Abstract
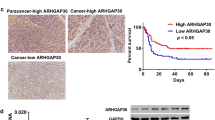
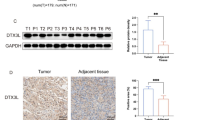
CRKL is an adapter protein which is overexpressed in many malignant tumors and plays crucial roles in tumor progression. However, expression pattern and biological roles of CRKL in pancreatic cancer have not been examined. In the present study, we found that CRKL expression in pancreatic cancer specimens was higher than that in normal pancreatic tissues. Colony formation assay and Matrigel invasion assay showed that the overexpression of CRKL in Bxpc3 and Capan2 cell lines with low endogenous expression increased cell proliferation and invasion. Flow cytometry showed that CRKL promoted cell proliferation by facilitating cell cycle. Further analysis of cell cycle- and invasion-related molecules showed that CRKL upregulated cyclin D1, cyclin A, matrix metalloproteinase 2 (MMP2) expression, and phosphorylated extracellular signal (ERK)-regulated kinase. In conclusion, our study demonstrated that CRKL was overexpressed in human pancreatic cancers and contributed to pancreatic cancer cell proliferation and invasion through ERK signaling.





Similar content being viewed by others
References
Bardeesy N, DePinho RA. Pancreatic cancer biology and genetics. Nat Rev Cancer. 2002;2(12):897–909.
Hidalgo M. Pancreatic cancer. N Engl J Med. 2010;362(17):1605–17.
Lillemoe KD, Yeo CJ, Cameron JL. Pancreatic cancer: state-of-the-art care. CA Cancer J Clin. 2000;50(4):241–68.
Siegel R et al. Cancer statistics, 2014. CA Cancer J Clin. 2014;64(1):9–29.
Rhodes J et al. CrkL functions as a nuclear adaptor and transcriptional activator in Bcr-Abl-expressing cells. Exp Hematol. 2000;28(3):305–10.
Feller SM. Crk family adaptors-signalling complex formation and biological roles. Oncogene. 2001;20(44):6348–71.
Fidler IJ, Kripke ML. Genomic analysis of primary tumors does not address the prevalence of metastatic cells in the population. Nat Genet. 2003;34(1):23. author reply 25.
ten Hoeve J et al. Isolation and chromosomal localization of CRKL, a human crk-like gene. Oncogene. 1993;8(9):2469–74.
ten Hoeve J et al. Cellular interactions of CRKL, and SH2-SH3 adaptor protein. Cancer Res. 1994;54(10):2563–7.
Beroukhim R et al. The landscape of somatic copy-number alteration across human cancers. Nature. 2010;463(7283):899–905.
Senechal K, Halpern J, Sawyers CL. The CRKL adaptor protein transforms fibroblasts and functions in transformation by the BCR-ABL oncogene. J Biol Chem. 1996;271(38):23255–61.
van’t Veer LJ et al. Gene expression profiling predicts clinical outcome of breast cancer. Nature. 2002;415(6871):530–6.
Cheung HW et al. Amplification of CRKL induces transformation and epidermal growth factor receptor inhibitor resistance in human non-small cell lung cancers. Cancer Discov. 2011;1(7):608–25.
Reebye V et al. Intracellular adaptor molecules and AR signalling in the tumour microenvironment. Cell Signal. 2011;23(6):1017–21.
Mintz PJ et al. An unrecognized extracellular function for an intracellular adapter protein released from the cytoplasm into the tumor microenvironment. Proc Natl Acad Sci U S A. 2009;106(7):2182–7.
Singer CF et al. Active (p)CrkL is overexpressed in human malignancies: potential role as a surrogate parameter for therapeutic tyrosine kinase inhibition. Oncol Rep. 2006;15(2):353–9.
Kim YH et al. Genomic and functional analysis identifies CRKL as an oncogene amplified in lung cancer. Oncogene. 2010;29(10):1421–30.
Fathers KE et al. Crk adaptor proteins act as key signaling integrators for breast tumorigenesis. Breast Cancer Res. 2012;14(3):R74.
Hanashiro K et al. Roles of cyclins A and E in induction of centrosome amplification in p53-compromised cells. Oncogene. 2008;27(40):5288–302.
Jezierska A, Motyl T. Matrix metalloproteinase-2 involvement in breast cancer progression: a mini-review. Med Sci Monit. 2009;15(2):RA32–40.
Tokuraku M et al. Activation of the precursor of gelatinase A/72 kDa type IV collagenase/MMP-2 in lung carcinomas correlates with the expression of membrane-type matrix metalloproteinase (MT-MMP) and with lymph node metastasis. Int J Cancer. 1995;64(5):355–9.
Kenny HA et al. The initial steps of ovarian cancer cell metastasis are mediated by MMP-2 cleavage of vitronectin and fibronectin. J Clin Invest. 2008;118(4):1367–79.
Murnane MJ et al. Active MMP-2 effectively identifies the presence of colorectal cancer. Int J Cancer. 2009;125(12):2893–902.
Fernandez-Medarde A, Santos E. Ras in cancer and developmental diseases. Genes Cancer. 2011;2(3):344–58.
Dong QZ et al. Derlin-1 is overexpressed in non-small cell lung cancer and promotes cancer cell invasion via EGFR-ERK-mediated up-regulation of MMP-2 and MMP-9. Am J Pathol. 2013;182(3):954–64.
Uemura N, Griffin JD. The adapter protein Crkl links Cbl to C3G after integrin ligation and enhances cell migration. J Biol Chem. 1999;274(53):37525–32.
Sebolt-Leopold JS, Herrera R. Targeting the mitogen-activated protein kinase cascade to treat cancer. Nat Rev Cancer. 2004;4(12):937–47.
Webb CP et al. Signaling pathways in ras-mediated tumorigenicity and metastasis. Proc Natl Acad Sci U S A. 1998;95(15):8773–8.
Lin, F., et al. CRKL promotes lung cancer cell invasion through ERK-MMP9 pathway. Mol Carcinog, 2014.
Wilson MB et al. Selective pyrrolo-pyrimidine inhibitors reveal a necessary role for Src family kinases in Bcr-Abl signal transduction and oncogenesis. Oncogene. 2002;21(53):8075–88.
Ito H et al. Prostaglandin E2 enhances pancreatic cancer invasiveness through an Ets-1-dependent induction of matrix metalloproteinase-2. Cancer Res. 2004;64(20):7439–46.
Han F, Zhu HG. Caveolin-1 regulating the invasion and expression of matrix metalloproteinase (MMPs) in pancreatic carcinoma cells. J Surg Res. 2010;159(1):443–50.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fu, L., Dong, Q., Xie, C. et al. CRKL protein overexpression enhances cell proliferation and invasion in pancreatic cancer. Tumor Biol. 36, 1015–1022 (2015). https://doi.org/10.1007/s13277-014-2706-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-014-2706-2