Abstract
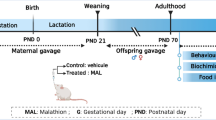
It has been proposed that developmental exposure to pesticides contributes to increasing prevalence of neurodevelopmental disorders in children, such as attention deficit with hyperactivity (ADHD) and to alterations in coordination skills. However, the mechanisms involved in these alterations remain unclear. We analyzed the effects on spontaneous motor activity and motor coordination of developmental exposure to a representative pesticide of each one of the four main chemical families: organophosphates (chlorpyrifos), carbamates (carbaryl), organochlorines (endosulfan), and pyrethroids (cypermethrin). Pesticides were administered once a day orally, in a sweet jelly, from gestational day 7 to post natal day 21. Spontaneous motor activity was assessed by an actimeter and motor coordination using the rotarod, when rats were adults. The effects were analyzed separately in males and females. Extracellular GABA in cerebellum and NMDA receptor subunits in hippocampus were assessed as possible underlying mechanisms of motor alterations. Motor coordination was impaired by developmental exposure to endosulfan, cypermethrin, and chlorpyrifos in females but not in males. The effect of endosulfan and cypermethrin would be due to increased extracellular GABA in cerebellum, which remains unaltered in male rats. Chlorpyrifos increased motor activity in males and females. Cypermethrin decreased motor activity mainly in males. In male rats, but not in females, expression of the NR2B subunit of NMDA receptor in hippocampus correlated with motor activity. These results show sex-specific effects of different pesticides on motor activity and coordination, associated with neurotransmission alterations. These data contribute to better understand the relationship between developmental exposure to the main pesticide families and motor disorders in children.





Similar content being viewed by others
References
Banji D, Banji OJ, Ragini M, Annamalai AR (2014) Carbosulfan exposure during embryonic period can cause developmental disability in rats. Environ Toxicol Pharmacol 38(1):230–238. https://doi.org/10.1016/j.etap.2014.05.009
Barr JL, Forster GL, Unterwald EM (2014) Repeated cocaine enhances ventral hippocampal-stimulated dopamine efflux in the nucleus accumbens and alters ventral hippocampal NMDA receptor subunit expression. J Neurochem 130(4):583–590. https://doi.org/10.1111/jnc.12764
Boix J, Cauli O, Felipo V (2010a) Developmental exposure to polychlorinated biphenyls 52, 138 or 180 affects differentially learning or motor coordination in adult rats. Mech Involved Neurosci 167:994–1003
Boix J, Cauli O, Leslie H, Felipo V (2010b) Differential long-term effects of developmental exposure to polychlorinated biphenyls 52, 138 or 180 on motor activity and neurotransmission. Gender dependence and mechanisms involved. Neurochem Int 58(1):69–77. https://doi.org/10.1016/j.neuint.2010.10.014
Bouchard MF, Chevrier J, Harley KG, Kogut K, Vedar M, Calderon N, Trujillo C, Johnson C, Bradman A, Barr DB et al (2011) Prenatal exposure to organophosphate pesticides and IQ in 7-year-old children. Environ Health Perspect 119(8):1189–1195
Brudzynski SM, Gibson CJ (1997) Release of dopamine in the nucleus accumbens caused by stimulation of the subiculum in freely moving rats. Brain Res Bull 42(4):303–308
Burns CJ, McIntosh LJ, Mink PJ, Jurek AM, Li AA (2013) Pesticide exposure and neurodevelopmental outcomes: review of the epidemiologic and animal studies. J Toxicol Environ Health B Crit Rev 16((3–4)):127–283. https://doi.org/10.1080/10937404.2013.783383 Review. Erratum in: J Toxicol Environ Health B Crit Rev. 2013; 16(6):395–8
Cabaleiro T, Caride A, Romero A, Lafuente A (2008) Effects of in utero and lactational exposure to endosulfan in prefrontal cortex of male rats. Toxicol Lett 176(1):58–67
Carr RL, Chambers HW, Guarisco JA, Richardson JR, Tang J, Chambers JE (2001) Effects of repeated oral postnatal exposure to chlorpyrifos on open-field behavior in juvenile rats. Toxicol Sci 59(2):260–267
