Abstract
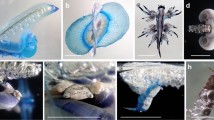
Hyperiidean amphipods are a major prey for fish and seabirds. In the Southern Ocean, they are particularly abundant, with distributions ranging from the Polar Frontal Zone to Antarctic shelf waters. The species Hyperiella dilatata has previously been reported to show a peculiar anti-predatory behaviour: It captures chemically protected, gymnosome pteropods in the water column and carries them on its dorsum, like a backpack. We report this association at four oceanic sampling sites between latitudes 45° and 71° S. Molecular barcodes of both hosts and pteropods are provided and compared with those of other hyperiidean and pteropod specimens. Morphological identifications as well as molecular analyses show a so far undocumented association of Hyperiella antarctica with the pteropod Spongiobranchaea australis in the Polar Frontal Zone (Lazarev Sea). H. dilatata carried Clione limacina antarctica specimens in the Weddell Sea, as recorded previously for the Ross Sea. Lengths of the abducted pteropods varied between 1 and 5 mm, with the biggest pteropod measuring more than half the host’s size. One of the abducting amphipods was a female carrying eggs. The formation of such tandem is known to be very efficient as protection from visually hunting icefish in the crystal-clear coastal waters around the Antarctic continent; however, in the open ocean, this behaviour was so far undocumented. Here, we develop hypotheses on its origin and function.



Similar content being viewed by others
References
Asao H, Nakamura Y, Furuya Y, Kuwahara S, Baker BJ, Kiyota H (2010) Synthesis of pteroenone and its stereoisomers, a defensive metabolite of the abducted Antarctic pteropod Clione antarctica. Helv Chim Acta 93(10):1933–1944
Barnard KH (1930) Crustacea. Part XI - Amphipoda. British Antarctic (“Terra Nova”) Expedition, 1910. Natural history Rep, Zoolog 8(4):307–454
Barrera-Oro E (2003) Analysis of dietary overlap in Antarctic fish (Notothenioidei) from the South Shetland Islands: no evidence of food competition. Polar Biol 26:631–637
Böer M, Graeve M, Kattner G (2006) Impact of feeding and starvation on the lipid metabolism of the Arctic pteropod Clione limacina. J Exp Mar Biol Ecol 328:98–112
Boulenger GA (1902) Pisces. Report on the collections of natural history made in the Antarctic regions during the voyage of the ‘Southern Cross’. Brit Mus (natural history), London, pp 174-189
Bovallius C (1887) Systematical list of the Amphipoda Hyperiidea. Kungliga Svenska Vetenskapsakademiens Handlingar 11:16, 50 pp
Bowman TE (1973) Pelagic amphipods of the genus Hyperia and closely related genera. Smithson Contrib Zool 136:1–76
Browne WE, Haddock SHD, Martindale MQ (2007) Phylogenetic analysis of lineage relationships among hyperiid amphipods as revealed by examination of the mitochondrial gene, cytochrome oxidase 1 (CO1). Integr Comp Biol 47(6):815–830
Bryan PJ, Yoshida WY, McClintock JB, Baker BJ (1995) Ecological role for pteroenone, a novel antifeedant from the conspicuous Antarctic pteropod Clione antarctica (Gymnosomata: Gastropoda). Mar Biol 122:271–277
Casaux RJ, Mazzotta AS, Barrera-Oro ER (1990) Seasonal aspects of the biology and diet of nearshore nototheniid fish at Potter Cove, South Shetland Islands, Antarctica. Polar Biol 11:63–72
