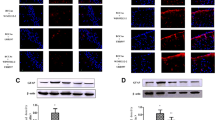
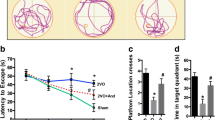
Abstract
Chronic cerebral hypoperfusion (CCH) is a major contributor to cognitive decline and degenerative processes leading to Alzheimer’s disease, vascular dementia, and aging. However, the delicate mechanism of CCH-induced neuronal damage, and therefore proper treatment, remains unclear. WIN55,212-2 (WIN) is a nonselective cannabinoid receptor agonist that has been shown to have effects on hippocampal neuron survival. In this study, we investigated the potential roles of WIN, as well as its underlying mechanism in a rat CCH model of bilateral common carotid artery occlusion. Hippocampal morphological changes and mitochondrial ultrastructure were detected using hematoxylin and eosin staining and electron microscopy, respectively. Various biomarkers, such as reactive oxidative species (ROS), superoxide dismutase (SOD), catalase (CAT), and malondialdehyde (MDA) were used to assess the level of oxidative stress in the hippocampus. Furthermore, the expression levels of neuronal nuclei (NeuN), apoptosis signal-regulating kinase 1 (ASK1)-p38 signaling proteins, cleaved Caspase-9 and -3, and cytochrome-c (Cyt-C) were accessed by western blotting. CCH decreased the levels of NeuN, Cyt-C (mitochondrial), SOD, and CAT, and increased the levels of MDA, phosphorylated ASK1 and phosphorylated p38, cleaved Caspase-9 and -3, and Cyt-C (cytoplasm), which were reversed by WIN treatment. Chronic treatment with WIN also improved CCH-induced neuronal degeneration and mitochondrial fragmentation. These findings indicated that WIN may be a potential therapeutic agent for ischemic neuronal damage, involving a mechanism associated with the suppression of oxidative stress and ASK1-p38 signaling.




Similar content being viewed by others
References
Akhter F, Chen D, Yan SF, Yan SS (2017) Mitochondrial perturbation in Alzheimer's disease and diabetes. Prog Mol Biol Transl Sci 146:341–361. https://doi.org/10.1016/bs.pmbts.2016.12.019
Arrazola MS, Ramos-Fernandez E, Cisternas P, Ordenes D, Inestrosa NC (2017) Wnt Signaling prevents the Abeta oligomer-induced mitochondrial permeability transition pore opening preserving mitochondrial structure in hippocampal neurons. PLoS One 12:e0168840. https://doi.org/10.1371/journal.pone.0168840
Cenini G, Lloret A, Cascella R (2019) Oxidative stress in neurodegenerative diseases: from a mitochondrial point of view. Oxidative Med Cell Longev 2019:2105607. https://doi.org/10.1155/2019/2105607
Chang CY, Liang MZ, Chen L (2019) Current progress of mitochondrial transplantation that promotes neuronal regeneration. Transl Neurodegener 8:17. https://doi.org/10.1186/s40035-019-0158-8
Cheon SY, Cho KJ, Kim SY, Kam EH, Lee JE, Koo BN (2016a) Blockade of apoptosis signal-regulating kinase 1 attenuates matrix metalloproteinase 9 activity in brain endothelial cells and the subsequent apoptosis in neurons after ischemic injury. Front Cell Neurosci 10:213. https://doi.org/10.3389/fncel.2016.00213
Cheon SY, Cho KJ, Song J, Kim GW (2016b) Knockdown of apoptosis signal-regulating kinase 1 affects ischaemia-induced astrocyte activation and glial scar formation. Eur J Neurosci 43:912–922. https://doi.org/10.1111/ejn.13175
Cheon SY, Kim EJ, Kim JM, Koo BN (2018) Cell type-specific mechanisms in the pathogenesis of ischemic stroke: the role of apoptosis signal-regulating kinase 1. Oxidative Med Cell Longev 2018:2596043. https://doi.org/10.1155/2018/2596043