Carter RJ, Morton J, Dunnett SB. (2001). Motor coordination and balance in rodents. Curr Protoc Neurosci. Chapter 8: Unit 8.12. https://doi.org/10.1002/0471142301.ns0812s15
Cauli O, Mansouri MT, Agusti A, Felipo V (2009a) Hyperammonemia increases GABAergic tone in the cerebellum but decreases it in the rat cortex. Gastroenterology 136:1359–1367
Cauli O, Rodrigo R, Piedrafita B, Llansola M, Mansouri MT, Felipo V (2009b) Neuroinflammation contributes to hypokinesia in rats with hepatic encephalopathy. Ibuprofen restores its motor activity. J Neurosci Res 87:1369–1374
Cauli O, Piedrafita B, Llansola M, Felipo V (2013) Gender differential effects of developmental exposure to methyl-mercury, polychlorinated biphenyls 126 or 153, or its combinations on motor activity and coordination. Toxicology 311(1–2):61–68
Chiu CS, Brickley S, Jensen K, Southwell A, Mckinney S, Cull-Candy S, Mody I, Lester HA (2005) GABA transporter deficiency causes tremor, ataxia, nervousness, and increased GABA-induced tonic conductance in cerebellum. J Neurosci 25:3234–3245
Dalsenter PR, de Araújo SL, de Assis HC, Andrade AJ, Dallegrave E (2003) Pre and postnatal exposure to endosulfan in Wistar rats. Hum Exp Toxicol 22(4):171–175
Dam K, Seidler FJ, Slotkin TA (2000) Chlorpyrifos exposure during a critical neonatal period elicits gender-selective deficits in the development of coordination skills and locomotor activity. Brain Res Dev Brain Res 121(2):179–187
Dong W, Gilmour MI, Lambert AL, Selgrade MK (1998) Enhanced allergic responses to house dust mite by oral exposure to carbaryl in rats. Toxicol Sci 44(1):63–69
Ostrea Jr EM, Reyes A, Villanueva-Uy E, Pacifico R, Benitez B, Ramos E, Bernardo RC, Bielawski DM, Delaney-Black V, Chiodo L, Janisse JJ, Ager JW (2012) Fetal exposure to propoxur and abnormal child neurodevelopment at 2 years of age. NeuroToxicology 33 (4):669-675
Felipo V, Minana MD, Grisolia S (1988) Long term ingestion of ammonium increases acetylglutamate and urea levels without affecting the amount of carbamyl phosphate synthase. Eur J Biochem 176:567–571
Grandjean P, Landrigan PJ (2014) Neurobehavioural effects of developmental toxicity. Lancet Neurol 13(3):330–338
Guillette EA, Meza MM, Aquilar MG, Soto AD, Garcia IE (1998) An anthropological approach to the evaluation of preschool children exposed to pesticides in Mexico. Environ Health Perspect 106(6):347–353
Hanchar HJ, Dodson PD, Olsen RW, Otis TS, Wallner M (2005) Alcohol-induced motor impairment caused by increased extrasynaptic GABAA receptor activity. Nat Neurosci 8:339–345
Harari R, Julvez J, Murata K, Barr D, Bellinger DC, Debes F, Grandjean P (2010) Neurobehavioral deficits and increased blood pressure in school-age children prenatally exposed to pesticides. Environ Health Perspect 118(6):890–896. https://doi.org/10.1289/ehp.0901582
Hermenegildo C, Montoliu C, Llansola M, Munoz MD, Gaztelu JM, Minana MD, Felipo V (1998) Chronic hyperammonemia impairs the glutamate-nitric oxide-cyclic GMP pathway in cerebellar neurons in culture and in the rat in vivo. Eur J Neurosci 10:3201–3209
Hönack D, Löscher W (1993) Sex differences in NMDA receptor mediated responses in rats. Brain Res 620(1):167–170
Jensen V, Rinholm JE, Johansen TJ, Medin T, Storm-Mathisen J, Sagvolden T, Hvalby O, Bergersen LH (2009) N-Methyl-D-aspartate receptor subunit dysfunction at hippocampal glutamatergic synapses in an animal model of attention-deficit/hyperactivity disorder. Neuroscience 158(1):353–364. https://doi.org/10.1016/j.neuroscience.2008.05.016
Jones BJ, Roberts DJ (1968) The quantiative measurement of motor inco-ordination in naive mice using an acelerating rotarod. J Pharm Pharmacol 20:302–304
Kamboj A, Sandhir R (2007) Perturbed synaptosomal calcium homeostasis and behavioral deficits following carbofuranexposure: neuroprotection by N-acetylcysteine. Neurochem Res 32(3):507–516
Kim JH, Vezina P (1997) Activation of metabotropic glutamate receptors in the rat nucleus accumbens increases locomotor activity in a dopamine-dependent manner. J Pharmacol Exp Ther 283:962–968
