Abstract
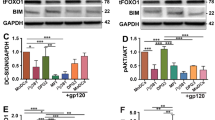
Periodontitis is the most common chronic inflammatory condition occurring in the human oral cavity, but our knowledge on its contribution to oral cancer is rather limited. To define crosstalk between chronic periodontitis and oral cancer, we investigated whether Porphyromonas gingivalis, a major pathogen of chronic periodontitis, plays a role in oral cancer progression. To mimic chronic irritation by P. gingivalis in the oral cavity, oral squamous cell carcinoma (OSCC) cells were infected with P. gingivalis twice a week for 5 weeks. Repeated infection of oral cancer cells by P. gingivalis resulted in morphological changes of host cancer cells into an elongated shape, along with the decreased expression of epithelial cell markers, suggesting acquisition of an epithelial-to-mesenchymal transition (EMT) phenotype. The prolonged exposure to P. gingivalis also promoted migratory and invasive properties of OSCC cells and provided resistance against a chemotherapeutic agent, all of which are described as cellular characteristics undergoing EMT. Importantly, long-term infection by P. gingivalis induced an increase in the expression level of CD44 and CD133, well-known cancer stem cell markers, and promoted the tumorigenic properties of infected cancer cells compared to non-infected controls. Furthermore, increased invasiveness of P. gingivalis-infected OSCC cells was correlated with enhanced production of matrix metalloproteinase (MMP)-1 and MMP-10 that was stimulated by interleukin-8 (IL-8) release. This is the first report demonstrating that P. gingivalis can increase the aggressiveness of oral cancer cells via epithelial-mesenchymal transition-like changes and the acquisition of stemness, implicating P. gingivalis as a potential bacterial risk modifier.







Similar content being viewed by others
References
Beaugerie L, Svrcek M, Seksik P, Bouvier AM, Simon T, Allez M, et al. Risk of colorectal high-grade dysplasia and cancer in a prospective observational cohort of patients with inflammatory bowel disease. Gastroenterology. 2013;145(1):166–75. doi:10.1053/j.gastro.2013.03.044.
Hiotis SP, Rahbari NN, Villanueva GA, Klegar E, Luan W, Wang Q, et al. Hepatitis B vs hepatitis C infection on viral hepatitis-associated hepatocellular carcinoma. BMC Gastroenterol. 2012;12:64. doi:10.1186/1471-230X-12-64.
Natarajan E, Eisenberg E. Contemporary concepts in the diagnosis of oral cancer and precancer. Dent Clin North Am. 2011;55(1):63–88. doi:10.1016/j.cden.2010.08.006.
Ahn J, Segers S, Hayes RB. Periodontal disease, Porphyromonas gingivalis serum antibody levels and orodigestive cancer mortality. Carcinogenesis. 2012;33(5):1055–8. doi:10.1093/carcin/bgs112.
Tezal M, Sullivan MA, Hyland A, Marshall JR, Stoler D, Reid ME, et al. Chronic periodontitis and the incidence of head and neck squamous cell carcinoma. Cancer Epidemiol Biomarkers Prev. 2009;18(9):2406–12. doi:10.1158/1055-9965.EPI-09-0334.
Meyer MS, Joshipura K, Giovannucci E, Michaud DS. A review of the relationship between tooth loss, periodontal disease, and cancer. Cancer Causes Control. 2008;19(9):895–907. doi:10.1007/s10552-008-9163-4.
Fitzpatrick SG, Katz J. The association between periodontal disease and cancer: a review of the literature. J Dent. 2010;38(2):83–95. doi:10.1016/j.jdent.2009.10.007.
Seymour G, Ford P, Cullinan M, Leishman S, Yamazaki K. Relationship between periodontal infections and systemic disease. Clin Microbiol Infect. 2007;13(s4):3–10.
Pidgeon GP, Harmey JH, Kay E, Da Costa M, Redmond HP, Bouchier-Hayes DJ. The role of endotoxin/lipopolysaccharide in surgically induced tumour growth in a murine model of metastatic disease. Br J Cancer. 1999;81(8):1311–7. doi:10.1038/sj.bjc.6694369.
