Abstract
The global spread of enteric disease, the increasingly limited options for antimicrobial treatment and the need for effective eradication programs have resulted in an increased demand for glycoconjugate enteric vaccines, made with carbohydrate-based membrane components of the pathogen, and their precise characterisation. A set of physico-chemical and immunological tests are employed for complete vaccine characterisation and to ensure their consistency, potency, safety and stability, following the relevant World Health Organization and Pharmacopoeia guidelines. Variable requirements for analytical methods are linked to conjugate structure, carrier protein nature and size and O-acetyl content of polysaccharide. We investigated a key stability-indicating method which measures the percent free saccharide of Salmonella enterica subspecies enterica serovar Typhi capsular polysaccharide, by detergent precipitation, depolymerisation and HPAEC-PAD quantitation. Together with modern computational approaches, a more precise design of glycoconjugates is possible, allowing for improvements in solubility, structural conformation and stability, and immunogenicity of antigens, which may be applicable to a broad spectrum of vaccines. More validation experiments are required to establish the most effective and suitable methods for glycoconjugate analysis to bring uniformity to the existing protocols, although the need for product-specific approaches will apply, especially for the more complex vaccines. An overview of current and emerging analytical approaches for the characterisation of vaccines against Salmonella Typhi and Shigella species is described in this paper. This study should aid the development and licensing of new glycoconjugate vaccines aimed at the prevention of enteric diseases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
According to a systematic analysis performed for The Global Burden of Disease Study (GBD) in 2019, the diarrhoeal disease was ranked as the third leading cause of mortality in children and the ninth among all age groups worldwide, contributing to a total of 1.53 million deaths per year (Global Burden of Diseases 2019; Global Burden of Diseases 2019 Risk Factor Collaborators 2020). Bacterial pathogens such as Salmonella subspecies enterica serovar Typhi (S. Typhi), Shigella spp., E. coli and rotavirus account for the largest proportion of deaths from all infectious diseases. The fast emergence of antibiotic resistance observed in bacterial enteric pathogens is of global significance (Humphries and Schuetz 2015; Tribble 2017; Khalil et al. 2018). The Global Antimicrobial Resistance Surveillance System (GLASS) listed these as priority pathogens in 2018 (WHO 2018).
Estimates from The GBD Study suggested that the burden of enteric fevers was around 9.24 million due to S. Typhi in 2019 and 14.3 million from S. Typhi and S. Paratyphi together in 2017. Of 136,000 deaths, the vast majority of deaths (86%) were due to typhoid fever from S. Typhi, of which 67% of deaths occurred in South Asia (Stanaway et al. 2019). Young children and endemic populations for these pathogens in South and Southeast Asia and sub-Saharan Africa are at the most risk. S. Paratyphi A results in the most enteric fever cases in Asia, while bacteraemia in children in Africa is mainly caused by S. Typhimurium and S. Enteritidis (MacLennan et al. 2014). The main risk factors are unsafe water, inadequate hand hygiene and poor sanitation (Global Burden of Diseases 2019; Global Burden of Diseases 2019 Risk Factor Collaborators 2020). High prevalence of the disease in low- and middle-income countries (LMICs), as well as its spread into the wider population highlight the urgent need for effective protection.
In addition to S. Typhi and S. Paratyphi, other Salmonella enterica serovars are responsible for a high burden of disease-causing death and morbidity globally (0.6 to 3.4 million cases annually, especially among children in sub-Saharan Africa (MacLennan and Steele 2019). The most important etiological agents of invasive nontyphoidal Salmonella (iNTS) disease are S. Typhimurium and S. Enteritidis. Nontyphoidal Salmonella are responsible for up to 29% of community-acquired (non-malarial) bloodstream infections in sub-Saharan Africa with an average case fatality rate of 15–20% (Reddy et al. 2010; MacLennan and Steele 2019). Also, for these pathogens, the effectiveness of antibiotic treatment is hampered by the difficulty in making a precise diagnosis, the sudden and rapid onset of the disease, and the growing levels of multidrug resistance.
Shigella is the second leading cause of diarrhoeal disease-related mortality after rotavirus and a primary cause of mortality in individuals older than 5 years old (Troeger et al. 2018). With an annual burden of 165 million cases mostly in developing countries, shigellosis accounted for 213,000 deaths in all age groups worldwide, 30% of which were children < 5 years old (Khalil et al. 2018; WHO 2018). Shigellosis often leads to complications like dysentery that needs antibiotic treatment, which is an emerging challenge due to its resistance. The current WHO recommendations to treat shigellosis include fluoroquinolones as a first line and β-lactams and cephalosporins as the second line with almost no alternative medicines available (Williams and Berkley 2016; Cohen and Muhsen 2019).
Since capsular polysaccharides and serotype-specific O-polysaccharides are tightly linked with the virulence of enteric pathogenic bacteria, global efforts are being taken to develop vaccine candidates using these antigens to provide long-term antibody-mediated protection from enteric diseases (Cohen and Muhsen 2019). Low immunogenicity of many bacterial glycans can be compensated via carrier protein conjugation that helps to induce a T-cell-dependent response (Rappuoli et al. 2018; Micoli et al. 2018a, b, c; MacCalman et al. 2019; Berti and Micoli 2020). Poor immunogenicity and heterogeneity of bacterial polysaccharides make glycoconjugate vaccine development a challenging but important task, as early successes show. Vaccines can become a rapid solution for the control of enteric disease in both LMICs and high-income countries. To reach the people in need, these vaccines must be licensed in accordance with WHO recommendations, national or regional pharmacopoeias and specific approvals issued by the National Regulatory Authorities (NRAs). From the early development stage, the identity, purity, physico-chemistry, stability, composition, size, efficacy and safety profiles of glycoconjugate vaccines must be analysed and meet standards set by the NRA (Ravenscroft et al. 2015b). The global prevalence of these enteric pathogens and the unmet need for effective and accessible vaccines reflect the demand for more rapid and precise characterisation methods that would provide superior quality control in line with existing regulations. This paper reviews current and emerging approaches to vaccine design and analytical methods aiding development and quality control of glycoconjugate vaccines based on Vi capsular polysaccharide of S. Typhi and lipopolysaccharide O-antigen (O-Ag) polysaccharide of Shigella spp.
Part I
S. Typhi and Shigella spp.: strain diversity and pathogenesis
S. Typhi and S. Paratyphi and Shigella spp. are Gram-negative bacteria, which are responsible for typhoid/paratyphoid fevers and shigellosis, respectively (Riddle et al. 2018). At a relatively low dosage, these pathogens also can cause asymptomatic infections, with Salmonella being capable to infect starting at about 105–108 organisms and Shigella spp. with a load of only 10 bacteria (Puzari et al. 2018; Liu et al. 2016). Salmonella enterica serovars Typhi and Paratyphi A (rarely B or C) are the main cause of disease. In contrast, shigellosis can be caused by representatives of the Shigella family, which is subdivided into four groups (Cohen et al. 2019). Within each defined group, multiple serotypes exist (Table 1). Up to 90% of the shigellosis cases are caused by the S. flexneri and S. sonnei serotypes (Page et al. 2016; Kotloff et al. 2018). S. flexneri is mostly endemic in LMICs, while S. sonnei tends to be present in developed countries, where it overrides other serotypes. The enhanced ability of S. sonnei to develop resistance to broad-spectrum antimicrobials makes it an important target pathogen, especially in developed countries, where it has started to become more prevalent. This serotype can accept horizontally transferred DNA and maintain it with better stability than S. flexneri, which results in a growing dominance. DNA transfer results in O-antigen switching, and O-antigens are the main immunogens in vaccines (Anderson et al. 2016; Thompson et al. 2015; Das and Mandal 2019).
Complications of the infection include intestinal immunosuppression that may overpower the established immunity to Shigella giving rise to repeat infections (Brunner et al. 2019). Diversity among pathogenic serotypes, high infectious capacity, variations among clinically relevant strains especially within the S. flexneri family, and disease seasonality and geographic distribution and prevalence complicate the development of an effective vaccine (Muthuirulandi Sethuvel et al. 2016; Knirel et al. 2015; Barel and Mulard 2019; Das and Mandal 2019).
Antibiotic resistance
Recent reports alert on rising extensive drug resistance (XDR) in S. Typhi to common antimicrobial agents, such as co-trimoxazole, ampicillin, chloramphenicol and trimethoprim-sulfamethoxazole, fluoroquinolones and third-generation cephalosporins (Klemm et al. 2018). In endemic populations with high prevalence of XDR Salmonella, not many alternative treatments exist apart from azithromycin and carbapenems (Klemm et al. 2018). But today, bacteria are still capable of developing resistance to azithromycin when used as a second-line agent (Ahsan and Rahman 2019). Emerging advantageous genetic mutations for pathogen survival and ciprofloxacin resistance (recommended as first-line treatment) were recently reported by Pathogenwatch. With increasingly limited treatment options for S. Typhi, control strategies like vaccination and hygiene maintenance must be established (Argimón et al. 2020).
In Shigella, multiple studies have found rising resistance to ciprofloxacin, and quinolones, especially among isolates from children (WHO 2005; Williams and Berkley 2018). Moreover, in 2014, WHO published a detailed report on the geographical burden of fluoroquinolone resistance among Shigella isolates, which showed worrying rates especially for the South-East Asia region which accounts as one of the endemic regions for shigellosis (Table 2) (WHO 2014a). A retrospective review showed that multidrug resistance among Shigella isolates varies from 36–98% (Muthuirulandi Sethuvel et al. 2016). Despite this, some of these antibiotics remain in the official guidelines globally (Table 3). The Infectious Diseases Society of America (IDSA) strongly recommended precise diagnostic tests and strict adherence to public health policies when working with patients suffering from enteric infections in high transmission risk areas and advised cautious prescription of antimicrobial agents (Shane et al. 2017). For example, the WHO recommendations for treatment of dysentery had minimal alterations from 2005 to 2013 and feature ciprofloxacin as the first-line antibiotic and pivmecillinam, ceftriaxone and azithromycin as second-line options. Note that pivmecillinam has no paediatric formulation and azithromycin is not recommended for paediatric treatment, while ceftriaxone is recommended for injection in children and not adults (WHO 2005).
Polysaccharides as target antigens
Salmonella Typhi capsule
Pathogenic bacteria often produce extracellular polysaccharide capsules that serves as virulence factors in promoting attachment and colonisation and in protecting the bacteria from opsonisation, complement binding and opsonophagocytosis, as well as providing hydration and transport functions. The Vi capsular polysaccharide of S. Typhi is immunogenic and is represented by non-stoichiometrically O-acetylated α-1,4-linked N-acetylgalactosaminuronic acid residues and is crucial for host infection, making it a major vaccine target (Liston et al. 2016). The effectiveness of Vi as an immunogen has been an important research topic that led to a promising route to a safer and more protective vaccine design (Robbins and Robbins 1984).
Variability of O-antigen targets in Vi-negative Salmonella and Shigella
Not all enteric Salmonella contain a capsular polysaccharide, leaving the bacterial cell surface-exposed O-Ag as a promising vaccine target. In common with other Gram-negative organisms, the cell envelope of Salmonella and Shigella contains a lipopolysaccharide (LPS) complex. This consists of an outer O-polysaccharide coat, a middle portion (the R core) and an inner hydrophobic lipid A chain. The LPS moiety may function as an endotoxin and O-polysaccharide repeating units confer O-Ag specificity and virulence (Whitfield et al. 2020). For example, rough serotypes of Salmonella and Shigella spp. with incomplete or no surface O-polysaccharide are usually avirulent or attenuated compared to the smooth serotypes which have a complete O-Ag moiety. S. Typhi serovars are positive for LPS O9 and O12 antigens and Vi capsular polysaccharide (Crump et al. 2015). But, S. Typhi may have a Vi-negative serotype, so O-Ag protein conjugation approaches may need to be applied (Salman et al. 2017).
Non-typhoidal Salmonella are the representatives of non-encapsulated enteric pathogens that contain LPS as an outer membrane component. While lipid A is an exceedingly conserved element among Gram-negative bacteria, which is responsible for toxicity, the nature and structures of the repeating glycans in the O-antigen chain are often serotype specific and important for pathogenicity. The R core has a proximal inner region composed of heptose residues, where phosphate, phosphorylethanolamine or pyrophosphorylethanolamine groups are often contained as substitutions; the outer core is usually built of neutral or amino hexoses (Whitfield et al. 2020). In glycoconjugate vaccines against iNTS, the core and O-Ag portions of the LPS are used to induce a protective response. Recently, flagellin proteins native to iNTS serovars were tested in the role of secondary antigens and shown to be a promising approach for the development of a highly potent vaccine (Baliban et al. 2018; Baliban et al. 2020).