Chi OZ, Barsoum S, Grayson J, Hunter C, Liu X, Weiss HR (2012) Effects of cannabinoid receptor agonist WIN 55,212-2 on blood-brain barrier disruption in focal cerebral ischemia in rats. Pharmacology 89:333–338. https://doi.org/10.1159/000338755
Dallak M, Al-Ani B, Abdel Kader DH, Eid RA, Haidara MA (2019) Insulin suppresses type 1 diabetes mellitus-induced ventricular cardiomyocyte damage associated with the inhibition of biomarkers of inflammation and oxidative stress in rats. Pharmacology 1–9. https://doi.org/10.1159/000500898
Daulatzai MA (2017) Cerebral hypoperfusion and glucose hypometabolism: key pathophysiological modulators promote neurodegeneration, cognitive impairment, and Alzheimer's disease. J Neurosci Res 95:943–972. https://doi.org/10.1002/jnr.23777
Duncombe J, Kitamura A, Hase Y, Ihara M, Kalaria RN, Horsburgh K (2017) Chronic cerebral hypoperfusion: a key mechanism leading to vascular cognitive impairment and dementia. Closing the translational gap between rodent models and human vascular cognitive impairment and dementia. Clin Sci (Lond) 131:2451–2468. https://doi.org/10.1042/CS20160727
Fernandez-Lopez D, Pradillo JM, Garcia-Yebenes I, Martinez-Orgado JA, Moro MA, Lizasoain I (2010) The cannabinoid WIN55212-2 promotes neural repair after neonatal hypoxia-ischemia. Stroke 41:2956–2964. https://doi.org/10.1161/STROKEAHA.110.599357
Fernandez-Ruiz J, Hernandez M, Ramos JA (2010) Cannabinoid-dopamine interaction in the pathophysiology and treatment of CNS disorders. CNS Neurosci Ther 16:e72–e91. https://doi.org/10.1111/j.1755-5949.2010.00144.x
Flameng W, Borgers M, Daenen W, Stalpaert G (1980) Ultrastructural and cytochemical correlates of myocardial protection by cardiac hypothermia in man. J Thorac Cardiovasc Surg 79:413–424
Gamdzyk M, Doycheva DM, Malaguit J, Enkhjargal B, Tang J, Zhang JH (2018) Role of PPAR-beta/delta/miR-17/TXNIP pathway in neuronal apoptosis after neonatal hypoxic-ischemic injury in rats. Neuropharmacology 140:150–161. https://doi.org/10.1016/j.neuropharm.2018.08.003
Gao Y, Tang H, Nie K, Zhu R, Gao L, Feng S, Wang L, Zhao J, Huang Z, Zhang Y, Wang L (2019) Hippocampal damage and white matter lesions contribute to cognitive impairment in MPTP-lesioned mice with chronic cerebral hypoperfusion. Behav Brain Res 368:111885. https://doi.org/10.1016/j.bbr.2019.03.054
Goncalves ED, Dutra RC (2019) Cannabinoid receptors as therapeutic targets for autoimmune diseases: where do we stand? Drug Discov Today. https://doi.org/10.1016/j.drudis.2019.05.023
Gooch J, Wilcock DM (2016) Animal models of vascular cognitive impairment and dementia (VCID). Cell Mol Neurobiol 36:233–239. https://doi.org/10.1007/s10571-015-0286-3
Haghani M, Shabani M, Javan M, Motamedi F, Janahmadi M (2012) CB1 cannabinoid receptor activation rescues amyloid beta-induced alterations in behaviour and intrinsic electrophysiological properties of rat hippocampal CA1 pyramidal neurones. Cell Physiol Biochem 29:391–406. https://doi.org/10.1159/000338494
Hai J, Lin Q, Li ST, Pan QG (2004) Chronic cerebral hypoperfusion and reperfusion injury of restoration of normal perfusion pressure contributes to the neuropathological changes in rat brain. Brain Res Mol Brain Res 126:137–145. https://doi.org/10.1016/j.molbrainres.2004.04.004
