Abstract
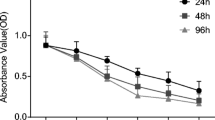
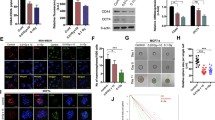
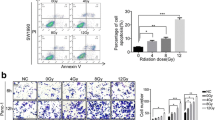
The present study aims to evaluate whether repression of the Numb/Notch signaling pathway affects the radiosensitivity of human pancreatic cancer cell lines. Different doses of X-rays (0, 2, 3, 4, and 5 Gy) were applied to the PANC-1, SW1990, and MIA PaCa-2 human pancreatic cancer cell lines, and the Numb/Notch pathway inhibitor DAPT was added at different doses (0, 1, 3, and 5 μmol/l). MTT assay, colony formation assay, flow cytometry, scratch assay, and Transwell experiments were performed, and qRT-PCR and Western blot were conducted for the detection of Numb expression. Tumorigenicity assay in nude mice was carried out to verify the influence of blocker of the Numb/Notch signaling pathway on the radiosensitivity of xenograft tumors. The MTT assay, colony formation assay and flow cytometry experiments revealed that proliferation decreased as radiation dose increased. The viability of PANC-1 cells at 5 Gy, SW 1990 cells at 4 Gy and 5 Gy, and MIA PaCa-2 cells at 2–5 Gy was significantly lower than that of non-irradiated cells (all P < 0.05). The migration and invasion assays indicated that the PANC-1 cell line was least radiosensitive, while the MIA PaCa-2 cell line was the most radiosensitive. Numb expression significantly increased with increasing radiation dose, whereas the expression of Hes1, Notch1, and Hes5 significantly decreased compared to non-irradiated cells (P < 0.05). Compared to untreated control cells, DAPT dose dependently increased Numb expression and inhibited Notch1, Hes1, and Hes5 expressions at 2 Gy (P < 0.05). Subcutaneous tumorigenicity assay in nude mice demonstrated that DAPT increased the radiosensitivity of PANC-1, SW 1990, and MIA PaCa-2 cells. These findings suggest that Numb/Notch signaling in pancreatic cancer cells is associated with X-ray radiation and that inhibition of the Numb/Notch signaling pathway can enhance radiosensitivity, suggesting that inhibition of the Numb/Notch signaling pathway may serve as a potential target for clinical improvement of the radiosensitivity of pancreatic cancer.










Similar content being viewed by others
References
Stathis A, Moore MJ. Advanced pancreatic carcinoma: current treatment and future challenges. Nat Rev Clin Oncol. 2010;7(3):163–72.
Oberstein PE, Olive KP. Pancreatic cancer: why is it so hard to treat? Therap Adv Gastroenterol. 2013;6(4):321–37.
Vincent A, Herman J, Schulick R, et al. Pancreatic cancer. Lancet. 2011;378(9791):607–20.
Loehrer Sr PJ, Feng Y, Cardenes H, et al. Gemcitabine alone versus gemcitabine plus radiotherapy in patients with locally advanced pancreatic cancer: an Eastern Cooperative Oncology Group trial. J Clin Oncol. 2011;29(31):4105–12.
Torres-Roca JF, Stevens CW. Predicting response to clinical radiotherapy: past, present, and future directions. Cancer Control. 2008;15(2):151–6.
Guan HT, Xue XH, Dai ZJ, et al. Down-regulation of survivin expression by small interfering RNA induces pancreatic cancer cell apoptosis and enhances its radiosensitivity. World J Gastroenterol. 2006;12(18):2901–7.
Rebeiz M, Miller SW, Posakony JW. Notch regulates numb: integration of conditional and autonomous cell fate specification. Development. 2011;138(2):215–25.
Borggrefe T, Oswald F. The Notch signaling pathway: transcriptional regulation at Notch target genes. Cell Mol Life Sci. 2009;66(10):1631–46.
Ma J et al. Notch signaling pathway in pancreatic cancer progression. Pancreat Disord Ther. 2013;3(114).
Bao B, Wang Z, Ali S, et al. Notch-1 induces epithelial-mesenchymal transition consistent with cancer stem cell phenotype in pancreatic cancer cells. Cancer Lett. 2011;307(1):26–36.