Koirala B, Alele PE, Devaud LL (2008) Influence of hormonal status on behavioral responses to an acute ethanol challenge during ethanol withdrawal in male and female rats. Pharmacol Biochem Behav 90(4):691–700. https://doi.org/10.1016/j.pbb.2008.05.013
Kovács AD, Pearcel DA (2013) Location- and sex-specific differences in weight and motor coordination in two commonly used mouse strains. Scientific Reports 3:2116. https://doi.org/10.1038/srep02116
Lee I, Eriksson P, Fredriksson A, Buratovic S, Viberg H (2015) Developmental neurotoxic effects of two pesticides: behavior and neuroprotein studies on endosulfan and cypermethrin. Toxicology 335:1–10. https://doi.org/10.1016/j.tox.2015.06.010
Lemaire G, Mnif W, Mauvais P, Balaguer P, Rahmani R (2006) Activation of alpha- and beta-estrogen receptors by persistent pesticides in reporter cell lines. Life Sci 79(12):1160–1169
Levin ED, Addy N, Baruah A, Elias A, Christopher NC, Seidler FJ, Slotkin TA (2002) Prenatal chlorpyrifos exposure in rats causes persistent behavioral alterations. Neurotoxicol Teratol 24(6):733–741
Li AA, Lowe KA, McIntosh LJ, Mink PJ (2012) Evaluation of epidemiology and animal data for riskassessment: chlorpyrifos developmental neurobehavioral outcomes. J Toxicol Environ Health B Crit Rev 15(2):109–184. https://doi.org/10.1080/10937404.2012.645142 Review
Mansour SA, Mossa AH (2010) Adverse effects of lactational exposure to chlorpyrifos in suckling rats. Hum Exp Toxicol. 29(2):77–92. https://doi.org/10.1177/0960327109357276
Mansouri MT, Naghizadeh B, López-Larrubia P, Cauli O (2013) Behavioral deficits induced by lead exposure are accompanied by serotonergic and cholinergic alterations in the prefrontal cortex. Neurochem Int 62(3):232–239. https://doi.org/10.1016/j.neuint.2012.12.009
McDaniel KL, Padilla S, Marshall RS, Phillips PM, Podhorniak L, Qian Y, Moser VC (2007) Comparison of acute neurobehavioral and cholinesterase inhibitory effects of N-methylcarbamates in rat. Toxicol Sci 98(2):552–560
Moser VC, Phillips PM, McDaniel KL (2015) Assessment of biochemical and behavioral effects of carbaryl and methomyl in Brown-Norway rats from preweaning to senescence. Toxicology 331:1–13. https://doi.org/10.1016/j.tox.2015.02.006
Mrema EJ, Rubino FM, Brambilla G, Moretto A, Tsatsakis AM, Colosio C (2013) Persistent organochlorinated pesticides and mechanisms of their toxicity. Toxicology 307:74–88
Nevalainen T (2014) Animal husbandry and experimental design. ILAR J 55(3):392–398
Olvera-Hernández S, Fernández-Guasti A (2011) Sex differences in the burying behavior test in middle-aged rats: effects of diazepam. Pharmacol Biochem Behav 99(4):532–539. https://doi.org/10.1016/j.pbb.2011.05.030
Olvera-Hernández S, Chavira R, Fernández-Guasti A (2013) Sex- and endocrine-stage-differences in middle-aged rats in an animal model of OCD. Prog Neuro-Psychopharmacol Biol Psychiatry 44:81–87. https://doi.org/10.1016/j.pnpbp.2013.01.021
Ostrea EM Jr, Bielawski DM, Posecion NC Jr, Corrion M, Villanueva-Uy E, Bernardo RC, Jin Y, Janisse JJ, Ager JW (2009) Combined analysis of prenatal (maternal hair and blood) and neonatal (infant hair, cord blood and meconium) matrices to detect fetal exposure to environmentalpesticides. Environ Res 109(1):116–122. https://doi.org/10.1016/j.envres.2008.09.004
Patro N, Shrivastava M, Tripathi S, Patro IK (2009) S100beta upregulation: a possible mechanism of deltamethrin toxicity and motor coordination deficits. Neurotoxicol Teratol 31(3):169–176. https://doi.org/10.1016/j.ntt.2008.12.001
Peleg-Raibstein D, Feldon J (2006) Effects of dorsal and ventral hippocampal NMDA stimulation on nucleus accumbens core and shell dopamine release. Neuropharmacology 51(5):947–957
Rauh VA, Garfinkel R, Perera FP, Andrews HF, Hoepner L, Barr DB, Whitehead R, Tang D, Whyatt RW (2006) Impact of prenatal chlorpyrifos exposure on neurodevelopment in the first 3 years of life among inner-city children. Pediatrics 118(6):e1845–e1859