Hai J, Yu F, Lin Q, Su SH (2012) The changes of signal transduction pathways in hippocampal regions and postsynaptic densities after chronic cerebral hypoperfusion in rats. Brain Res 1429:9–17. https://doi.org/10.1016/j.brainres.2011.10.023
Huizenga MN, Forcelli PA (2019) Neuroprotective action of the CB1/2 receptor agonist, WIN 55,212-2, against DMSO but not phenobarbital-induced neurotoxicity in immature rats. Neurotox Res 35:173–182. https://doi.org/10.1007/s12640-018-9944-9
Ilyasov AA, Milligan CE, Pharr EP, Howlett AC (2018) The endocannabinoid system and oligodendrocytes in health and disease. Front Neurosci 12:733. https://doi.org/10.3389/fnins.2018.00733
Inaba T et al (2019) Protective role of levetiracetam against cognitive impairment and brain white matter damage in mouse prolonged cerebral hypoperfusion. Neuroscience 414:255–264. https://doi.org/10.1016/j.neuroscience.2019.07.015
Jing H, Yan H, Guo D, Zhiqiang W, Xiang Y, Qingsong W (2019) Lasting spatial learning and memory deficits following chronic cerebral hypoperfusion are associated with hippocampal mitochondrial aging in rats. Neuroscience. https://doi.org/10.1016/j.neuroscience.2019.04.044
Kho AR et al (2018) Effects of Protocatechuic acid (PCA) on global cerebral ischemia-induced hippocampal neuronal death. Int J Mol Sci 19. https://doi.org/10.3390/ijms19051420
Kirschmann EK, McCalley DM, Edwards CM, Torregrossa MM (2017) Consequences of adolescent exposure to the cannabinoid receptor agonist WIN55,212-2 on working memory in female rats. Front Behav Neurosci 11:137. https://doi.org/10.3389/fnbeh.2017.00137
Kirschmann EK, Pollock MW, Nagarajan V, Torregrossa MM (2019) Development of working memory in the male adolescent rat. Dev Cogn Neurosci 37:100601. https://doi.org/10.1016/j.dcn.2018.11.003
Li L, Zuo Z (2011) Isoflurane postconditioning induces neuroprotection via Akt activation and attenuation of increased mitochondrial membrane permeability. Neuroscience 199:44–50. https://doi.org/10.1016/j.neuroscience.2011.10.022
Li P et al (2018) Oxidative stress and DNA damage after cerebral ischemia: potential therapeutic targets to repair the genome and improve stroke recovery. Neuropharmacology 134:208–217. https://doi.org/10.1016/j.neuropharm.2017.11.011
Li L et al (2019) Polydatin prevents the induction of secondary brain injury after traumatic brain injury by protecting neuronal mitochondria. Neural Regen Res 14:1573–1582. https://doi.org/10.4103/1673-5374.255972
Lin B, Xu J, Feng DG, Wang F, Wang JX, Zhao H (2018) DUSP14 knockout accelerates cardiac ischemia reperfusion (IR) injury through activating NF-kappaB and MAPKs signaling pathways modulated by ROS generation. Biochem Biophys Res Commun 501:24–32. https://doi.org/10.1016/j.bbrc.2018.04.101
Marchalant Y, Brothers HM, Wenk GL (2009) Cannabinoid agonist WIN-55,212-2 partially restores neurogenesis in the aged rat brain. Mol Psychiatry 14:1068–1069. https://doi.org/10.1038/mp.2009.62
Perez-Pinzon MA, Xu GP, Born J, Lorenzo J, Busto R, Rosenthal M, Sick TJ (1999) Cytochrome C is released from mitochondria into the cytosol after cerebral anoxia or ischemia. J Cereb Blood Flow Metab 19:39–43. https://doi.org/10.1097/00004647-199901000-00004
Pertwee RG (2006) The pharmacology of cannabinoid receptors and their ligands: an overview. Int J Obes 30(Suppl 1):S13–S18. https://doi.org/10.1038/sj.ijo.0803272