Taketomi A, Takenaka K, Matsumata T, Shimada M, Higashi H, Shirabe K, et al. Circulating intercellular adhesion molecule-1 in patients with hepatocellular carcinoma before and after hepatic resection. Hepatogastroenterology. 1997;44(14):477–83.
Mikami T, Cheng J, Maruyama S, Kobayashi T, Funayama A, Yamazaki M, et al. Emergence of keratin 17 vs. loss of keratin 13: their reciprocal immunohistochemical profiles in oral carcinoma in situ. Oral Oncol. 2011;47(6):497–503. doi:10.1016/j.oraloncology.2011.03.015.
Masui T, Ota I, Yook JI, Mikami S, Yane K, Yamanaka T, et al. Snail-induced epithelial-mesenchymal transition promotes cancer stem cell-like phenotype in head and neck cancer cells. Int J Oncol. 2014;44(3):693–9. doi:10.3892/ijo.2013.2225.
Wang X, Ling MT, Guan XY, Tsao SW, Cheung HW, Lee DT, et al. Identification of a novel function of TWIST, a bHLH protein, in the development of acquired taxol resistance in human cancer cells. Oncogene. 2004;23(2):474–82. doi:10.1038/sj.onc.1207128.
Vega S, Morales AV, Ocaña OH, Valdés F, Fabregat I, Nieto MA. Snail blocks the cell cycle and confers resistance to cell death. Genes Dev. 2004;18(10):1131–43.
Polyak K, Weinberg RA. Transitions between epithelial and mesenchymal states: acquisition of malignant and stem cell traits. Nat Rev Cancer. 2009;9(4):265–73. doi:10.1038/nrc2620.
Kong D, Li Y, Wang Z, Sarkar FH. Cancer stem cells and epithelial-to-mesenchymal transition (EMT)-phenotypic cells: are they cousins or twins? Cancers (Basel). 2011;3(1):716–29. doi:10.3390/cancers30100716.
Dean M, Fojo T, Bates S. Tumour stem cells and drug resistance. Nat Rev Cancer. 2005;5(4):275–84. doi:10.1038/nrc1590.
Shi C, Tian R, Wang M, Wang X, Jiang J, Zhang Z, et al. CD44+ CD133+ population exhibits cancer stem cell-like characteristics in human gallbladder carcinoma. Cancer Biol Ther. 2010;10(11):1182–90.
Prince ME, Sivanandan R, Kaczorowski A, Wolf GT, Kaplan MJ, Dalerba P, et al. Identification of a subpopulation of cells with cancer stem cell properties in head and neck squamous cell carcinoma. Proc Natl Acad Sci U S A. 2007;104(3):973–8. doi:10.1073/pnas.0610117104.
Okamoto A, Chikamatsu K, Sakakura K, Hatsushika K, Takahashi G, Masuyama K. Expansion and characterization of cancer stem-like cells in squamous cell carcinoma of the head and neck. Oral Oncol. 2009;45(7):633–9. doi:10.1016/j.oraloncology.2008.10.003.
Cao L, Zhou Y, Zhai B, Liao J, Xu W, Zhang R, et al. Sphere-forming cell subpopulations with cancer stem cell properties in human hepatoma cell lines. BMC Gastroenterol. 2011;11:71. doi:10.1186/1471-230X-11-71.
Chiou SH, Yu CC, Huang CY, Lin SC, Liu CJ, Tsai TH, et al. Positive correlations of Oct-4 and Nanog in oral cancer stem-like cells and high-grade oral squamous cell carcinoma. Clin Cancer Res. 2008;14(13):4085–95. doi:10.1158/1078-0432.CCR-07-4404.
Fernando RI, Castillo MD, Litzinger M, Hamilton DH, Palena C. IL-8 signaling plays a critical role in the epithelial–mesenchymal transition of human carcinoma cells. Cancer Res. 2011;71(15):5296–306.
Curran S, Murray G. Matrix metalloproteinases: molecular aspects of their roles in tumour invasion and metastasis. Eur J Cancer. 2000;36(13):1621–30.
Mauviel A. Cytokine regulation of metalloproteinase gene expression. J Cell Biochem. 1993;53(4):288–95.