Cheng F, Wang M, Sun S, Li C, Zhang Y (2013) DNA barcoding of Antarctic marine zooplankton for species identification and recognition. Adv Polar Sci 24(2):119–127
Claus C (1879) Die Gattungen und Arten der Platysceliden in systematischer Übersicht. Arbeiten aus dem Zoologischen Institut der Universität zu Wien 2:147–198
Conover RJ, Lalli CM (1974) Feeding and growth in Clione limacina (Phipps), a pteropod mollusc. II. Assimilation, metabolism, and growth efficiency. J Exp Mar Biol Ecol 16(2):131–154
Costa FO, deWaard JR, Boutillier J, Ratnasingjam S, Dooh RT, Hajibabaei M, Hebert PDN (2007) Biological identifications through DNA barcodes: the case of the Crustacea. Can J Fish Aquat Sci 64(2):272–295
Dall WH (1871) Descriptions of sixty new forms of molluscs and brachiopods of the southeastern coast of North America and the North Pacific Ocean, with notes on others already described. Am J Conch 7(2):93–180
Dana JD (1853) On the classification and geographical distribution of Crustacea. The report on Crustacea of the U. S. exploring expedition during the years 1838-42 under the command of Charles Wilkes, U.S.N. 13(2):689–1618. Philadelphia
Duhamel G, Hulley PA, Causse R, Koubbi P, Vacchi M, Pruvost P, Vigetta S, Irisson JO, Mormède S, Belchier M, Dettai A, Detrich W, Gutt J, Jones CD, Kock KH, Abellan LJL, Van de Putte A (2014) Biogeographic patterns of fish. In: De Broyer C, Koubbi P, Griffiths HJ, Raymond B, Udekem d’Acoz C d’ et al. (eds) Biogeographic Atlas of the Southern Ocean. Scientific Committee on Antarctic Research, Cambridge, pp 323–362
Fanta E, Donatti L, Freiberger S (1999) Visual sufficiency in food detection and initiation of feeding behaviour in the Antarctic fish Trematomus newnesi Boulanger. Antarct Rec 43(2):221–236
Filin AA, Gorchinsky KV, Kiseleva VM (1991) Biomass of myctophids in the Atlantic sector of the Southern Ocean as estimated by acoustic surveys. Selected scientific papers (SC-CAMLR-SSP/7). CCAMLR, Hobart, Australia, pp 417–431
Flores H, van Franeker JA, Cisewski B, Leach H, Van de Putte AP, Meesters E, Bathmann U, Wolff WJ (2011) Macrofauna under sea ice and in open surface layer of the Lazarev Sea, Southern Ocean. Deep-Sea Res II 58:1948–1961
Folmer O, Black M, Hoeh R, Lutz R, Vrijenhoek R (1994) DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol Mar Biol Biotechn 3(5):294–299
Foster BA (1987) Composition and abundance of zooplankton under the spring sea-ice of McMurdo Sound, Antarctica. Polar Biol 8(1):41–48
Foster BA (1989) Time and depth comparisons of sub-ice zooplankton in McMurdo Sound, Antarctica. Polar Biol 9(7):431–435
Foster BA, Montgomery JC (1993) Planktivory in benthic nototheniid fish in McMurdo Sound, Antarctica. Environ Biol Fish 36(3):313–318
Foster BA, Cargill JM, Montgomery JC (1987) Planktivory in Pagothenia borchgrevinki (Pisces: Nototheniidae) in McMurdo Sound, Antarctica. Polar Biol 8(1):49–54
Gasca R, Haddock SHD (2004) Associations between gelatinous zooplankton and hyperiid amphipods (Crustacea: Peracarida) in the Gulf of Mexico. Hydrobiologia 530(531):529–535
Guérin FE (1825) Encyclopédie Méthodique Histoire Naturelle. Entomologie, ou histoire naturelle des Crustacés, des Arachnides et des Insectes par M. Latreille. Tome 10. Paris