Rose J, Brian C, Woods J, Pappa A, Panayiotidis MI, Powers R, Franco R (2017) Mitochondrial dysfunction in glial cells: implications for neuronal homeostasis and survival. Toxicology 391:109–115. https://doi.org/10.1016/j.tox.2017.06.011
Saez TM, Aronne MP, Caltana L, Brusco AH (2014) Prenatal exposure to the CB1 and CB2 cannabinoid receptor agonist WIN 55,212-2 alters migration of early-born glutamatergic neurons and GABAergic interneurons in the rat cerebral cortex. J Neurochem 129:637–648. https://doi.org/10.1111/jnc.12636
Santabarbara-Ruiz P et al (2019) Ask1 and Akt act synergistically to promote ROS-dependent regeneration in Drosophila. PLoS Genet 15:e1007926. https://doi.org/10.1371/journal.pgen.1007926
Sbarski B, Akirav I (2018) Chronic exposure to cannabinoids before an emotional trauma may have negative effects on emotional function. Eur Neuropsychopharmacol 28:955–969. https://doi.org/10.1016/j.euroneuro.2018.05.008
Serita T et al (2019) Dietary magnesium deficiency impairs hippocampus-dependent memories without changes in the spine density and morphology of hippocampal neurons in mice. Brain Res Bull 144:149–157. https://doi.org/10.1016/j.brainresbull.2018.11.019
Sharon H, Brill S (2019) Cannabis-based medicines for chronic pain management: current and future prospects. Curr Opin Anaesthesiol 32:623–628. https://doi.org/10.1097/ACO.0000000000000775
Su SH, Wu YF, Lin Q, Yu F, Hai J (2015) Cannabinoid receptor agonist WIN55,212-2 and fatty acid amide hydrolase inhibitor URB597 suppress chronic cerebral hypoperfusion-induced neuronal apoptosis by inhibiting c-Jun N-terminal kinase signaling. Neuroscience 301:563–575. https://doi.org/10.1016/j.neuroscience.2015.03.021
Su SH, Wang YQ, Wu YF, Wang DP, Lin Q, Hai J (2016) Cannabinoid receptor agonist WIN55,212-2 and fatty acid amide hydrolase inhibitor URB597 may protect against cognitive impairment in rats of chronic cerebral hypoperfusion via PI3K/AKT signaling. Behav Brain Res 313:334–344. https://doi.org/10.1016/j.bbr.2016.07.009
Su BX, Chen X, Huo J, Guo SY, Ma R, Liu YW (2017a) The synthetic cannabinoid WIN55212-2 ameliorates traumatic spinal cord injury via inhibition of GAPDH/Siah1 in a CB2-receptor dependent manner. Brain Res 1671:85–92. https://doi.org/10.1016/j.brainres.2017.06.020
Su SH, Wu YF, Lin Q, Hai J (2017b) Cannabinoid receptor agonist WIN55,212-2 and fatty acid amide hydrolase inhibitor URB597 ameliorate neuroinflammatory responses in chronic cerebral hypoperfusion model by blocking NF-kappaB pathways. Naunyn Schmiedeberg's Arch Pharmacol 390:1189–1200. https://doi.org/10.1007/s00210-017-1417-9
Su SH, Wu YF, Wang DP, Hai J (2018) Inhibition of excessive autophagy and mitophagy mediates neuroprotective effects of URB597 against chronic cerebral hypoperfusion. Cell Death Dis 9:733. https://doi.org/10.1038/s41419-018-0755-y
Sun J et al (2013) WIN55, 212-2 promotes differentiation of oligodendrocyte precursor cells and improve remyelination through regulation of the phosphorylation level of the ERK 1/2 via cannabinoid receptor 1 after stroke-induced demyelination. Brain Res 1491:225–235. https://doi.org/10.1016/j.brainres.2012.11.006
Venkat P, Chopp M, Chen J (2015) Models and mechanisms of vascular dementia. Exp Neurol 272:97–108. https://doi.org/10.1016/j.expneurol.2015.05.006
Wang D, Lin Q, Su S, Liu K, Wu Y, Hai J (2017a) URB597 improves cognitive impairment induced by chronic cerebral hypoperfusion by inhibiting mTOR-dependent autophagy. Neuroscience 344:293–304. https://doi.org/10.1016/j.neuroscience.2016.12.034