Knoblich JA. Mechanisms of asymmetric stem cell division. Cell. 2008;132(4):583–97.
Ding X, Zhu F, Li T, et al. Numb protects renal proximal tubular cells from puromycin aminonucleoside-induced apoptosis through inhibiting Notch signaling pathway. Int J Biol Sci. 2011;7(3):269–78.
Guo Z, Jin X, Jia H. Inhibition of ADAM-17 more effectively down-regulates the Notch pathway than that of gamma-secretase in renal carcinoma. J Exp Clin Cancer Res. 2013;32:26.
Su F, Zhu S, Ruan J, et al. Combination therapy of RY10-4 with the gamma-secretase inhibitor DAPT shows promise in treating HER2-amplified breast cancer. Oncotarget. 2016;7(4):4142–54.
Huang Y, Yang X, Wu Y, et al. Gamma-secretase inhibitor induces adipogenesis of adipose-derived stem cells by regulation of Notch and PPAR-gamma. Cell Prolif. 2010;43(2):147–56.
Hallahan AR, Pritchard JI, Hansen S, et al. The SmoA1 mouse model reveals that notch signaling is critical for the growth and survival of sonic hedgehog-induced medulloblastomas. Cancer Res. 2004;64(21):7794–800.
Baskar R, Lee KA, Yeo R, et al. Cancer and radiation therapy: current advances and future directions. Int J Med Sci. 2012;9(3):193–9.
Connell PP, Hellman S. Advances in radiotherapy and implications for the next century: a historical perspective. Cancer Res. 2009;69(2):383–92.
Wang ZM, Lu J, Zhang LY, et al. Biological effects of low-dose-rate irradiation of pancreatic carcinoma cells in vitro using 125I seeds. World J Gastroenterol. 2015;21(8):2336–42.
Vignard J, Mirey G, Salles B. Ionizing-radiation induced DNA double-strand breaks: a direct and indirect lighting up. Radiother Oncol. 2013;108(3):362–9.
Kozono S, Ohuchida K, Ohtsuka T, et al. S100A4 mRNA expression level is a predictor of radioresistance of pancreatic cancer cells. Oncol Rep. 2013;30(4):1601–8.
Artavanis-Tsakonas S, Rand MD, Lake RJ. Notch signaling: cell fate control and signal integration in development. Science. 1999;284(5415):770–6.
Wang Z, Li Y, Banerjee S, et al. Emerging role of Notch in stem cells and cancer. Cancer Lett. 2009;279(1):8–12.
Reedijk M. Notch signaling and breast cancer. Adv Exp Med Biol. 2012;727:241–57.
Ma YC, Shi C, Zhang YN, et al. The tyrosine kinase c-Src directly mediates growth factor-induced Notch-1 and Furin interaction and Notch-1 activation in pancreatic cancer cells. PLoS One. 2012;7(3):e33414.
Crawford TQ, Roelink H. The notch response inhibitor DAPT enhances neuronal differentiation in embryonic stem cell-derived embryoid bodies independently of sonic hedgehog signaling. Dev Dyn. 2007;236(3):886–92.
Purow BW, Haque RM, Noel MW, et al. Expression of Notch-1 and its ligands, Delta-like-1 and Jagged-1, is critical for glioma cell survival and proliferation. Cancer Res. 2005;65(6):2353–63.
Kageyama R, Ohtsuka T, Kobayashi T. The Hes gene family: repressors and oscillators that orchestrate embryogenesis. Development. 2007;134(7):1243–51.
Liu X, Xu QR, Xie WF, et al. DAPT suppresses the proliferation of human glioma cell line SHG-44. Asian Pac J Trop Med. 2014;7(7):552–6.
Acknowledgments
This study was supported by a grant from the Beijing Municipal Science and Technology Commission (NO. Z151100004015213). We acknowledge the helpful comments on this paper received from our reviewers.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Additional information
Yi-Liang Bi and Min Min contributed equally to this work.
Rights and permissions
About this article
Cite this article
Bi, YL., Min, M., Shen, W. et al. Numb/Notch signaling pathway modulation enhances human pancreatic cancer cell radiosensitivity. Tumor Biol. 37, 15145–15155 (2016). https://doi.org/10.1007/s13277-016-5311-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-016-5311-8