Inoue K, Slaton JW, Eve BY, Kim SJ, Perrotte P, Balbay MD, et al. Interleukin 8 expression regulates tumorigenicity and metastases in androgen-independent prostate cancer. Clin Cancer Res. 2000;6(5):2104–19.
de Martel C, Ferlay J, Franceschi S, Vignat J, Bray F, Forman D, et al. Global burden of cancers attributable to infections in 2008: a review and synthetic analysis. Lancet Oncol. 2012;13(6):607–15. doi:10.1016/S1470-2045(12)70137-7.
Porta C, Riboldi E, Sica A. Mechanisms linking pathogens-associated inflammation and cancer. Cancer Lett. 2011;305(2):250–62. doi:10.1016/j.canlet.2010.10.012.
Parkin DM. The global health burden of infection-associated cancers in the year 2002. Int J Cancer. 2006;118(12):3030–44. doi:10.1002/ijc.21731.
Smith JS, Bosetti C, Muñoz N, Herrero R, Bosch FX, Eluf-Neto J, et al. Chlamydia trachomatis and invasive cervical cancer: a pooled analysis of the IARC multicentric case–control study. Int J Cancer. 2004;111(3):431–9. doi:10.1002/ijc.20257.
Kocazeybek B. Chronic Chlamydophila pneumoniae infection in lung cancer, a risk factor: a case–control study. J Med Microbiol. 2003;52(Pt 8):721–6.
Ellmerich S, Schöller M, Duranton B, Gossé F, Galluser M, Klein JP, et al. Promotion of intestinal carcinogenesis by Streptococcus bovis. Carcinogenesis. 2000;21(4):753–6.
Horikawa T, Yang J, Kondo S, Yoshizaki T, Joab I, Furukawa M, et al. Twist and epithelial-mesenchymal transition are induced by the EBV oncoprotein latent membrane protein 1 and are associated with metastatic nasopharyngeal carcinoma. Cancer Res. 2007;67(5):1970–8. doi:10.1158/0008-5472.CAN-06-3933.
Yang SZ, Zhang LD, Zhang Y, Xiong Y, Zhang YJ, Li HL, et al. HBx protein induces EMT through c-Src activation in SMMC-7721 hepatoma cell line. Biochem Biophys Res Commun. 2009;382(3):555–60. doi:10.1016/j.bbrc.2009.03.079.
Iqbal J, McRae S, Banaudha K, Mai T, Waris G. Mechanism of hepatitis C virus (HCV)-induced osteopontin and its role in epithelial to mesenchymal transition of hepatocytes. J Biol Chem. 2013;288(52):36994–7009. doi:10.1074/jbc.M113.492314.
Yin Y, Grabowska AM, Clarke PA, Whelband E, Robinson K, Argent RH, et al. Helicobacter pylori potentiates epithelial:mesenchymal transition in gastric cancer: links to soluble HB-EGF, gastrin and matrix metalloproteinase-7. Gut. 2010;59(8):1037–45. doi:10.1136/gut.2009.199794.
Chandrakesan P, Roy B, Jakkula LU, Ahmed I, Ramamoorthy P, Tawfik O, et al. Utility of a bacterial infection model to study epithelial-mesenchymal transition, mesenchymal-epithelial transition or tumorigenesis. Oncogene. 2014;33(20):2639–54. doi:10.1038/onc.2013.210.
Baud J, Varon C, Chabas S, Chambonnier L, Darfeuille F, Staedel C. Helicobacter pylori initiates a mesenchymal transition through ZEB1 in gastric epithelial cells. PLoS One. 2013;8(4), e60315. doi:10.1371/journal.pone.0060315.
Grünert S, Jechlinger M, Beug H. Diverse cellular and molecular mechanisms contribute to epithelial plasticity and metastasis. Nat Rev Mol Cell Biol. 2003;4(8):657–65. doi:10.1038/nrm1175.
Na DC, Lee JE, Yoo JE, Oh BK, Choi GH, Park YN. Invasion and EMT-associated genes are up-regulated in B viral hepatocellular carcinoma with high expression of CD133-human and cell culture study. Exp Mol Pathol. 2011;90(1):66–73. doi:10.1016/j.yexmp.2010.10.003.