Shigella spp. do not possess a capsular component but all contain LPS anchored to the outer membrane via lipid A. Antigenic O-polysaccharide chains attached at the distal end of the LPS core define the serotype affiliation and strain-dependent immune specificity of Shigella. The length and structural availability of this O-Ag play a key role in pathogenesis and host resistance (Lindberg et al. 1991; Caboni et al. 2015). The O-Ag is unique for each serotype, reflecting the genetic diversity of Shigella spp. (Perepelov et al. 2012).
Part II
Vaccines on the market and in the pipeline
In the 1960s, the first live-attenuated S. Typhi vaccine was created and gave up to 7 years of protection (Ashcroft et al. 1967). Nevertheless, systemic adverse events following the use of such vaccines excluded them from modern treatment approaches (Ashcroft et al. 1964). Today, efforts are taken in the direction of native and synthetic polysaccharide formulations, protein-conjugated antigens, combination vaccines and alternative delivery systems like GMMA (generalised modules for membrane antigens) (Rossi et al. 2014). Four Vi conjugate vaccines have been licensed with one so far receiving WHO prequalification. Vi-DT (diphtheria toxoid) and others are in late-stage development or waiting for approval. There are several Vi PS based and a number of bivalent conjugate vaccines against Typhi and Paratyphi in development. Examples can be seen in Table 4.
Salmonella Typhimurium and S. Enteritidis bivalent vaccine could represent a valuable public health intervention. Two approaches, both O-Ag based, have been evaluated. The first approach is based on glycoconjugation, where S. Typhimurium and S. Enteritidis O-Ag are independently linked to CRM197 or a homologous flagellin protein as carriers. The second approach is represented by the development of a bivalent formulation of S. Typhimurium and S. Enteritidis genetically modified outer membrane vesicles (OMV) termed “GMMA” (Micoli et al. 2018a, b, c). Several glycoconjugates are now in preclinical development for iNTS, and some employ flagellin from S. Enteritidis to function as both a carrier protein for bacterial O-Ag and a secondary antigen in an attempt to increase the efficacy. This preparation is scalable and allows bivalent/multivalent formulations for different O-PS antigens. This conjugation strategy may contribute to an enhanced immunogenicity and safety of this potential vaccine candidate and is likely to come at a lower production cost, which is important for implementation in LMICs (MacLennan and Steele 2019; Baliban et al. 2020).
With over 50 serotypes of Shigella arising from O-Ag variations, the development of an appropriate target for a vaccine is challenging. Antigen variability complicates the protection of the population in non-endemic areas. Thus, a vaccine should preferably be multivalent covering the most pathogenic Shigella species such as S. flexneri 1b, 2a, 3a and 6, and S. sonnei according to the WHO, and a recent meta-study on antibiotic resistance (Mani et al. 2016; Das and Mandal 2019; Micoli et al. 2018a, b, c; Raso et al. 2020; WHO 2018, 2020a). The global enteric multi-centre study suggests that such a multivalent formulation would cover 72% of Shigella strains protecting directly and cross-protect for up to 89% of all strains (Livio et al. 2014). Shigella dysenteriae type 1 used to be linked to rapid spread and high mortality rates; however, no epidemic cases were reported in the last 20 years and it is rarely included in serotype-specific vaccines (Kotloff et al. 2018). Several vaccines have now been developed with a few entering phase III clinical trials: most candidates are based on S. flexneri 2a LPS conjugates (Table 5). Although Shigella vaccines lack WHO guidelines for vaccine development, a recent draft version suggests that quality and safety must be monitored at least to the standard of currently licensed glycoconjugate vaccines (WHO 2020a, b). New standards for Shigella are also going to be established soon to aid the development of a vaccine evaluation protocol (NIBSC 2020).
Factors that play a role in immunogenicity for Vi and Shigella O-Ag glycoconjugates
Antigen chemistry and O-acetyl content and modifications
The structural implications of glycan antigens to become an efficient immunogen are still unclear. Apart from carbohydrate chain length, other important viability properties affect the resultant antigen success: the stereochemistry of glycosidic linkages, glycan position within a repeating unit, locations of branching points and biochemical composition, which complicate design and analysis. Size/span of the epitope, terminal glycan residues, presence/absence of branching points, substituent groups, such as O-acetyl, and number of repeating units may be considered as key parameters to measure (Anish et al. 2014; Berti et al. 2018). Table 6 lists the repeating unit structures of S. Typhi capsular polysaccharide and the O-antigens of vaccine-relevant Salmonella and Shigella species.
For Vi PS, O-acetylation is a major factor for immune potency (Szu et al. 1991; Hitri et al. 2019). Vi PS is variably O-acetylated at the carbon 3 of the repeating monosaccharide. The O-acetyl groups are the most solvent exposed (Szu et al. 1991; Hitri et al. 2019) and the primary epitope exposed for antibody binding since carboxyl and N-acetyl groups are buried within the PS helix, making them less available as antibody targets, which are only be exposed in de-O-acetylated Vi (Hitri et al. 2019). Antibodies reacting with carboxyl and/or N-acetyl groups in de-O-acetylated Vi have been reported, but these groups are unlikely to be dominant in Vi vaccines, which meet the recommended level of O-acetylation (Qadri et al. 1990; Szu et al. 1991). The relatively high viscosity of Vi makes it more challenging to characterise with physicochemical methods like HPLC-SEC or matrix-based sample separation methods, for example ultrafiltration membranes or solid-phase extraction cartridges in a free polysaccharide assay (Hitri et al. 2019). Partially, O-acetylated PS may be structurally beneficial in a formulation to give access to additional epitope(s) (Hitri et al. 2019). Nevertheless, the key to an effective Vi vaccine is to ensure that the O-acetyl content will be at least 52% or 2.0 mmol/g of PS as specified by WHO recommendations (Szu et al. 1991; Lemercinier et al. 2000; WHO 2014b).
With iNTS and Shigella, the level and position of O-acetylation of the O-Ag and any chemical modifications of the LPS in the infectious strains mediate the strength of the immune response and are considered to be important variables for vaccine safety and efficacy (Kubler-Kielb et al. 2007; Giardina et al. 2005). The O-polysaccharide portion of the LPS is the most exposed and is extremely diverse in chain length, chain distribution and composition, which all affect virulence (Carter et al. 2007; Morona et al. 2003; West 2005). The O-Ag can be composed of multiple repeating units (RUs), which contain two to six monosaccharides per RU and are heterogeneously distributed on the LPS molecule. The chain numbers and lengths are serotype dependent (Barel and Mulard 2019). Techniques currently used for characterisation of LPS and O-Ag in vaccine preparations are 1D and 2D NMR, GC, GC-MS and HPAEC-PAD (Micoli et al. 2014; Gerke et al. 2015; De Benedetto et al. 2017a; Raso et al. 2020).
Carrier proteins and conjugation chemistry
To confer immunogenicity, the saccharide component is chemically or biologically conjugated to a carrier protein. Currently licensed carriers of Vi PS include tetanus toxoid (TT), diphtheria toxoid (DT) and cross reacting material 197 (CRM197), while other glycoconjugates utilise Haemophilus protein D and outer membrane protein complex from serogroup B meningococcus and pneumococcal proteins. The recombinant non-toxic form of Pseudomonas aeruginosa exotoxin A (rEPA) has been evaluated as a carrier protein with Shigella antigens and Vi PS (Cohen et al. 1997; Kubler-Kielb et al. 2007; Thiem et al. 2011; Kämpf et al. 2015). Most emerging protein carriers are recombinant, and alternatives such as OMV, GMMA, inorganic nanoparticles and virus-like particles are being investigated. The GMMA approach may be promising for vaccine development in LMICs if it is cheaper to produce and its efficacy meets accepted standards. More methods like one-pot (single reaction mixture) and chemo-enzymatic protocols, along with HPLC-based automated oligosaccharide construction methods and advances in the assembly of synthetic glycan and alternative nanocarriers that are being developed will speed up vaccine development (Safari et al. 2012; Micoli et al. 2018a, b, c; Wen et al. 2018). Such new protocols will also require characterisation, consistency of production and verification of their quality and safety by currently recommended assays.
Glycoconjugate vaccines are produced by either chemical, bioconjugation or GMMA approach, conferring variations in conjugation chemistries that affect the ability to properly define and standardise a molecule. For example, chemical conjugation protocols expose or modify each oligo- or poly-saccharide separately; a similar situation is seen in “sun-type” bioconjugate vaccines with a single point of conjugation. “Lattice-type” conjugates where carrier protein and O-antigens interlink several times may be more challenging to analyse (Barel and Mulard 2019). The integrity of protective saccharide epitopes should be verified following modifications.
Immune responses to vaccines can also be affected by complexity of assembly, linker structure and carrier attachment site number or the structure, conformation and size distribution of the conjugates (Avci et al. 2019). In constructing Vi conjugates, Arcuri et al. discovered that actual conjugation chemistry in terms of linker number conjugated to the carrier protein (in this case CRM197) did not impact vaccine immunogenicity as well as did not inter-relate to linker length or the saccharide loading on the carrier protein (2017). While the highest IgG response post-primary dose was achieved with a full-length Vi PS-conjugate versus fragmented, the requirements for an optimal boost varied. For a full-sized antigen, the response was similar irrespective of protein conjugate, but in formulation with fragmented Vi, after second immunisation a booster response was only observed with TT but not with CRM197 and DT that may be linked to the larger size of TT as well as its T-cell epitopes (Arcuri et al. 2017; Wessels et al. 1998; Lockyer et al. 2020). With a CRM197 conjugate, Vi PS of less than 50 kDa gave a booster dose, while full-length Vi PS led to hypo-responsiveness (Micoli et al. 2020a, b). Although Vi-CRM197 and Vi-rEPA behaved differently when tested in infants, the source and size of Vi PS were different, and these factors should also be considered (Thiem et al. 2011; Bhutta et al. 2014; Micoli et al. 2020a, b). Another study has also confirmed that immune response to Vi-DT in mice was stronger with an increasing amount of cross-linking and the subsequent size of the conjugate (An et al. 2011). Recently, Sun et al. showed that while conjugation chemistry was not critical for immune response activation, polysaccharide stability and structure dictate the unique antigen presentation method to the T-helper cells, elucidating adaptive immune response (Sun et al. 2018). Moreover, for Vi-CRM197, studies in mice indicated that shorter polysaccharide have less risk of inducing unfavourable T-independent immune response and hypo-responsiveness compared to long-chain Vi conjugates. Therefore, the elimination of T-independent responses elucidated by potential glycan candidates may be the next important control used for rational vaccine design and testing (Micoli et al. 2020a, b). For Shigella, use of a succinylated carrier protein was found to produce more immunogenic glycoconjugates compared to the unaltered version so chemical modifications of carrier proteins are also important to consider and need to be analysed (Barel and Mulard 2019). These findings highlight that in the development of novel Vi PS vaccines, the PS chain length and the carrier protein of candidate vaccines may need careful monitoring to determine their optimal immunogenicity.
International guidelines of vaccine development and control: overview
In 2014, WHO developed Guidelines for the evaluation and lot release of typhoid glycoconjugate vaccines, and these were replaced in 2020 by more comprehensive WHO recommendations (WHO 2014a, b, 2020b). Physicochemical tests can ensure batch consistency of conjugate vaccines and were exemplified by the WHO and Pharmacopoeia guidelines based on predecessor conjugate and unconjugated polysaccharide vaccines. Molecular size distribution, the saccharide and protein quantity and their ratios must be considered when determining the structure of the conjugate with appropriate analytical methods. The key testing areas required for vaccine quality control are described below, including the techniques used to measure the relevant structures (Table 7).
Identity and compositional analysis of polysaccharide-based vaccines
The identity of a glycoconjugate vaccine for enteric pathogens can be defined in monosaccharide composition, O-acetyl group quantitation and purity of the sample. 1H-nuclear magnetic resonance is validated as a sole routine release method for PS identity and purity, which is recommended by the United States Pharmacopeia (USP) and WHO for S. Typhi Vi PS and is a superior technology to less precise colorimetric techniques (although it cannot measure residual nucleic acid or protein) (Lemercinier et al. 2000; WHO 2017b; United States Pharmacopeial Convention 2018). NMR provides data on the PS structure, stability and degradation pathways, and can be used with a quantitative standard as a qNMR method to get relative molecular mass and content (WHO 2017a). Uni-dimensional and two-dimensional NMR spectroscopy can be used for S. Typhi and Shigella O-Ag in either glycoconjugate or GMMA preparations, and is a relatively rapid and information-rich characterisation method (Berti and Ravenscroft 2015; Raso et al. 2020). NMR spectroscopy has also proven valuable for the detection, identification and quantification of process impurities in the polysaccharides (Xu et al. 2005; Beri et al. 2019). Nevertheless, spectra are less resolved for the hydrophobic and rigid structure of tightly packed substituents in the sugar ring of Vi due to the rapid spin-spin relaxation rates, which is not ideal for differentially O-acetylated serotypes (Lemercinier et al. 2000).