Ricceri L, Venerosi A, Capone F, Cometa MF, Lorenzini P, Fortuna S, Calamandrei G (2006) Developmental neurotoxicity of organophosphorous pesticides: fetal and neonatal exposure to chlorpyrifos alters sex-specific behaviors at adulthood in mice. Toxicol Sci 93(1):105–113. https://doi.org/10.1093/toxsci/kfl032
Rice D, Barone S Jr (2000) Critical periods of vulnerability for the developing nervous system: evidence from humans and animal models. Environ Health Perspect 108(Suppl 3):511–533 Review
Roberts EM, English PB, Grether JK, Windham GC, Somberg L, Wolff C (2007) Maternal residence near agricultural pesticide applications and autism spectrum disorders among children in the California Central Valley. Environ Health Perspect 115(10):1482–1489
Ruppert PH, Cook LL, Dean KF, Reiter LW (1983) Acute behavioral toxicity of carbaryl and propoxur in adult rats. Pharmacol Biochem Behav 18(4):579–584
Sachana M, Flaskos J, Nikolaidis E, Hargreaves A, Alexaki-Tzivanidou E (2001) Inhibition of rat platelet 5-hydroxytryptamine uptake by chlorpyrifos and carbaryl. Pharmacol Toxicol 9(4):195–200
Sachana M, Flaskos J, Alexaki E, Hargreaves AJ (2003) Inhibition of neurite outgrowth in N2a cells by leptophos and carbaryl: effects on neurofilament heavy chain, GAP-43 and HSP-70. Toxicol in Vitro 17(1):115–120
Shelton JF, Geraghty EM, Tancredi DJ, Delwiche LD, Schmidt RJ, Ritz B, Hansen RL, Hertz-Picciotto I (2014) Neurodevelopmental disorders and prenatal residential proximity to agricultural pesticides: the CHARGE study. Environ Health Perspect. 122(10):1103–1109. https://doi.org/10.1289/ehp.1307044 Erratum in: Environ Health Perspect. 2014 122(10):A266
Singh A, Yadav S, Srivastava V, Kumar R, Singh D, Sethumadhavan R, Parmar D (2013) Imprinting of cerebral and hepatic cytochrome p450s in rat offsprings exposed prenatally to low doses of cypermethrin. Mol Neurobiol 8(1):128–140. https://doi.org/10.1007/s12035-013-8419-5
Terry AV Jr (2012) Functional consequences of repeated organophosphate exposure: potential non-cholinergic mechanisms. Pharmacol Ther 134(3):355–365
Udvardi PT, Föhr KJ, Henes C, Liebau S, Dreyhaupt J, Boeckers TM, Ludolph AG (2013) Atomoxetine affects transcription/translation of the NMDA receptor and the norepinephrine transporter in the rat brain—an in vivo study. Drug Des Devel Ther. 7:1433–1446. https://doi.org/10.2147/DDDT.S50448
Venerosi A, Ricceri L, Scattoni ML, Calamandrei G (2009) Prenatal chlorpyrifos exposure alters motor behavior and ultrasonic vocalization in cd-1 mouse pups. Environ Health 8:12. https://doi.org/10.1186/1476-069X-8-12
Venerosi A, Tait S, Stecca L, Chiarotti F, De Felice A, Cometa MF, Volpe MT, Calamandrei G, Ricceri L (2015) Effects of maternal chlorpyrifos diet on social investigation and brain neuroendocrine markers in the offspring—a mouse study. Environ Health 14:32. https://doi.org/10.1186/s12940-015-0019-6
Xiao J, Kannan G, Jones-Brando L, Brannock C, Krasnova IN, Cadet JL, Pletnikov M, Yolken RH (2012) Sex-specific changes in gene expression and behavior induced by chronic toxoplasma infection in mice. Neuroscience 206:39–48. https://doi.org/10.1016/j.neuroscience.2011.12.051
Funding
This study was funded by the European Commission (FP7-ENV-2011 no. 282957, DENAMIC project), the Ministerio de Ciencia e Innovación (SAF2011-23051), and the Consellería de Educación de la Generalitat Valenciana (PROMETEOII/2014/033).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
“All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.” European Union for the protection of animals used for scientific purposes (Directive 2010/63/EU). “All procedures performed in studies involving animals were in accordance with the ethical standards of the institution (CIPF).”
Rights and permissions
About this article
Cite this article
Gómez-Giménez, B., Felipo, V., Cabrera-Pastor, A. et al. Developmental Exposure to Pesticides Alters Motor Activity and Coordination in Rats: Sex Differences and Underlying Mechanisms. Neurotox Res 33, 247–258 (2018). https://doi.org/10.1007/s12640-017-9823-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-017-9823-9