Wang DP, Liu KJ, Kasper G, Lin Q, Hai J (2017b) Inhibition of SENP3 by URB597 ameliorates neurovascular unit dysfunction in rats with chronic cerebral hypoperfusion. Biomed Pharmacother 91:872–879. https://doi.org/10.1016/j.biopha.2017.05.021
Wang DP, Yin H, Kang K, Lin Q, Su SH, Hai J (2018) The potential protective effects of cannabinoid receptor agonist WIN55,212-2 on cognitive dysfunction is associated with the suppression of autophagy and inflammation in an experimental model of vascular dementia. Psychiatry Res 267:281–288. https://doi.org/10.1016/j.psychres.2018.06.012
Wang DP et al (2019) Andrographolide enhances hippocampal BDNF signaling and suppresses neuronal apoptosis, astroglial activation, neuroinflammation, and spatial memory deficits in a rat model of chronic cerebral hypoperfusion. Naunyn Schmiedeberg's Arch Pharmacol. https://doi.org/10.1007/s00210-019-01672-9
Winkelheide U, Engelhard K, Kaeppel B, Winkler J, Hutzler P, Werner C, Kochs E (2008) Cerebral ischemia and neurogenesis: a two-time comparison. Neurocrit Care 9:387–393. https://doi.org/10.1007/s12028-008-9121-8
Xie YC, Yao ZH, Yao XL, Pan JZ, Zhang SF, Zhang Y, Hu JC (2018) Glucagon-like peptide-2 receptor is involved in spatial cognitive dysfunction in rats after chronic cerebral hypoperfusion. J Alzheimers Dis 66:1559–1576. https://doi.org/10.3233/JAD-180782
Xie X et al (2019) Prostaglandin E1 alleviates cognitive dysfunction in chronic cerebral hypoperfusion rats by improving hemodynamics. Front Neurosci 13:549. https://doi.org/10.3389/fnins.2019.00549
Xue LX et al (2017) Neuroprotective effects of Activin A on endoplasmic reticulum stress-mediated apoptotic and autophagic PC12 cell death. Neural Regen Res 12:779–786. https://doi.org/10.4103/1673-5374.206649
Yang JL, Mukda S, Chen SD (2018) Diverse roles of mitochondria in ischemic stroke. Redox Biol 16:263–275. https://doi.org/10.1016/j.redox.2018.03.002
Zhang Q, Zhang G (2002) Activation and autophosphorylation of apoptosis signal-regulating kinase 1 (ASK1) following cerebral ischemia in rat hippocampus. Neurosci Lett 329:232–236
Zhang S et al (2016) Chronic intermittent hybobaric hypoxia protects against cerebral ischemia via modulation of mitoKATP. Neurosci Lett 635:8–16. https://doi.org/10.1016/j.neulet.2016.10.025
Zhu W et al (2018) Anti-oxidative and anti-apoptotic effects of acupuncture: role of thioredoxin-1 in the hippocampus of vascular dementia rats. Neuroscience 379:281–291. https://doi.org/10.1016/j.neuroscience.2018.03.029
Funding
The study was supported by the National Nature Science Foundation of China (Grant Nos. 81771410, 81860664).
Author information
Authors and Affiliations
Contributions
Methodology: Kai Kang; Formal analysis and investigation: Qi Lin and Kai-Yan Jin; Writing - original draft preparation: Da-Peng Wang; Writing - review and editing: Qiao-Li Lv; Supervision: Da-Peng Wang and Jian Hai.
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, DP., Lv, QL., Lin, Q. et al. The Cannabinoid Receptor Agonist WIN55,212-2 Ameliorates Hippocampal Neuronal Damage After Chronic Cerebral Hypoperfusion Possibly Through Inhibiting Oxidative Stress and ASK1-p38 Signaling. Neurotox Res 37, 847–856 (2020). https://doi.org/10.1007/s12640-019-00141-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-019-00141-8