Other methods for Vi polysaccharide quantitation include an acridine orange dye colorimetric method (Stone and Szu 1988), rate nephelometry and rocket immunoelectrophoresis (WHO 2017b). High-performance anion-exchange chromatography-pulsed amperometric detection (HPAEC-PAD) is a common technique applied to quantitate Vi saccharide content in drug substance and drug product using an alkaline hydrolysis method (Micoli et al. 2011). Measuring the Vi content of conjugated or non-conjugated polysaccharide vaccines by six laboratories with HPAEC-PAD resulted in good inter-laboratory comparability and produced improved results distribution of Vi content when the homologous Vi PS standard was used to quantitate the Vi content in a vaccine from the same source (Citrobacter freundii or S. Typhi). The rocket immuno-electrophoresis was performed by four laboratories but could have limitations when used for bulk conjugates, due to the standard-sample molecular mass disagreement. It, however, gave good results for the PS alone (WHO 2017b; Gao et al. 2019).
LPS and glycan composition in Shigella can be determined with the methods described above as well. The 1H diffusion-ordered spectroscopy (DOSY) NMR coupled with HPLC-SEC (size exclusion chromatography) with differential refractive index (dRI) can be used to detect LPS chain length, size and structure variations in S. flexneri GMMAs. The number of LPS core reducing end KDOs (2-keto-3-deoxy-octonate) is isolated and quantified by HPLC-SEC as well. The percentage of O-Ag can be determined as the molar ratio of their KDO to total KDO. O-Ag sugar content is also possible to quantify using the Dische colorimetric method (methyl pentoses (6-deoxyhexoses)) (Dische and Shettles 1948). Colorimetry is less precise so physical or immuno-detection methods may be prioritised. Total O-acetyl content, as well as O-acetyl content variations, can be confirmed with 2D NMR (Raso et al. 2020). NMR and HPLC-SEC are often applied to check glycoconjugate purity as well (Berti and Ravenscroft 2015; Ravenscroft et al. 2015b). HPAEC-PAD run with appropriate standard or standard mixtures can be applied for GMMA or complex O-Ag saccharide chains to calculate the relative molar concentration of glucans (Gerke et al. 2015).
The number of polysaccharide chains bound to the carrier protein can be deduced with limited information if chain lengths are known; however, these methods do not give information on their position. The conjugation positions can be identified by mass spectrometry, but it is still a challenge to map conjugation sites in populations of polydisperse molecules. Some reports on capsular polysaccharides (CPS) show that solid-state NMR spectroscopy is a working alternative to determine PS/protein conjugation pattern and the degree of conjugation; however, it has not yet been reported for Salmonella Typhi, iNTS or Shigella conjugated vaccines (Giuntini et al. 2017). To determine sugar:protein ratio, colorimetric tests are still popular due to their low cost and relative simplicity, for example anthrone and a colorimetric protein concentration assay (Leyva et al. 2008; Ravenscroft et al. 2015b).
Standardised ELISA methods are available for immunogenicity determination. Currently, two methods have been accredited as repeatable and reliable for serological ELISAs of serum responses against Vi-based conjugates from clinical studies: commercial VaccZyme and in-house Vi-poly-l-lysine (PLL)-based immunoassays, where Vi is pre-coated with PLL to give superior results to native coating, as PS is negatively charged allowing PLL to adhere and improve Vi interaction with solid phase (Rijpkema et al. 2018; Rigsby et al. 2020). The biotinylated Vi ELISA can be used for control comparison of potencies but not for the geometric mean potencies, or potency estimates, as it gives alternative reads to the VaccZyme ELISA (Rijpkema et al. 2018). Following the request of the WHO Expert Committee on Biological Standardization (ECBS), Vi-PLL ELISA has been finally assessed in 2020 as a suitable substitute to the VaccZyme ELISA, allowing to use this uniform non-commercial, and thus more available, method to use at a lower cost. The Vi-PLL procedure is publicly available and uses standard biological agents, as opposed to VaccZyme (WHO 2018; Rigsby et al. 2020). Key considerations for implementation and smooth performance are the level of reagent standardisation (reference standard), validity criteria (system and sample suitability) and the inclusion of assay-specific run controls which may be study samples representative of Vi PS vaccines (Rigsby et al. 2020).
Determining O-acetyl content
The most basic, cheap and straightforward method for O-acetyl content estimation is the colorimetric Hestrin assay (Hestrin 1949) which is routinely used with purified glycoconjugate vaccines or polysaccharide components but gives less precise information (Gao et al. 2019). Another potential approach to determine the degree of O-acetylation could be an enzyme immunoassay using an O-acetyl-specific monoclonal antibody. However, due to the importance of the O-acetyl pattern for vaccine immunogenicity, complementary methods to colorimetry and immunoassays were developed, enabling acetyl group detection, content and positioning on the purified CPSs. These are currently NMR, and HPAEC with conductivity detection (HPAEC-CD), although this is not yet widely used (Lemercinier et al. 2000; Kao and Tsai 2004; Hitri et al. 2019). NMR is considered to give more information on O-acetyl position in general and has benefits over wet chemical methods since it can be used for CPS fingerprinting and gives a wider spectrum of information; however, it becomes difficult to interpret when more complex cross-linked conjugates are evaluated. Nevertheless, 1H and 13C NMR coupled with mass spectrometry was applied successfully to detect O- and N-linkages in Vi PS in a GMMA construct (2020). HPAEC-CD is up to 20 times more sensitive compared with the Hestrin test, requires a smaller amount of material compared to NMR and is a superior method to evaluate monosaccharide composition (Mulard 2017).
While circular dichroism (CD) is commonly applied for the characterisation of carrier protein integrity, effect of conjugation and stability determination, a recent study has shown its potential for monitoring O-acetylation (Jones et al. 2020). A strong signal from the Vi, unusual to polysaccharides, is observed in the far-UV from O-acetylated and non-O-acetylated residues in the Vi. With a larger amount of information generated by CD for Vi compared to other conjugates, it may become a useful method to monitor vaccine stability by analysing the degradation or de-O-acetylation behaviour of the polysaccharide. This method poses larger uncertainty when reading weak spectra. To assess the suitability of CD for this application, more detailed data comparisons from orthogonal methods and corrections for specific saccharides are required (Jones et al. 2020).
Detection of polysaccharide degradation or detachment in vaccine products
Free, unconjugated PS content is the main indicator of vaccine stability and inversely correlates with its potency, or effective dose; a product with increased free saccharide content could be less immunogenic, due to a lower amount of conjugated saccharide, and the potential for unconjugated saccharide to neutralise pre-existing antibody. It can be quantified successfully by only a limited number of methods that allow reproducibility. This measurement is reflecting the stability of the glycoconjugates. To determine free PS content, two steps are required: free PS separation and its quantification. The first step can routinely be done by chromatography (size, ion-exchange, hydrophobic interaction), chemical precipitation with acid or detergents, ammonium sulphate or ammonium adsorption, capillary electrophoresis, gel filtration, centrifugal ultrafiltration, ultracentrifugation, solid-phase extraction or immunoprecipitation. The second step can be performed using colorimetry or immunoblotting or by using physicochemical techniques (HPLC, HPAEC-PAD or gas chromatography (GC) post-hydrolysis), which are more robust approaches (Ravenscroft et al. 2015b).
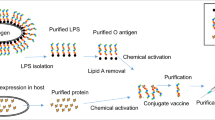
Giannelli et al. found that UV-coupled HPLC-SEC detection of free Vi PS proved to be problematic due to the variable extinction coefficient of Vi that depends on O-acetyl content and chemical modifications required for carrier attachment (2017). An alternative free PS purification method was introduced using the Capto Adhere resin separation that worked irrespective of conjugated protein and delivered a more sensitive and quantitative measurement for a range of Vi lengths. It works by entrapment of intact conjugates and HPAEC-PAD quantification of the Vi component recovered in the filtrate. Capto Adhere resin separation coupled with HPAEC-PAD achieved 75–120% recovery of free Vi PS. The absence of conjugates in the buffer strip solutions was verified by a dot-blot assay and HPLC-SEC chromatograms (Giannelli et al. 2017). Their most recent paper on Vi detection suggests an optimised hydrolysis conditions for the HPAEC-PAD detection method to get more accurate data on the de-O- and de-N-acetylated Vi PS monomer, discussed later in the review. A deoxycholate (DOC) precipitation method (Lei et al. 2000), in which free saccharide is recovered in the supernatant of DOC- precipitated protein samples, has also been shown to measure reproducible % free saccharide levels (Fig. 1).
Another one-step immunoprecipitation method based on anti-carrier protein antibody-coupled Sepharose beads was developed at the Serum Institute of India. The method claims to overcome HP-SEC limitations and can be applied for free PS from multivalent polysaccharide-protein conjugate vaccine formulations, total PS content and percent adsorption of polysaccharides (Serum Institute of India 2013).
Molecular size profiling and solution properties of glycoconjugate vaccines
Glycoconjugates manufactured by conjugating PS to a carrier protein will have increased mass, altered shape and possibly changes in behaviour in solution compared to their individual components. Flexible interactions in solution and inter-molecular interactions can be influenced by the character of the polysaccharide as well as the protein, and changes to protein sites required for antigen processing and T-cell epitope accessibilities could be affected. The type of carrier protein that is conjugated may influence the choice of analytical techniques. While monovalent “sun type”, for example CRM197 conjugates may be more straightforward to characterise, TT-based cross-conjugated glyco-vaccines (e.g. “lattice type”) may be more challenging (Barel and Mulard 2019). Proven methods to measure molecular size distribution, hydrodynamic size, viscosity and solution behaviour (of the glycoconjugate or the separate polysaccharides and/or carrier proteins) include SEC-MALS/UV/RI/Viscosity, degree of polymerisation (repeating unit analysis), analytical ultracentrifugation and optical spectroscopy (MacCalman et al. 2019).
When determining molecular integrity of polysaccharides in solution, challenges linked to the polydisperse nature of the saccharides arise, in addition to their higher non-ideality (through co-exclusion and polyelectrolyte effects) compared to proteins. SEC-MALS is considered a useful technique with minimal non-ideality effect due to the dilution from the columns. However, it relies on interaction with a matrix, is dependent on % recovery and sometimes ignores hydrodynamic changes brought about by the PS moiety detachment or protein unfolding which increases the apparent relative size, as if the conjugate structures were intact (Ravenscroft et al. 2015b). In addition, the separation columns have a size separation limit, and the assumption must be made; there are no anomalous interactions with the polymer matrix which makes up the separation columns. SEC-MALS/RI/viscosity of Vi polysaccharides in different eluents demonstrated the need to consider column choice and buffers salts to obtain representative recoveries and highlighted the differences that salt composition makes to hydrodynamic size and viscosity (Hitri et al. 2019). See also the following examples of other vaccine-relevant oligo- and poly-saccharide components (Jumel et al. 2002; Bardotti et al. 2008; Harding et al. 2012; Lockyer et al. 2015).
Analytical ultracentrifugation (AUC)—a matrix-free method—has also been extensively applied for molecular distribution and molecular weight of polysaccharides (see, for example Harding et al. 2010 and 2015 & references cited therein). The two main types of AUC are sedimentation velocity and sedimentation equilibrium. Sedimentation velocity provides separation based on size and shape, and analysis, normally from Rayleigh interference optical records. Sedimentation coefficient distributions provide a measure of heterogeneity, and if conformational information is known, the distributions can be converted into molecular weight distributions using the “Extended Fujita” method. Sedimentation equilibrium—at slower speeds than for sedimentation velocity—gives optical records that are directly related to molecular weight. Some caution is necessary with regard to non-ideality, either by working at low concentration to minimise these effects (the lowest working concentrations are ~ 0.05 mg/mL for sedimentation velocity and ~ 0.4 mg/mL for sedimentation equilibrium) or by making measurements at different concentrations and performing an appropriate extrapolation to zero concentration where non-ideality effects vanish. Conformation and flexibility can be estimated using power-law coefficients (relation between sedimentation coefficient and molecular weight) and the Wales-van Holde ratio (ratio of the “non-ideality” concentration dependence sedimentation coefficient parameter to the intrinsic viscosity) and using the persistence length calculations based on “HYDFIT” combinations of SEC-MALS, viscosity and AUC data (see Abdelhameed et al. 2012, 2016a, b) for examples with Hib and meningococcal-TT conjugates.