Kong QL, Hu LJ, Cao JY, Huang YJ, Xu LH, Liang Y, et al. Epstein-Barr virus-encoded LMP2A induces an epithelial-mesenchymal transition and increases the number of side population stem-like cancer cells in nasopharyngeal carcinoma. PLoS Pathog. 2010;6(6), e1000940. doi:10.1371/journal.ppat.1000940.
Bessede E, Staedel C, Amador LA, Nguyen P, Chambonnier L, Hatakeyama M, et al. Helicobacter pylori generates cells with cancer stem cell properties via epithelial–mesenchymal transition-like changes. Oncogene. 2014;33(32):4123–31.
Oliveira MJ, Costa AC, Costa AM, Henriques L, Suriano G, Atherton JC, et al. Helicobacter pylori induces gastric epithelial cell invasion in a c-Met and type IV secretion system-dependent manner. J Biol Chem. 2006;281(46):34888–96. doi:10.1074/jbc.M607067200.
Wu CY, Wang CJ, Tseng CC, Chen HP, Wu MS, Lin JT, et al. Helicobacter pylori promote gastric cancer cells invasion through a NF-kappaB and COX-2-mediated pathway. World J Gastroenterol. 2005;11(21):3197–203.
Chang YJ, Wu MS, Lin JT, Chen CC. Helicobacter pylori-induced invasion and angiogenesis of gastric cells is mediated by cyclooxygenase-2 induction through TLR2/TLR9 and promoter regulation. J Immunol. 2005;175(12):8242–52.
Lan YY, Yeh TH, Lin WH, Wu SY, Lai HC, Chang FH, et al. Epstein-Barr virus Zta upregulates matrix metalloproteinases 3 and 9 that synergistically promote cell invasion in vitro. PLoS One. 2013;8(2), e56121. doi:10.1371/journal.pone.0056121.
Fravalo P, Menard C, Bonnaure-Mallet M. Effect of Porphyromonas gingivalis on epithelial cell MMP-9 type IV collagenase production. Infect Immun. 1996;64(12):4940–5.
Inaba H, Sugita H, Kuboniwa M, Iwai S, Hamada M, Noda T, et al. Porphyromonas gingivalis promotes invasion of oral squamous cell carcinoma through induction of proMMP9 and its activation. Cell Microbiol. 2014;16(1):131–45.
DeCarlo A, Windsor L, Bodden M, Harber G, Birkedal-Hansen B, Birkedal-Hansen H. Activation and novel processing of matrix metalloproteinases by a thiol-proteinase from the oral anaerobe Porphyromonas gingivalis. J Dent Res. 1997;76(6):1260–70.
Pattamapun K, Tiranathanagul S, Yongchaitrakul T, Kuwatanasuchat J, Pavasant P. Activation of MMP‐2 by Porphyromonas gingivalis in human periodontal ligament cells. J Periodontal Res. 2003;38(2):115–21.
Justilien V, Regala RP, Tseng I-C, Walsh MP, Batra J, Radisky ES, et al. Matrix metalloproteinase-10 is required for lung cancer stem cell maintenance, tumor initiation and metastatic potential. PLoS One. 2012;7(4), e35040.
Inaba H, Sugita H, Kuboniwa M, Iwai S, Hamada M, Noda T, et al. Porphyromonas gingivalis promotes invasion of oral squamous cell carcinoma through induction of proMMP9 and its activation. Cell Microbiol. 2014;16(1):131–45. doi:10.1111/cmi.12211.
Acknowledgments
This work was supported by Basic Science Research Program through National Research Foundation of Korea (NRF) funded by the Ministry of Education (2014R1A1A2A16050554).
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Additional information
Na Hee Ha and Bok Hee Woo contributed equally to this work.
Rights and permissions
About this article
Cite this article
Ha, N.H., Woo, B.H., Kim, D.J. et al. Prolonged and repetitive exposure to Porphyromonas gingivalis increases aggressiveness of oral cancer cells by promoting acquisition of cancer stem cell properties. Tumor Biol. 36, 9947–9960 (2015). https://doi.org/10.1007/s13277-015-3764-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-3764-9