape, entrapment efficiency, in vitro drug release, and in vivo pharmacokinetic parameters. The morphological characterization of microspheres was done using a scanning electron microscope. The microspheres were spherical and had particle size in the range of 400 to 525 μm. The percent drug entrapment efficiency varied between 56.12 ± 1.32 % to 87.32 ± 5.28 %. The drug release was decreased at higher polymer concentrations. Nearly two times higher AUC0–24 value of acyclovir-loaded piperine containing microspheres (15614.13 ± 6953.13 ng h ml−1) was observed as compared to the drug solution (7552.33 ± 3219.09 ng h ml−1). Under the accelerated storage conditions, the best selected formulation was found to be stable for 90 days. The preliminary results of this study suggest that the developed microspheres containing acyclovir could enhance drug entrapment efficiency, reduce initial burst release, and prolong the drug release with enhanced bioavailability.






Similar content being viewed by others
References
Awasthi R, Kulkarni GT. Decades of research in drug targeting to the upper gastrointestinal tract using gastroretention technology: where do we stand? Drug Deliv. 2016;23:378–94.
O’Brien JJ, Campoli-Richards DM. Acyclovir: an updated review of its antiviral activity, pharmacokinetic properties and therapeutic efficacy. Drugs. 1989;37:233–309.
Park GB, Shao Z, Mitra AK. Acyclovir permeation enhancement across intestinal and nasal mucosae by bile salt-acylcarnitine mixed micelles. Pharm Res. 1992;9:1262–7.
Atal CK, A breakthrough in drug bioavailability-a clue from age old wisdom of Ayurveda, IDMA Bulletin. 1979;483–84.
Annamalai AR, Manavalan R. Effects of Trikatu and its individual components and piperine on gastrointerstinal tracts: Trikatua bioavailable enhancer. Ind Drugs. 1989;27:595–604.
Atal CK, Dubey RK, Singh J. Biochemical basis of enhanced drug bioavailability by piperine: evidence that piperine is a potent inhibitor of drug metabolism. J Pharmacol Exp Ther. 1985;232:258–62.
O'Donnell PB, McGinity JW. Preparation of microspheres by the solvent evaporation technique. Adv Drug Deliv Rev. 1997;28:25–42.
Awasthi R, Kulkarni GT. Development and characterization of amoxicillin loaded floating microballoons for the treatment of Helicobacter pylori induced gastric ulcer. Asian J Pharm Sci. 2013;8:174–80.
Dhaliwal S, Jain S, Singh HP, Tiwary AK. Mucoadhesive microspheres for gastroretentive delivery of acyclovir: in vitro and in vivo evaluation. AAPS J. 2008;10:322–30.
Awasthi R, Kulkarni GT, Pawar VK, Garg G. Optimization studies on gastroretentive floating system using response surface methodology. AAPS PharmSciTech. 2012a;13:85–93.
Awasthi R, Kulkarni GT. Development of novel gastroretentive floating particulate drug delivery system of gliclazide. Curr Drug Deliv. 2012b;9:437–51.
Awasthi R, Kulkarni GT. Development of novel gastroretentive drug delivery system of gliclazide: Hollow beads. Drug Dev Ind Pharm. 2014;40:398–408.
Khajuria A, Thusu N, Zutshi U. Piperine modulates permeability characteristics of intestine by inducing alterations in membrane dynamics: influence on brush border membrane fluidity, ultrastructure and enzyme kinetics. Phytomed. 2002;9:224–31.
Singh A, Pawar VK, Jakmola V, Pariva MH, Awasthi R, Sharma G. In vivo assessment of enhanced bioavailability of metronidazole with piperine in rabbit. Research Journal of Pharmaceutical, Biological and Chemical Sciences. 2010;1:273–8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Khatri, S., Awasthi, R. Piperine containing floating microspheres: an approach for drug targeting to the upper gastrointestinal tract. Drug Deliv. and Transl. Res. 6, 299–307 (2016). https://doi.org/10.1007/s13346-016-0285-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-016-0285-z