Optimal spectroscopic methods such as near-UV CD and Trp fluorescence spectroscopy also can report on the mobility and solvent accessibility of aromatic amino acid side chains. The effects of conjugation on these groups, as well as the secondary structure and folding of a carrier protein, as determined by far-UV CD, are important characterisation methods and will remain important for the initial characterisation of novel recombinant carrier proteins. Carrier protein and conjugate molecule characterisation and assessment of thermal stability have been described for these carrier proteins: CRM197 (Crane and Bolgiano 1997; Bolgiano et al. 2001; Ho et al. 2001; Bardotti et al. 2008), TT (Ho et al. 2002; Lockyer et al. 2015), rEPA (Ho et al. 2006), DT and protein D (Lockyer et al. 2015). Structural effects on conjugated carrier proteins have been demonstrated to correlate with reduction in the immune response against the carrier (Beresford et al. 2017).
Novel Shigella vaccines: glycoconjugates and GMMA
Several vaccines in clinical trials are currently targeting the most common serotypes of Shigella (Table 5); however, a more promising direction to take maybe towards multiprotection formulations (WHO 2020a). The WHO suggested existing recommendations for glycoconjugate vaccines control to be followed in product guidance for Shigella vaccines (WHO 2020a). Since Shigella vaccines are based on the O-polysaccharides anchored to the LPS, they are more challenging to produce and control. Conjugation chemistry of multiple antigens is structurally complex, and LPS requires laborious extraction and detoxification, which may affect molecular integrity. LPS and lipoproteins in GMMA act as immunostimulatory components, which alter their reactogenicity: the level of LPS acylation is important for determining the potency and safety of vaccines (De Benedetto et al. 2017a).
Alternatives to multistep chemical conjugation include bioconjugation using transformed E. coli, GMMA vesicles and synthetic conjugates (Kis et al. 2018). GMMA vaccines are already manufactured in GMP-quality conditions against S. sonnei, S. flexneri and nontyphoidal Salmonella strains (Gerke et al. 2015). Novel GMMA formulations are usually characterised by conventional analytical methods used for other glycoconjugates, being verified for their use (Micoli et al. 2020a, b; Raso et al. 2020). For GMMA, O-Ag core sugar content can be quantified by HPAEC-PAD, after performing acid hydrolysis directly on GMMA. After O-Ag extraction, total sugar content can be done using a suitable colorimetric method, or NMR, which also provides structural information and assesses purity (Raso et al. 2020). The concentration of O-Ag can be determined with a combination of NMR and HPAEC-PAD (Gerke et al. 2015). Protein and lipid content will need to be determined to calculate LPS/protein ratios.
The GMMA size distribution can be determined by dynamic light scattering (DLS). Other methods described for GMMA particle size include multiangle light scattering (MALS) that can be coupled to the high-performance liquid chromatography–size exclusion chromatography (HPLC–SEC/MALS, which also helps to check sample purity) and nanoparticle tracking analysis (NTA) (De Benedetto et al. 2017b).
Meloni et al. used hydrolysed GMMA (1% acetic acid v/v, neutralised with 28% NH4OH) from Salmonella Typhimurium SL1344 ΔtolR O-antigen (O-Ag) PS purified by a tangential flow filtration method to separate and analyse O-Ag, and it was revealed that such method strips off the low molecular mass O-Ag completely in GMMA and wild-type bacteria, shown by HPLC analysis of the batches (2015). Saccharide content was then analysed by HPAEC-PAD, confirming the differential monosaccharide ratios of low and medium molecular mass antigens in GMMA and wild-type bacteria. This O-Ag has more similarity to an LPS, which may be relevant for Shigella GMMA characterisation. This method highlights that O-Ag purification must be optimal to fully characterise GMMA constituents; however, the approach may be useful to evaluate the proportion of larger Mw components with HPAEC-PAD, and seems to work well as a direct O-Ag separation method in enteric Salmonella (Micoli et al. 2013).
Ravenscroft et al. described analytical techniques used to prove the structural integrity of S. dysenteriae type 1-EPA glycoconjugate produced by innovative biosynthetic Escherichia coli glycosylation that are suitable for characterisation of other Shigella serotypes and based on conventional approaches like (RP/NP)HPLC-SEC/UV, immunoblot detection, 1D and 2D NMR and MALDI MS/MS post-trifluoroacetic acid (TFA) hydrazinolysis (2016). The recovery of the Rha repeating unit was low as the free 6-deoxy hexose decomposed due to hydrolysis employed to release GlcNAc, suggesting the importance of optimal hydrolysis conditions. The S. dysenteriae type 1 O-PS structure was fully characterised by 1D and 2D NMR spectroscopy with full information provided on the Gal, two Rha and GlcNacspin systems evaluated by the use of 1D TOCSY, 2D 1H–1H (COSY and TOCSY) and 1H–13C (HSQC, HMBC and HSQC-TOCSY) experiments (Ravenscroft et al. 2016).
Part III
Emerging techniques to aid glycoconjugate design
Structural vaccinology
Structurally complex polysaccharides conjugated to carrier proteins may pose a risk of exerting immunodominance over a less immunogenic but essential epitopes in a combination vaccine, which is highly undesirable for immunisation. Neoepitopes that form due to degradation or modification of polysaccharides may adversely impact the immunogenicity of native polysaccharides. Getting access to comprehensive structural evaluation of the mechanisms of binding and epitope characterisation tools may help to expand current understanding of how immunodominance is dictated in glycoconjugates and even provide novel data allowing for the enhancement of the antigen structure to achieve adequate and balanced protection (Anish et al. 2014).
Computational methods to facilitate synthetic antigen design
Establishing and evaluating carbohydrate structure-function relationships that affect immunogenicity are topics of high interest. Glycan shape complexity, chain flexibility, dynamic properties of pyranose rings and diversity of functional groups make structural analysis a priority that would benefit from multiple approaches. Where 3D physicochemical methods are not informative enough, computational methods may supplement the characterisation of structural determinants of affinity, specificity and antigenicity. In silico simulations could allow for a study of conjugation and binding at an atomic level when they cannot be experimentally deduced. Computational methods allow for tracking molecular effects on saccharide antigenicity (through enthalpy and entropy changes), which is important for the selection of the right candidate for synthesis. Moreover, currently used methods, such as X-ray diffraction or NMR spectroscopy, may provide biased results for large saccharides in more dilute solutions. While NMR techniques are relevant for homogeneous saccharide characterisation, implications linked to molecular mass limit the scope of applications for large antibody-PS complexes and often need coupling with in silico molecular dynamics (MD) simulations for structural definition (Anish et al. 2014). MD simulations of vaccine-relevant PS that consider chain length and counterions in a hydrated environment have identified potential epitopes and conformer distributions for Vi PS and O-Ag (Hitri et al. 2019; Hlozek et al. 2020).
Characterisation and structural information for glycoconjugates cannot be achieved by a single method; thus, multiple biophysical techniques combined with computational methods allow deduction of carbohydrate structure-function analysis and antibody interaction (Anish et al. 2014). Molecular modelling coupled with NMR spectra of the molecular structure has been implemented for more optimal design and immunogenicity of Shigella SD1 synthetic glycoconjugates. It allowed identification of a preferred hairpin conformation of a synthetic O-Ag (the ABCD tetrasaccharide) that was representing the native O-Ag more closely. A hairpin conformation for ABCDA’ saccharide and larger O-Ag parts in a helix conformation allowed partial exposure of B and D monosaccharides, thereby inducing antibody recognition that could lead to an enhanced anti-O-Ag antibody response, which is a key parameter for vaccine potency and safety considerations (Barel and Mulard 2019). These methods provide alternatives to the 3D structural analysis of polysaccharides by small-angle X-ray/neutron scattering, X-ray crystallography and NMR (Anish et al. 2014).
Current efforts for better polysaccharide characterisation
Modified methodologies of HPAEC-PAD for more precise Vi saccharide quantitation
HPAEC-PAD is currently the most robust and detailed quantification technique used for PS compositional analysis of bacterial vaccines (Rohrer 2020). However, due to the low susceptibility of Vi and other uronic acids to PS depolymerisation, this method is constantly being optimised for more accurate and precise readings. While strong alkaline-based depolymerisation is efficient for Vi in monovalent formulations, combinations with S. sonnei O-antigen, where co-elution with Vi may occur, are likely coming from the degradation of the alturonic acid (Gerke et al. 2015; Giannelli et al. 2020). Giannelli et al. further improved the hydrolysis step for HPAEC-PAD by suggesting acid hydrolysis with simultaneous use of trifluoroacetic and hydrochloric acid (TFA:HCl in a 2:13 v/v ratio), to prevent such co-elution and verified product formation using 1H NMR, 13C NMR, COSY and HSQC (Giannelli et al. 2020). The presence of 2-amino-galacturonic acid monosaccharide in equilibrium between α and β conformations was also observed for Vi. Recovery of 101% was reported of reduced (de-O- and de-N acetylated) monosaccharides with increased sensitivity due to the high yield of hydrolysis product(s) compared to the depolymerisation method. This procedure could facilitate the development of standards in enteric vaccines containing both Vi PS and O-Antigens that also contain amino uronic acids (Giannelli et al. 2020).
We have compared the novel HCl:TFA hydrolysis method for Vi PS quantitation of a typhoid conjugate vaccine containing S. Typhi Vi PS, with the standard NaOH hydrolysis conditions (Gao et al. 2019) using WHO International Standards for the polysaccharides (Fig. 1).
The content of Vi saccharide in a typhoid conjugate vaccine was quantified using WHO International Standards for Vi PS (Gao et al. 2019). Standards and vaccine samples were base depolymerized with 2 M sodium hydroxide for 4 h at 110 °C (panels a to c) or acid-hydrolysed with 10% TFA-8 M HCl for 4.5 h at 80 °C (panel d), following the recovery of unconjugated PS in the supernatant of a vaccine sample precipitated by sodium deoxycholate (1% w/v) pH 8.0 and HCl (0.04 M) (Lei et al. 2000). The separation was performed on a CarboPac PA-1 column (2 mm) at 25 °C with an Amino Trap as a guard column. Base-treated samples were eluted at a flow rate of 0.25 mL/min for 30 min, and with elution conditions of 0–2 min, 100 mM NaOH, 40 mM NaNO3; 2–22, 100 mM NaOH, 40–150 mM NaNO3 (Gao et al. 2019); acid-hydrolysed Vi was run at 0.375 mL/min using isocratic elution with 400 mM NaOH for 15 min. N-Acetyl glucosamine-6-phosphate (10 μg/mL) (peak 1) was injected in samples and standards to normalise the Vi signal in base-treated samples. Standard curves were constructed using 27, 9, 3, 1 and 0.5 μg/mL Vi PS shown in panels a and b as alternating solid and dotted lines (peak 2) from C. freundii (panel a) or S. Typhi (panel b) polysaccharide samples. The vaccine sample (panel c) injected was 10 μg/mL Vi PS to quantify free, unconjugated (dotted line) or total Vi PS (solid line). Panel D of acid-hydrolysed standards and sample shows from top to bottom, 27 (μg/mL) C. freundii Vi, 27 S. Typhi Vi, 9 C. freundii Vi, 9 S. Typhi Vi and total saccharide vaccine sample. Samples were injected in duplicate. For base-treated samples, Vi eluted within the expected time interval, at about 14.3 min, and both standards yielded R2 > 0.98. For acid-hydrolysed samples, Vi eluted at about 5.4 min and the signal was 6 times higher.
Both the Vi PS concentration (μg per dose) and % free Vi PS were lower using the C. freundii sample by 7.7% and 4.3%, respectively, compared to native Vi PS standard under base treatment. This suggested that C. freundii is an imperfect alternative to the native Vi that could underestimate Vi content in the sample. Homologous source Vi PS should be used for quantitation (Gao et al. 2019). Linear standard curves were obtained (up to 10 ug/ml Vi) with the acid hydrolysis method, which has the potential to separate amino-uronic acids and O-LPS with higher sensitivity. The establishment of an acid-stable internal standard for quantitation is required.
Methods for robust characterisation of multivalent combinations and Shigella antigens
Shigella LPS and O-Ag analysis may be a laborious task due to a vast diversity of antigens in different serotypes. Currently, primary testing to identify the O-Ag usually limited to techniques using specific identifiers for the serotype detection, such as membrane insertion and ELISAs (Stromberg et al. 2017). The lack of sensitive and selective antigen ligands suitable for every serogroup remains a major problem. Ravenscroft et al. also applied capillary gel electrophoresis for Shigella dysenteriae 1-EPA bioconjugates mentioned above to get better resolution of individual repeating units, as well as to monitor their integrity (Ravenscroft et al. 2016).
Combination methods suggested for future analysis and vaccine design
Coupling high-resolution mass spectrometry and 1H NMR techniques was tested with Neisseria meningitidis serogroup B, enabling simultaneous analysis of conjugation reaction course and final products (Yu et al. 2018). However, the utilisation of mass spectrometry with polydisperse macromolecules (10–100 kDa), like S. Typhi O-PS, provides less information as in the case of glycoconjugates derived from synthetic antigens or less complex glycans (Méndez et al. 2018).
The NEWCARBOVAX project was launched in 2017, which showcases an effective vaccine design platform that has been tested with CPSs from S. Typhi, Hib and type Ib group B Streptococcus, that successfully induced T helper cells (T carbs) successfully (CORDIS | European Commission 2020). This platform is going to be a helpful approach if coupled with research into novel conjugation chemistry approaches in this field.
Any of the new and existing conjugates under development may benefit from the application of Quality by Design principles in the development and testing of vaccines and intermediates. Current technologies discussed in this review are focused on more common glycoconjugate formulations. Nanoparticle-based vaccines, protein capsular matrix vaccines and liposomal encapsulation of polysaccharides are becoming a new trend in vaccine design and will require new validated methods of quality control.
Lastly, classical approaches to evaluating thermal unfolding and stability for lipid-based vaccines destined for hot climates may need to be reconsidered to account for their membrane dynamics. The lipid composition of the membranes may affect membrane phase transitions at relatively lower temperatures than expected for the polysaccharides and some protein domains, and the impact of such dynamics on immunogenicity, safety and protection will need to be considered as shown for Salmonella and Shigella GMMAs using differential scanning calorimetry in combination with monoclonal antibody binding and immunogenicity (Palmieri et al. 2021).
Summary
Limited antibiotic treatment options against the emerging burden of typhoid fever and shigellosis infections and the continuing need for cleaner water, hygiene and sanitation emphasise the urgent need for effective preventions against communicable enteric diseases. Glycoconjugate vaccines could become a promising solution targeting the most infectious strains of these bacteria. With the updated WHO recommendations on the quality, safety and efficacy of typhoid conjugate vaccines adopted by the ECBS in 2020 and SAGE recommendations for typhoid conjugate vaccines to be used as preventive measures in endemic countries or where there is a high burden of antimicrobial resistance (WHO 2017a), analytical methods for evaluating typhoid conjugate vaccines are becoming established. The efforts to produce protocols for Shigella and non-typhoidal Salmonella vaccine development will be supported by academia, institutions, public health bodies and funding organisations over the next few years (NIBSC 2020). These are major steps towards faster commercialisation of glycoconjugates and more complex vaccines to address an emerging global need for enteric disease prevention.
This review highlights applications of conventional methods of glycoconjugate and more complex vaccine structures. Alterations to experimental conditions are constantly made and protocols are being modified together with reference standards to serve the demand for higher accuracy for glycoconjugates that are less susceptible for hydrolysis or behave differently in solution. Together with modern computational approaches, a more precise design of glycoconjugates is possible, which allowing better predictions on vaccine behaviour, structural changes in solution and immunogenicity, which can be applied to a broad spectrum of vaccines. More validation is required to establish the most effective and suitable methods for glycoconjugate and more complex vaccine analysis to bring uniformity to existing protocols, aiding future achievements in this area of research.
References
Abdelhameed AS, Morris GA, Adams GG, Rowe AJ, Laloux O, Cerny L, Bonnier B, Duvivier P, Conrath K, Lenfant C, Harding SE (2012) An asymmetric and slightly dimerized structure for the tetanus toxoid protein used in glycoconjugate vaccines. Carbohydr Polym 90:1831–1835. https://doi.org/10.1016/j.carbpol.2012.07.032
Abdelhameed AS, Adams GG, Morris GA, Almutairi FM, Duvivie P, Conrath K, Harding SE (2016a) A glycoconjugate of Haemophilus influenzae type b capsular polysaccharide with tetanus toxoid protein: hydrodynamic properties mainly influenced by the carbohydrate. Sci Rep 6:22208. https://doi.org/10.1038/srep22208
Abdelhameed AS, Morris GA, Almutairi F, Adams GG, Duvivier P, Conrath K, Harding SE (2016b) Solution conformation and flexibility of capsular polysaccharides from Neisseria meningitidis and glycoconjugates with the tetanus toxoid protein. Sci Rep 6:35588. https://doi.org/10.1038/srep35588
Adisinsight.springer.com (2020a) Typhoid conjugate vaccine - International Vaccine Institute/Incepta/SK Bioscience/PT Biofarma - Adisinsight. https://adisinsight.springer.com/drugs/800048060. Accessed 8 Aug 2020
Adisinsight.springer.com (2020b) Typhoid-tetanus toxoid conjugate vaccine - Cadila Healthcare – Adisinsight. https://adisinsight.springer.com/drugs/800047578. Accessed 8 Aug 2020
Ahsan S, Rahman S (2019) Azithromycin resistance in clinical isolates of Salmonella enterica serovars Typhi and Paratyphi in Bangladesh. Microb Drug Resist 25:8–13. https://doi.org/10.1089/mdr.2018.0109
An S, Yoon Y, Kothari S, Kothari N, Kim J, Lee E, Kim D, Park T, Smith G, Carbis R (2011) Physico-chemical properties of Salmonella Typhi Vi polysaccharide–diphtheria toxoid conjugate vaccines affect immunogenicity. Vaccine 29:7618–7623. https://doi.org/10.1016/j.vaccine.2011.08.019
Anderson M, Sansonetti P, Marteyn B (2016) Shigella diversity and changing landscape: insights for the twenty-first century. Front Cell Infect Microbiol 6:45. https://doi.org/10.3389/fcimb.2016.00045
Angelakopoulos H, Hohmann EL (2000) Pilot study of phoP/phoQ-deleted Salmonella enterica serovar typhimurium expressing Helicobacter pylori urease in adult volunteers. Infect Immun 68:2135–2141. https://doi.org/10.1128/iai.68.4.2135-2141.2000
Anish C, Schumann B, Pereira C, Seeberger P (2014) Chemical biology approaches to designing defined carbohydrate vaccines. Chem Biol 21:38–50. https://doi.org/10.1016/j.chembiol.2014.01.002
Arcuri M, Di Benedetto R, Cunningham AF, Saul A, MacLennan CA, Micoli F (2017) The influence of conjugation variables on the design and immunogenicity of a glycoconjugate vaccine against Salmonella Typhi. PLoS One 12(12):e0189100. https://doi.org/10.1371/journal.pone.0189100
Argimón S, Yeats, C, Goater R et al (2020) A global resource for genomic predictions of antimicrobial resistance and surveillance of Salmonella Typhi at Pathogenwatch. https://doi.org/10.1101/2020.07.03.186692
Ashcroft M, Ritchie J, Nicholson C (1964) Controlled field trial in British Guiana school children of heat-killed-phenolized and acetone-killed lyophilized typhoid vaccines. Am J Epidemiol 79:196–206. https://doi.org/10.1093/oxfordjournals.aje.a120376
Ashcroft M, Singh B, Nicholson C, Ritchie J, Sobryan E, Williams F (1967) A seven-year field trial of two typhoid vaccines in Guyana. Lancet 290:1056–1059. https://doi.org/10.1016/S0140-6736(67)90335-2
Avci F, Berti F, Dull P, Hennessey J, Pavliak V, Prasad A, Vann W, Wacker M, Marcq M (2019) Glycoconjugates: what it would take to master these well-known yet little-understood immunogens for vaccine development. mSphere 4(5):e00520–e00519. https://doi.org/10.1128/mSphere.00520-19
Baliban S, Curtis B, Toema D, Tennant S, Levine M, Pasetti M, Simon R (2018) Immunogenicity and efficacy following sequential parenterally-administered doses of Salmonella Enteritidis COPS:FliC glycoconjugates in infant and adult mice. PLoS Negl Trop Dis 12:e0006522. https://doi.org/10.1371/journal.pntd.0006522
Baliban S, Lu Y, Malley R (2020) Overview of the Nontyphoidal and Paratyphoidal Salmonella vaccine pipeline: current status and future prospects. Clin Infect Dis 71:S151–S154. https://doi.org/10.1093/cid/ciaa514
Bardotti A, Averani G, Berti F, Berti S, Carinci V, D’Ascenzi S, Fabbri B, Giannini S, Giannozzi A, Magagnoli C, Proietti D, Norelli F, Rappouli R, Ricci S, Costantino P (2008) Physicochemical characterisation of glycoconjugate vaccines for prevention of meningococcal diseases. Vaccine 26:2284–2296. https://doi.org/10.1016/j.vaccine.2008.01.022
Barel L, Mulard L (2019) Classical and novel strategies to develop a Shigella glycoconjugate vaccine: from concept to efficacy in human. Hum Vaccines Immunother 15:1338–1356. https://doi.org/10.1080/21645515.2019.1606972
Bayart C, Jean E, Paillagot M, Renoud A, Raillard A, Paladino J, Le Borgne M (2019) Comparison of SEC and AF4 analytical tools for size estimation of typhoid Vi polysaccharides. Anal Methods 11:4851–4858. https://doi.org/10.1039/c9ay00145j
Beresford NJ, Martino A, Feavers IM, Corbel MJ, Bai X, Borrow R, Bolgiano B (2017) Quality, immunogenicity and stability of meningococcal serogroups ACWY-CRM197, DT and TT glycoconjugate vaccines. Vaccine 35:3598–3606. https://doi.org/10.1016/j.vaccine.2017.03.066
Beri S, Gandhi D, Ravenscroft N (2019) Use of NMR as an analytical tool in the process development of conjugate vaccines against Haemophilus influenzae type b (Hib) and meningococcal serogroup A (MenA). Biologicals 62:102–106. https://doi.org/10.1016/j.biologicals.2019.10.005
Berti F, Micoli F (2020) Improving efficacy of glycoconjugate vaccines: from chemical conjugates to next generation constructs. Curr Opin Immunol 65:42–49. https://doi.org/10.1016/j.coi.2020.03.015
Berti F, Ravenscroft N (2015) Characterization of carbohydrate vaccines by NMR spectroscopy. In: Lepenies B (ed) Carbohydrate-based vaccines: methods and protocols. Methods in Molecular Biology 1331. Human Press, New York, pp 189–209
Berti F, De Ricco R, Rappuoli R (2018) Role of O-acetylation in the immunogenicity of bacterial polysaccharide vaccines. Molecules 23:1340. https://doi.org/10.3390/molecules23061340
Bhutta Z, Capeding M, Bavdekar A et al (2014) Immunogenicity and safety of the Vi-CRM197 conjugate vaccine against typhoid fever in adults, children, and infants in south and southeast Asia: results from two randomised, observer-blind, age de-escalation, phase 2 trials. Lancet Infect Dis 14:119–129. https://doi.org/10.1016/s1473-3099(13)70241-x
Bolgiano B, Mawas F, Yost SE, Crane DT, Lemercinier X, Corbel MJ (2001) Effect of physico-chemical modification on the immunogenicity of Haemophilus influenzae type b oligosaccharide-CRM197 conjugate vaccines. Vaccine 19:3189–3200. https://doi.org/10.1016/s0264-410x(01)00024-x
Brunner K, Samassa F, Sansonetti P, Phalipon A (2019) Shigella-mediated immunosuppression in the human gut: subversion extends from innate to adaptive immune responses. Hum Vaccines Immunother 15:1317–1325. https://doi.org/10.1080/21645515.2019.1594132
Caboni M, Pédron T, Rossi O, Goulding D, Pickard D, Citiulo F, MacLennan CA, Dougan G, Thomson NR, Saul A, Sansonetti PJ, Gerke C (2015) An O antigen capsule modulates bacterial pathogenesis in Shigella sonnei. PLoS Pathog 11(3):e1004749. https://doi.org/10.1371/journal.ppat.1004749
Carter J, Blondel C, Zaldívar M, Álvarez S, Marolda C, Valvano M, Contreras I (2007) O-antigen modal chain length in Shigella flexneri 2a is growth-regulated through RfaH-mediated transcriptional control of the wzy gene. Microbiol 153:3499–3507. https://doi.org/10.1099/mic.0.2007/010066-0
Cohen D, Muhsen K (2019) Vaccines for enteric diseases. Hum Vaccines Immunother 15:1205–1214. https://doi.org/10.1080/21645515.2019.1611200
Cohen D, Ashkenazi S, Green MS, Gdalevich M, Robin G, Slepon R, Yavzori M, Orr N, Block C, Ashkenazi I, Shemer J, Taylor DN, Hale TL, Sadoff JC, Pavliakova D, Schneerson R, Robbins JB (1997) Double-blind vaccine-controlled randomised efficacy trial of an investigational Shigella sonnei conjugate vaccine in young adults. Lancet 349:155–159. https://doi.org/10.1016/S0140-6736(96)06255-1
Cohen D, Meron-Sudai S, Bialik A, Asato V, Goren S, Ariel-Cohen O, Reizis A, Hochberg A, Ashkenazi S (2019) Serum IgG antibodies to Shigella lipopolysaccharide antigens – a correlate of protection against shigellosis. Hum Vaccines Immunother 15:1401–1408. https://doi.org/10.1080/21645515.2019.1606971
Cordis.europa.eu (2020) CORDIS | European Commission. [online] Available at: https://cordis.europa.eu/article/id/421458-a-novel-platform-for-the-design-of-effective-vaccines-against-bacteria. Accessed 15 Aug 2020
Crane D, Bolgiano JC (1997) Comparison of the diphtheria mutant toxin, CRM197, with a Haemophilus influenzae type-b polysaccharide-CRM197 conjugate by optical spectroscopy. Eur J Biochem 246(2):320–327. https://doi.org/10.1111/j.1432-1033.1997.00320.x
Crump J, Sjölund-Karlsson M, Gordon M, Parry C (2015) Epidemiology, clinical presentation, laboratory diagnosis, antimicrobial resistance, and antimicrobial management of invasive Salmonella infections. Clin Microbiol Rev 28:901–937. https://doi.org/10.1128/CMR.00002-15
Das A, Mandal J (2019) Extensive inter-strain diversity among clinical isolates of Shigella flexneri with reference to its serotype, virulence traits and plasmid incompatibility types, a study from south India over a 6-year period. Gut Pathog 11(1). https://doi.org/10.1186/s13099-019-0314-9
De Benedetto G, Alfini R, Cescutti P et al (2017a) Characterization of O-antigen delivered by generalized modules for membrane antigens (GMMA) vaccine candidates against nontyphoidal Salmonella. Vaccine 35:419–426. https://doi.org/10.1016/j.vaccine.2016.11.089
De Benedetto G, Cescutti P, Giannelli C, Rizzo R, Micoli F (2017b) Multiple techniques for size determination of generalized modules for membrane antigens from Salmonella typhimurium and Salmonella enteritidis. ACS Omega 2:8282–8289. https://doi.org/10.1021/acsomega.7b01173
Dharmasena MN, Osorio M, Filipova S, Marsh C, Stibitz S, Kopecko DJ (2016) Stable expression of Shigella dysenteriae serotype 1 O-antigen genes integrated into the chromosome of live Salmonella oral vaccine vector Ty21a. Pathogens Dis 74(8):ftw09. https://doi.org/10.1093/femspd/ftw098
Dische Z, Shettles LB (1948) A specific color reaction of methylpentoses and a spectrophotometric micromethod for their determination. J Biol Chem 175:595–603
Dubois M, Gilles K, Hamilton J, Rebers P, Smith F (1951) A colorimetric method for the determination of sugars. Nat 168:167–167. https://doi.org/10.1038/168167a0
Emami P, Motevalian S, Pepin E, Zydney A (2018) Impact of module geometry on the ultrafiltration behavior of capsular polysaccharides for vaccines. J Membr Sci 561:19–25. https://doi.org/10.1016/j.memsci.2018.05.024
Gao F, Swann C, Rigsby P, Rijpkema S, Lockyer K, Logan A, Bolgiano B, Vi IS, Working Group (2019) Evaluation of two WHO First International Standards for Vi PS from Citrobacter freundii and Salmonella enterica subspecies enterica serovar Typhi. Biologicals 57:34–45. https://doi.org/10.1016/j.biologicals.2018.11.004
Gerke C, Colucci A, Giannelli C et al (2015) Production of a Shigella sonnei vaccine based on generalized modules for membrane antigens (GMMA), 1790GAHB. PLoS One 10(8):e0134478. https://doi.org/10.1371/journal.pone.0134478
Giannelli C, Raso M, Palmieri E, De Felice A, Pippi F, Micoli F (2020) Development of a specific and sensitive HPAEC-PAD method for quantification of Vi polysaccharide applicable to other polysaccharides containing amino uronic acids. Anal Chem 92:6304–6311. https://doi.org/10.1021/acs.analchem.9b05107?ref=pdf
Giannelli C, Cappelletti E, Di Benedetto R, Pippi F, Arcuri M, Di Cioccio V, Martin LB, Saul A, Micoli F (2017) Determination of free polysaccharide in Vi glycoconjugate vaccine against typhoid fever. JPharm Biomed Anal 139:143–147
Giardina P, Longworth E, Evans-Johnson R et al (2005) Analysis of human serum immunoglobulin G against O-acetyl-positive and O-acetyl-negative serogroup W135 meningococcal capsular polysaccharide. Clin Diagn Lab Immunol 12:586–592. https://doi.org/10.1128/CDLI.12.5.586-592.2005
Giuntini S, Balducci E, Cerofolini L, Ravera E, Fragai M, Berti F, Luchinat C (2017) Characterization of the conjugation pattern in large polysaccharide-protein conjugates by NMR spectroscopy. Angew Chem Int Ed 56:14997–15001. https://doi.org/10.1002/anie.201709274
Global Burden of Diseases 2019 Disease and Injuries Collaborators (2020) Global burden of 369 diseases and injuries in 204 countries and territories, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 396:1204-22. https://doi.org/10.1016/S0140-6736(20)30925-9 Accessed 2 Nov 2020
Global Burden of Diseases 2019 Risk Factor Collaborators (2020) Global burden of 87 risk factors in 204 countries and territories, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 396:1223-1249. https://doi.org/10.1016/S0140-6736(20)30752-2 Accessed 2 Nov 2020
GSK (2018) Discontinuation of Typherix adult vaccine. https://au.gsk.com/en-au/media/press-releases/2018/discontinuation-of-typherix-adult-vaccine/. Accessed 8 Aug 2020
Harding SE (1992) Sedimentation analysis of polysaccharides. In: Harding SE, Rowe AJ, Horton JC (eds) Analytical ultracentrifugation in biochemistry and polymer science. Royal Society of Chemistry, Cambridge, pp 495–516 www.nottingham.ac.uk/ncmh/documents/papers/Paper96.pdf
Harding SE, Abdelhameed AS, Morris GA (2010) Molecular weight distribution evaluation of polysaccharides and glycoconjugates using analytical ultracentrifugation. Macromol Biosci 10:714–720. https://doi.org/10.1002/mabi.201000072
Harding SE, Abdelhameed AS, Morris GA, Adams G, Laloux O, Cerny L, Bonnier B, Duvivier P, Conrath K, Lenfant C (2012) Solution properties of capsular polysaccharides from Streptococcus pneumoniae. Carbohydr Polym 90:237–242. https://doi.org/10.1016/j.carbpol.2012.05.030
Harding SE, Adams GG, Almutairi F, Alzahrani Q, Erten T, Kök MS, Gillis RB (2015) Ultracentrifuge methods for the analysis of polysaccharides, glycoconjugates, and lignins. Methods Enzymol 562:391–439
Hestrin S (1949) The reaction of acetylcholine and other carboxylic acid derivatives with hydroxylamine, and its analytical application. J Biol Chem 180:249–261
Heyns K, Kiessling G (1967) Strukturaufklärung des Vi-antigens aus Citrobacter freundii (E. coli) 5396/38. Carbohydr Res 3(3):340–353. https://doi.org/10.1016/S0008-6215(00)82210-7
Hindle Z, Chatfield SN, Phillemore J, Bentley M, Johnson J et al (2002) Characterization of Salmonella enterica derivatives harboring defined aroC and Salmonella pathogenicity island 2 type III secretion system (ssaV) mutations by immunization of healthy volunteers. Infect Immun 70:3457–3467
Hitri K, Kuttel M, De Benedetto G et al (2019) O-acetylation of typhoid capsular polysaccharide confers polysaccharide rigidity and immunodominance by masking additional epitopes. Vaccine 37:3866–3875. https://doi.org/10.1016/j.vaccine.2019.05.050
Hlozek J, Ravenscroft N, Kuttel MM (2020) Effects of glucosylation and O-acetylation on the conformation of Shigella flexneri serogroup 2 O-antigen vaccine targets. J Phys Chem B 124:2806–2814. https://doi.org/10.1021/acs.jpcb.0c01595
Ho MM, Lemercinier X, Bolgiano B, Crane DT, Corbel MJ (2001) Conformational stability of the subunit components of meningococcal oligosaccharide C-CRM197 conjugate vaccines. Biotechnol Appl Biochem 33:91–98. https://doi.org/10.1042/BA20000078
Ho MM, Mawas F, Bolgiano B, Lemercinier X, Crane D, Huskisson R, Corbel (2002) Physico-chemical and immunological examination of the thermal stability of tetanus toxoid conjugate vaccines. Vaccine 20:3509–3522. https://doi.org/10.1016/S0264-410X(02)00342-0
Ho MM, Bolgiano B, Martino A, Kairo SK, Corbel MJ (2006) Preclinical laboratory evaluation of a bivalent Staphylococcus aureus saccharide exotoxin A protein conjugate vaccine. Hum Vaccines 2:89–98. https://doi.org/10.4161/hv.2755
Humphries R, Schuetz A (2015) Antimicrobial susceptibility testing of bacteria that cause gastroenteritis. Clin Lab Med 35:313–331. https://doi.org/10.1016/j.cll.2015.02.005
Jin C, Gibani MM, Moore M et al (2017) Efficacy and immunogenicity of a Vi-tetanus toxoid conjugate vaccine in the prevention of typhoid fever using a controlled human infection model of Salmonella typhi: a randomised controlled, phase 2b trial. Lancet 390:2472–2480. https://doi.org/10.1016/S0140-6736(17)32149-9
Jones C, An S, Yoon Y, Kothari S, Sahastrabuddhe S, Carbis R (2020) Spectroscopic characterisation of a series of Salmonella Typhi Vi-diphtheria toxoid glycoconjugate antigens differing in polysaccharide-protein ratio. J Pharm Biomed Anal 181:113100. https://doi.org/10.1016/j.jpba.2020.113100
Jumel K, Ho MM, Bolgiano B (2002) Evaluation of Meningococcal C oligosaccharide conjugate vaccines by size exclusion chromatography/multi-angle laser light scattering. Biotechnol Appl Biochem 36:219–226. https://doi.org/10.1042/BA20020066
Kaminski RW, Wu M, Turbyfill KR et al (2014) Development and preclinical evaluation of a trivalent, formalin-inactivated Shigella whole-cell vaccine. Clin Vaccine Immunol 21:366–382. https://doi.org/10.1128/CVI.00683-13
Kämpf MM, Braun M, Sirena D et al (2015) In vivo production of a novel glycoconjugate vaccine against Shigella flexneri 2a in recombinant Escherichia coli: identification of stimulating factors for in vivo glycosylation. Microb Cell Factories 14:12. https://doi.org/10.1186/s12934-015-0195-7
Kao G, Tsai C (2004) Quantification of O-acetyl, N-acetyl and phosphate groups and determination of the extent of O-acetylation in bacterial vaccine polysaccharides by high-performance anion-exchange chromatography with conductivity detection (HPAEC-CD). Vaccine 22:335–344. https://doi.org/10.1016/j.vaccine.2003.08.008
Khalil I, Troeger C, Blacker B et al (2018) Morbidity and mortality due to Shigella and enterotoxigenic Escherichia coli diarrhoea: The Global Burden of Disease Study 1990–2016. Lancet Infect Dis 18(11):1229–1240. https://doi.org/10.1016/S1473-3099(18)30475-4
Khan M, Franco-Paredes C, Sahastrabuddhe S, Ochiai R, Mogasale V, Gessner B (2017) Barriers to typhoid fever vaccine access in endemic countries. Res Rep Trop Med 8:37–44. https://doi.org/10.2147/RRTM.S97309
Kim MJ, Moon YH, Kim H et al (2018) Cross-protective Shigella whole-cell vaccine with a truncated O-polysaccharide chain. Front Microbiol 9:2609. https://doi.org/10.3389/fmicb.2018.02609
Kis Z, Shattock R, Shah N, Kontoravdi C (2018) Emerging technologies for low-cost, rapid vaccine manufacture. Biotechnol J 14:1800376. https://doi.org/10.1002/biot.201800376
Klemm E, Shakoor S, Page A et al (2018) Emergence of an extensively drug-resistant Salmonella enterica serovar Typhi clone harboring a promiscuous plasmid encoding resistance to fluoroquinolones and third-generation cephalosporins. mBio 9(1). https://doi.org/10.1128/mbio.00105-18
Knirel Y, Sun Q, Senchenkova S, Perepelov A, Shashkov A, Xu J (2015) O-Antigen modifications providing antigenic diversity of Shigella flexneri and underlying genetic mechanisms. Biochem Mosc 80:901–914. https://doi.org/10.1134/S0006297915070093
Kothari N, Genschmer K, Kothari S, Kim J, Briles D, Rhee D, Carbis R (2014) Preparation and testing of a Vi conjugate vaccine using pneumococcal surface protein A (PspA) from Streptococcus pneumoniae as the carrier protein. Vaccine 32:5755–5760. https://doi.org/10.1016/j.vaccine.2014.08.041
Kotloff K, Riddle M, Platts-Mills J, Pavlinac P, Zaidi A (2018) Shigellosis. Lancet 391:801–812. https://doi.org/10.1016/s0140-6736(17)33296-8
Kubler-Kielb J, Vinogradov E, Chu C, Schneerson R (2007) O-Acetylation in the O-specific polysaccharide isolated from Shigella flexneri serotype 2a. Carbohydr Res 342:643–647. https://doi.org/10.1016/j.carres.2006.09.017
Lei QP, Lamb DH, Heller R, Pietrobon P (2000) Quantitation of low level unconjugated polysaccharide in tetanus-toxoid conjugate vaccine by HPAEC-PAD following rapid separation by deoxycholate/HCl. J Pharm Biomed Anal 21:1087–1091. https://doi.org/10.1016/s0731-7085(99)00183-1
Lemercinier X, Martinez-Cabrera I, Jones C (2000) Use and validation of an NMR test for the identity and O -acetyl content of the Salmonella Typhi Vi Capsular Polysaccharide Vaccine. Biologicals 28:17–24. https://doi.org/10.1006/biol.1999.0238
Leyva A, Quintana A, Sánchez M, Rodríguez E, Cremata J, Sánchez J (2008) Rapid and sensitive anthrone–sulfuric acid assay in microplate format to quantify carbohydrate in biopharmaceutical products: method development and validation. Biologicals 36(2):134–141. https://doi.org/10.1016/j.biologicals.2007.09.001
Lindberg A, Karnell A, Weintraub A (1991) The lipopolysaccharide of Shigella bacteria as a virulence factor. Clin Infect Dis 13(Supplement_4):S279–S284. https://doi.org/10.1093/clinids/13.supplement_4.s279
Liston SD, Ovchinnikova OG, Whitfield C (2016) Unique lipid anchor attaches Vi antigen capsule to the surface of Salmonella enterica serovar typhi. Proc Natl Acad Sci U S A 113:6719–6724. https://doi.org/10.1073/pnas.1524665113
Liu B, Knirel YA, Feng L, Perepelov AV, Senchenkova SN, Wang Q, Reeves PR, Wang L (2008) Structure and genetics of Shigella O antigens. FEMS Microbiol Rev 32:627–653. https://doi.org/10.1111/j.1574-6976.2008.00114.x
Liu J, Platts-Mills J, Juma J et al (2016) Use of quantitative molecular diagnostic methods to identify causes of diarrhoea in children: a reanalysis of the GEMS case-control study. Lancet 388:1291–1301. https://doi.org/10.1016/s0140-6736(16)31529-x
Livio S, Strockbine N, Panchalingam S et al (2014) Shigella isolates from the global enteric multicenter study inform vaccine development. Clin Infect Dis 59(7):933–941. https://doi.org/10.1093/cid%2Fciu468
Lockyer K, Gao F, Derrick JP, Bolgiano B (2015) Structural correlates of carrier protein recognition in tetanus toxoid conjugated bacterial protein vaccines. Vaccine 33:1345–1352. https://doi.org/10.1016/j.vaccine.2015.01.046
Lockyer K, Gao F, Francis RJ, Eastwood D, Khatri B, Stebbings R, Derrick JP, Bolgiano B (2020) Higher mass meningococcal group C-tetanus toxoid vaccines conjugated with carbodiimide correlate with greater immunogenicity. Vaccine 38:2859–2869. https://doi.org/10.1016/j.vaccine.2020.02.012
Lyodov VA, Aparin PG (2014) Clinical studies of the vaccine preparation for prevention of Flexner dysentery on the basis of modified Shigella flexneri lipopolysaccharide (based on the results of the I phase of clinical trials). Acta Biomed Sci (East Siberian Biomed J) 2014(4):91–95
MacCalman T, Phillips-Jones M, Harding S (2019) Glycoconjugate vaccines: some observations on carrier and production methods. Biotechnol Genet Eng Rev 35:93–125. https://doi.org/10.1080/02648725.2019.1703614
MacLennan C, Steele D (2019) Invasive non-typhoid Salmonella vaccines. WHO Product Development for Vaccines Advisory Committee 26-28 June 2019. https://www.who.int/immunization/research/meetings_workshops/13_Steele_iNTS_PDVAC_2019.pdf?ua=1. Accessed 22 Oct 2020
MacLennan C, Martin L, Micoli F (2014) Vaccines against invasive Salmonella disease. Hum Vaccines Immunother 10:1478–1493. https://doi.org/10.4161/hv.29054
Mani S, Wierzba T, Walker R (2016) Status of vaccine research and development for Shigella. Vaccine 34:2887–2894. https://doi.org/10.1016/j.vaccine.2016.02.075
Martin L, Simon R, MacLennan C, Tennant S, Sahastrabuddhe S, Khan M (2016) Status of paratyphoid fever vaccine research and development. Vaccine 34:2900–2902. https://doi.org/10.1016/j.vaccine.2016.03.106
Martinez-Becerra FJ, Chen X, Dickenson NE et al (2013) Characterization of a novel fusion protein from IpaB and IpaD of Shigella spp. and its potential as a pan-Shigella vaccine. Infect Immun 81:4470–4477. https://doi.org/10.1128/IAI.00859-13
Meloni E, Colucci AM, Micoli F, Sollai L, Gavini M, Saul A, Di Cioccio V, Calman AM (2015) Simplified low-cost production of O-antigen from Salmonella typhimurium generalized modules for membrane antigens (GMMA). J Biotechnol 198:46–52. https://doi.org/10.1016/j.jbiotec.2015.01.020
Méndez Y, Chang J, Humpierre A et al (2018) Multicomponent polysaccharide–protein bioconjugation in the development of antibacterial glycoconjugate vaccine candidates. Chem Sci 9:2581–2588. https://doi.org/10.1039/C7SC05467J
Micoli F, Rondini S, Pisoni I, Proietti D, Berti F, Costantino P, Rappuoli R, Szu S, Saul A, Martin LB (2011) Vi-CRM197 as a new conjugate vaccine against Salmonella Typhi. Vaccine 29:712–720. https://doi.org/10.1016/j.vaccine.2010.11.022
Micoli F, Rondini S, Gavini M et al (2013) A scalable method for O-antigen purification applied to various Salmonella serovars. Anal Biochem 434:136–145. https://doi.org/10.1016/j.ab.2012.10.038
Micoli F, Ravenscroft N, Cescutti P, Stefanetti G, Londero S, Rondini S, MacLennan C (2014) Structural analysis of O-polysaccharide chains extracted from different Salmonella strains. Carbohydr Res 385:1–8. https://doi.org/10.1016/j.carres.2013.12.003
Micoli F, Adamo R, Costantino P (2018a) Protein carriers for glycoconjugate vaccines: history selection criteria characterization and new trends. Mol 23:1451. https://doi.org/10.3390/molecules23061451
Micoli F, Costantino P, Adamo R (2018b) Potential targets for next generation antimicrobial glycoconjugate vaccines. FEMS Microbiol Rev 42(3):388–423. https://doi.org/10.1093/femsre/fuy011
Micoli F, Rondini S, Alfini R et al (2018c) Comparative immunogenicity and efficacy of equivalent outer membrane vesicle and glycoconjugate vaccines against nontyphoidal Salmonella. Proc Natl Acad Sci 115:10428–10433. https://doi.org/10.1073/pnas.1807655115
Micoli F, Alfini R, Di Benedetto R, Necchi F, Schiavo F, Mancini F, Carducci M, Palmieri E, Balocchi C, Gasperini G, Brunelli B, Costantino P, Adamo R, Piccioli D, Saul A (2020a) GMMA is a versatile platform to design effective multivalent combination vaccines. Vaccines 8:540. https://doi.org/10.3390/vaccines8030540
Micoli F, Bjarnarson SP, Arcuri M, Aradottir Pind AA, Magnusdottir GJ, Necchi F, Di Benedetto R, Carducci M, Schiavo F, Giannelli C, Pisoni I, Martin LB, Del Giudice G, MacLennan CA, Rappuoli R, Jonsdottir I, Saul A (2020b) Short Vi-polysaccharide abrogates T-independent immune response and hyporesponsiveness elicited by long Vi-CRM197 conjugate vaccine. Proc Natl Acad Sci U S A 117:24443–24449. https://doi.org/10.1073/pnas.2005857117
Morona R, Daniels C, Van Den Bosch L (2003) Genetic modulation of Shigella flexneri 2a lipopolysaccharide O antigen modal chain length reveals that it has been optimized for virulence. Microbiol 149(4):925–939. https://doi.org/10.1099/mic.0.26141-0
Mulard L (2017) Bacterial polysaccharides as major surface antigens: interest in O-acetyl substitutions. In Rauter AP, Lindhorst T, Queneau Y (eds) Carbohydr Chemistry:Chemical and biological approaches. Vol 43, Ed. AP Rauter, T Lindhorst, Y Queneau. Roy Soc Chem, London. https://doi.org/10.1039/9781788010641-00071
Muthuirulandi Sethuvel D, Devanga Ragupathi N, Anandan S, Veeraraghavan B (2016) Update on: Shigella new serogroups/serotypes and their antimicrobial resistance. Lett Appl Microbiol 64:8–18. https://doi.org/10.1111/lam.12690
NIBSC (2020) NIBSC to develop International Standards for Shigella and Group B Streptococcus. https://www.nibsc.org/about_us/latest_news/shigella_gbs.aspx. Accessed 27 Sept 2020
NICE (2020) Summary of antimicrobial prescribing guidance – managing common Infections. https://www.nice.org.uk/Media/Default/About/what-we-do/NICE-guidance/antimicrobial%20guidance/summary-antimicrobial-prescribing-guidance.pdf, Accessed 7 Aug 2020
Noyes A, Boesch A, Godavarti R, Titchener-Hooker R, Coffman J, Mukhopadhyay T (2013) High throughput quantification of capsular polysaccharides for multivalent vaccines using precipitation with a cationic surfactant. Vaccine 31:5659–5665. https://doi.org/10.1016/j.vaccine.2013.09.075
Page M, MacLean D, Schudoma C (2016) blastjs: a BLAST+ wrapper for Node.js. BMC Res Notes 9(1). https://doi.org/10.1186/s13104-016-1938-1
Palmieri E, Arato V, Oldrini D, Ricchetti B, Aruta MG, Pansegrau W, Marchi S, Giusti F, Ferlenghi I, Rossi O, Alfini R, Giannelli C, Gasperini G, Necchi F, Micoli F (2021) Stability of Outer Membrane Vesicles-Based Vaccines, Identifying the Most Appropriate Methods to Detect Changes in Vaccine Potency. Vaccines 9(3):229
Pastor Y, Camacho AI, Zúñiga-Ripa A, Merchán A, Rosas P, Irache JM, Gamazo C (2018) Towards a subunit vaccine from a Shigella flexneri ΔtolR mutant. Vaccine 36:7509–7519. https://doi.org/10.1016/j.vaccine.2018.10.066
Perepelov A, Shekht M, Liu B, Shevelev S, Ledov V, Senchenkova S, L’vov V, Shashkov A, Feng L, Aparin P, Wang L, Knirel Y (2012) Shigella flexneri O-antigens revisited: final elucidation of the O-acetylation profiles and a survey of the O-antigen structure diversity. FEMS Immunol Med Microbiol 66:201–210
Puzari M, Sharma M, Chetia P (2018) Emergence of antibiotic resistant Shigella species: a matter of concern. J Infect Public Health 11:451–454. https://doi.org/10.1016/j.jiph.2017.09.025
Qadri A, Ghosh S, Talwar G (1990) Monoclonal antibodies against two discrete determinants on Vi capsular polysaccharide. J Immunoass 11(2):235–250. https://doi.org/10.1080/01971529008053271
Rappuoli R, De Gregorio E, Costantino P (2018) On the mechanisms of conjugate vaccines. Proc Natl Acad Sci 116:14–16. https://doi.org/10.1073/pnas.1819612116
Raso M, Gasperini G, Alfini R et al (2020) GMMA and glycoconjugate approaches compared in mice for the development of a vaccine against Shigella flexneri serotype 6. Vaccines 8:160. https://doi.org/10.3390/vaccines8020160
Ravenscroft N, Cescutti P, Gavini M, Stefanetti G, MacLennan CA, Martin LB, Micoli F (2015a) Structural analysis of the O-acetylated O-polysaccharide isolated from Salmonella Paratyphi A and used for vaccine preparation. Carbohydr Res 404:108–116. https://doi.org/10.1016/j.carres.2014.12.002
Ravenscroft N, Costantino P, Talaga P et al (2015b) Glycoconjugate vaccines. In: Nunnally BK, Sitrin RD, Turula VE (eds) Vaccine analysis: strategies, principles, and control. Springer-Verlag, Berlin Heidelberg, pp 301–381. https://doi.org/10.1007/978-3-662-45024-6_8
Ravenscroft N, Haeuptle M, Kowarik M et al (2016) Purification and characterization of a Shigella conjugate vaccine produced by glycoengineering Escherichia coli. Glycobiology 26(1):51–62. https://doi.org/10.1093/glycob/cwv077
Reddy EA, Shaw AV, Crump JA (2010) Community-acquired bloodstream infections in Africa: a systematic review and meta-analysis. Lancet Infect Dis 10:417–432. https://doi.org/10.1016/S1473-3099(10)70072-4
Riddle M, Chen W, Kirkwood C, MacLennan C (2018) Update on vaccines for enteric pathogens. Clin Microbiol Infect 24(10):1039–1045. https://doi.org/10.1016/j.cmi.2018.06.023
Rigsby P, Beamish E, Hockley J et al (2020) Evaluation of a standardised Vi poly-l-lysine ELISA for serology of Vi capsular polysaccharide antibodies. Biologicals 66:21–29. https://doi.org/10.1016/j.biologicals.2020.05.002
Rijpkema S, Hockley J, Logan A et al (2018) Establishment of the first International Standard for human anti-typhoid capsular Vi polysaccharide IgG. Biologicals 56:29–38. https://doi.org/10.1016/j.biologicals.2018.09.001
Robbins J, Robbins J (1984) Reexamination of the protective role of the capsular polysaccharide (Vi antigen) of Salmonella typhi. J Infect Dis 150:436–449. https://doi.org/10.1093/infdis/150.3.436
Rohrer JS (2020) Vaccine quality ensured by high-performance anion-exchange chromatography with pulsed amperometric detection. SLAS Technol 25(4):320–328. https://doi.org/10.1177/2472630319890309
Rosminzdrav (2020) Gosudarstvenniy Reestr Lekarstvennih Sredstv. Registered Clinical Study №161 (04.04.2019) http://grls.rosminzdrav.ru/CIPermissionMini.aspx?CIStatementGUID=23eee6a9-9c4e-47e2-992d-1ed6d781013a&CIPermGUID=CB5A344B-8A5B-4E1B-BB07-24C8D3ADCA68 . Accessed 29 Oct 2020
Rossi O, Pesce I, Giannelli C, Aprea S, Caboni M, Citiulo F, Valentini S, Ferlenghi I, MacLennan CA, D’Oro U, Saul A, Gerke C (2014) Modulation of endotoxicity of Shigella generalized modules for membrane antigens (GMMA) by genetic lipid A modifications: relative activation of TLR4 and TLR2 pathways in different mutants. J Biol Chem 289(36):24922–24935. https://doi.org/10.1074/jbc.m114.566570
Safari D, Marradi M, Chiodo F, Th Dekker H, Shan Y, Adamo R, Oscarson S, Rijkers G, Lahmann M, Kamerling J, Penadés S, Snippe H (2012) Gold nanoparticles as carriers for a synthetic Streptococcus pneumoniaetype 14 conjugate vaccine. Nanomedicine 7:651–662. https://doi.org/10.2217/nnm.11.151
Sahastrabuddhe S, Saluja T (2019) Overview of the typhoid conjugate vaccine pipeline: current status and future plans. Clin Infect Dis 68(Supplement_1):S22–S26. https://doi.org/10.1093/cid/ciy884
Salman M, St Michael F, Ali A, Jabbar A, Cairns C, Hayes AC, Rahman M, Iqbal M, Haque A, Cox AD (2017) First characterization of immunogenic conjugates of Vi negative Salmonella Typhi O-specific polysaccharides with rEPA protein for vaccine development. J Immunol Methods 450:27–33. https://doi.org/10.1016/j.jim.2017.07.005
Serum Institute of India (2013) A novel quantification method for vaccines. Patent application. WO2013046226A3
Shane A, Mody R, Crump J et al (2017) 2017 Infectious Diseases Society of America Clinical Practice Guidelines for the Diagnosis and Management of Infectious Diarrhea. Clin Infect Dis 65:1963–1973. https://doi.org/10.1093/cid/cix959
Stanaway J, Reiner R, Blacker B et al (2019) The global burden of typhoid and paratyphoid fevers: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Infect Dis 19:369–381. https://doi.org/10.1016/S1473-3099(18)30685-6
Steele AD, Carey ME, Kumar S, MacLennan CA, Ma LF, Diaz Z, Zaidi AKM (2020) Typhoid conjugate vaccines and enteric fever control: where to next? Clin Infect Dis 71(Supplement_2):S185–S190. https://doi.org/10.1093/cid/ciaa343
Stone AL, Szu SC (1988) Application of optical properties of the Vi capsular polysaccharide for quantitation of the Vi antigen in vaccines for typhoid fever. J Clin Microbiol 26:719–725
Stromberg L, Mendez H, Mukundan H (2017) Detection methods for lipopolysaccharides: past and present. In: Escherichia coli - recent advances on physiology, pathogenesis and biotechnological applications. InTech, pp 141–168. https://doi.org/10.5772/intechopen.68311
Sun X, Stefanetti G, Berti F, Kasper D (2018) Polysaccharide structure dictates mechanism of adaptive immune response to glycoconjugate vaccines. Proc Natl Acad Sci 116:193–198. https://doi.org/10.1073/pnas.1816401115
Syed K, Saluja T, Cho H et al (2020) Review on the recent advances on typhoid vaccine development and challenges ahead. Clin Infect Dis 71(Supplement_2):S141–S150. https://doi.org/10.1093/cid/ciaa504
Szu S, Li X, Stone A, Robbins J (1991) Relation between structure and immunologic properties of the Vi capsular polysaccharide. Infect Immun 59:4555–4561
Thiem V, Lin F, Canh D et al (2011) The Vi conjugate typhoid vaccine is safe elicits protective levels of IgG anti-Vi and is compatible with routine infant vaccines. Clin Vaccine Immunol 18:730–735. https://doi.org/10.1128/cvi.00532-10
Thompson CN, Duy PT, Baker S (2015) The rising dominance of Shigella sonnei: an intercontinental shift in the etiology of bacillary dysentery. PLoS Negl Trop Dis 9(6):e0003708. https://doi.org/10.1371/journal.pntd.0003708
Toapanta FR, Bernal PJ, Kotloff KL et al (2018) T cell mediated immunity induced by the live-attenuated Shigella flexneri 2a vaccine candidate CVD 1208S in humans. J Transl Med 16:61. https://doi.org/10.1186/s12967-018-1439-1
Tribble D (2017) Resistant pathogens as causes of traveller’s diarrhea globally and impact(s) on treatment failure and recommendations. J Travel Med 24(suppl_1):S6–S12. https://doi.org/10.1093/jtm/taw090
Troeger C, Blacker B, Khalil I et al (2018) Estimates of the global regional and national morbidity mortality and aetiologies of diarrhoea in 195 countries: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Infect Dis 18:1211–1228. https://doi.org/10.1016/S1473-3099(18)30362-1
United States Pharmacopeial Convention (2018) Nuclear magnetic resonance spectroscopy: identity testing of bacterial polysaccharides used in vaccine manufacturing. Chapter 198
Wang BZ (2003) Current status and development trend of dysentery vaccine. Progress in Microbiology and Immunology 4:54–59
Wen L, Edmunds G, Gibbons C, Zhang J, Gadi MR, Zhu H, Fang J, Liu X, Kong Y, Wang PG (2018) Toward Automated Enzymatic Synthesis of Oligosaccharides. Chem Rev 118(17):8151–8187
Wessels M, Paoletti L, Guttormsen H, Michon F, D’Ambra A, Kasper D (1998) Structural properties of group b Streptococcal type III polysaccharide conjugate vaccines that influence immunogenicity and efficacy. Infect Immun 66:2186–2192
West N (2005) Optimization of virulence functions through glucosylation of Shigella LPS. Science 307:1313–1317
Whitfield C, Williams DM, Kelly SD (2020) Lipopolysaccharide O-antigens: bacterial glycans made to measure. J Biol Chem 295:10593–10609. https://doi.org/10.10741/jbc.REV120.009402
WHO (2005) Guidelines for the control of shigellosis including epidemics due to Shigella dysenteriae type 1. Geneva, World Health Organization. http://apps.who.int/iris/bitstream/10665/43252/1/924159330X.pdf?ua=1&ua=1. Accessed 3 Aug 2020
WHO (2010) WHO recommendations on the management of diarrhoea and pneumonia in HIV infected infants and children. Geneva, World Health Organization. https://apps.who.int/iris/bitstream/handle/10665/44471/9789241548083_eng.pdf?sequence=1. Accessed 20 Oct 2020
WHO (2013) Pocket Book Of Hospital Care For Children. Geneva, World Health Organization. https://apps.who.int/iris/bitstream/handle/10665/81170/9789241548373_eng.pdf?sequence=1. Accessed 20 Oct 2020
WHO. (2014a) Antimicrobial resistance global report on surveillance. Geneva, World Health Organization. http://apps.who.int/iris/bitstream/10665/112642/1/9789241564748_eng.pdf. Accessed 3 Aug 2020
WHO (2014b) Guidelines on the quality, safety and efficacy of typhoid conjugate vaccines. Geneva, World Health Organization, WHO Tech Rep Ser. 987, Annex 3. https://www.who.int/biologicals/areas/vaccines/TRS_987_Annex3.pdf?ua=1. Accessed 13 Aug 2020
WHO (2017a) Background paper to SAGE on typhoid vaccine policy recommendations. Geneva, World Health Organization. https://www.who.int/immunization/sage/meetings/2017/october/1_Typhoid_SAGE_background_paper_Final_v3B.pdf?ua=1, accessed 05 Nov 2020
WHO (2017b) Evaluation of candidate international standards for Vi polysaccharide from citrobacter freundii and salmonella enterica subspecies enterica serovar Typhi. Geneva, World Health Organization, WHO/BS/2017.2310. https://www.who.int/biologicals/expert_committee/BS.2017.2310_Vi_PS_Report_for_WHO_Final.pdf. Accessed 3 Aug 2020
WHO (2017c) The selection and use of essential medicines. Geneva, World Health Organization, WHO Technical Report Series 1006 https://apps.who.int/iris/bitstream/handle/10665/259481/9789241210157-eng.pdf. Accessed 15 Aug 2020
WHO (2018) Global antimicrobial resistance surveillance system (GLASS) Report. Geneva, World Health Organization. https://apps.who.int/iris/bitstream/handle/10665/259744/9789241513449-eng.pdf?sequence=1. Accessed 20 Oct 2020
WHO (2020a) Draft WHO preferred product characteristics for vaccines against Shigella. Geneva, World Health Organization. https://www.who.int/immunization/research/ppc-tpp/PPC_Shigella_draft_for_review_april2020.pdf?ua=1. Accessed 14 Aug 2020
WHO (2020b) Recommendations to assume the quality, safety and efficacy of typhoid conjugate vaccines. Replacement of WHO Technical Report Series, No. 987, Annex 3. Geneva, World Health Organization. https://www.who.int/publications/m/item/tcv71-recommendations
Williams P and Berkley J (2016) Dysentery (shigellosis) current who guidelines and the WHO essential medicine list for children. https://www.who.int/selection_medicines/committees/expert/21/applications/s6_paed_antibiotics_appendix5_dysentery.pdf?ua=1. Accessed 12 Aug 2020
Williams P, Berkley J (2018) Guidelines for the treatment of dysentery (shigellosis): a systematic review of the evidence. Paediatr Int Child Health 38(sup1):S50–S65
Xu Q, Abeygunawardana C, Ng A, Sturgess A, Harmon B, Hennessey J (2005) Characterization and quantification of C-polysaccharide in Streptococcus pneumoniae capsular polysaccharide preparations. Anal Biochem 336:262–272
Yu H, An Y, Battistel M, Cipollo J, Freedberg D (2018) Improving analytical characterization of glycoconjugate vaccines through combined high-resolution MS and NMR: Application to Neisseria meningitidis serogroup B oligosaccharide-peptide glycoconjugates. Anal Chem 90:5040–5047
Acknowledgements
The authors are grateful to Dr. Gianluigi De Benedetto and Dr. Sjoerd Rijpkema for their review of the manuscript and to G.DeB. for method optimisation.
Funding
Funding to A.B. was provided by the National Centre for Macromolecular Hydrodynamics, School of Biosciences, University of Nottingham. S.E.H. was supported by the UK Biotechnology and Biological Sciences Research Council [grant number BB/T006404/1] and the Norwegian Ministry of Education and Research.
Author information
Authors and Affiliations
Contributions
Aleksandra Bazhenova performed the literature search and drafted the review, which was supervised by Stephen Harding, and critically revised by all of the authors. Aleksandra Bazhenova performed the HPAEC-PAD analysis, which was supervised by Fang Gao and Barbara Bolgiano.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bazhenova, A., Gao, F., Bolgiano, B. et al. Glycoconjugate vaccines against Salmonella enterica serovars and Shigella species: existing and emerging methods for their analysis. Biophys Rev 13, 221–246 (2021). https://doi.org/10.1007/s12551-021-00791-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12551-021-00791-z