Harbison GR, Biggs DC, Madin LP (1977) The associations of Amphipoda Hyperiidea with gelatinous zooplankton—II. Associations with Cnidaria, Ctenophora and Radiolaria. Deep-Sea Res 24:465–488
Hunt BPV, Pakhomov EA, Hosie GW, Siegel V, Ward P, Bernard K (2008) Pteropods in Southern Ocean ecosystems. Progr Oceanogr 78(3):193–221
Hunt B, Strugnell J, Bednarsek N, Linse K, Nelson RJ, Pakhomov E, Seibel B, Steinke D, Würzberg L (2010) Poles apart: the “bipolar” pteropod species Limacina helicina is genetically distinct between the Arctic and Antarctic oceans. PLoS One 5(3):e9835
Kattner G, Hagen W, Graeve M, Albers C (1998) Exceptional lipids and fatty acids in the pteropod Clione limacina from both polar oceans. Mar Chem 61:219–228
Klussmann-Kolb A, Dinapoli A (2006) Systematic position of the pelagic Thecosomata and Gymnosomata within Opisthobranchia (Mollusca, Gastropoda)—revival of the Pteropoda. J Zool Syst Evol Res 44:118–129
Laval P (1980) Hyperiid amphipods as crustacean parasitoids associated with gelatinous plankton. Oceanogr Mar Biol Ann Rev 18:11–56
Lesueur BHMD (1821) Hyale, Hyalaea (Malacoz). In: Cuvier F (ed) Dictionnaire des Sciences Naturelles Levrault, Strasbourg & Le Normant, Paris, vol 22, pp 65–83
Loeb VJ, Santora JA (2013) Pteropods and climate off the Antarctic Peninsula. Prog Ocean 116:31–48
Loeb V, Outram D, Puglise K, Armstrong WA, Hobday A, Nelson MM, Phleger CF, Sterling J, Watters G, Yender R (1998) Direct krill and zooplankton sampling. CAMLR 1996/97 Field season reports LJ-97. Southwest Fisheries Science Center, Antarctic Ecosystem Research Group
McClintock JB, Baker BJ (1998) Chemical ecology in Antarctic seas. Am Sci 86:254–263
McClintock JB, Janssen J (1990) Pteropod abduction as a chemical defence in a pelagic Antarctic amphipod. Nature 346:424–426
Minichev YS (1976) Subclass back-gilled molluscs. In: Animals and plants of Peter-the-Great Bay. Leningrad, Nauka, pp 92–95 (in Russian)
Montagu G (1815) Descriptions of several new or rare animals, principally marine, discovered on the south coast of Devonshire. Trans Linn Soc London 11(1):1–26
Müller F (1864) Für Darwin. Wilhelm Engelmann, Leipzig, 91 pp
Niebuhr C (1776) Icones rerum naturalium, quas in itinere orientali depingi curavit Petrus Forskål, prof. Haun., post mortem auctoris... edidit Carsten Niebuhr. Hauniae [Copenhagen], 43 pp.
Orbigny AD d' (1836) Voyage dans l’Amérique méridionale exécuté pendant les années 1826, 1827, 1828, 1829, 1830, 1831, 1832 et 1833, Tome 5, Partie 3. Mol Ther:758 pp
Pakhomov EA, Perissinotto R, Froneman PW (1999) Predation impact of carnivorous macrozooplankton and micronekton in the Atlantic sector of the Southern Ocean. J Mar Sys 19:47–64
Phleger CF, Nelson MM, Mooney B, Nichols PD (1999) Lipids of abducted Antarctic pteropods, Spongiobranchaea australis, and their hyperiid amphipod host. Comp Biochem Physiol B 124:295–307
Ratnasingham S, Hebert PD (2007) BOLD: The Barcode of Life Data System (http://www. barcodinglife. org). Mol Ecol Notes 7(3):355–364.
Roberts D, Hopcroft RR, Hosie GW (2014) Southern Ocean pteropods. In: De Broyer C, Koubbi P, Griffiths HJ, Raymond B, Udekem d’Acoz C d’ et al (eds) Biogeographic Atlas of the Southern Ocean. Scientific Committee on Antarctic Research, Cambridge, pp 276–283
Rodhouse G, White MG (1995) Cephalopods occupy the ecological niche of epipelagic fish in the Antarctic polar frontal zone. Biol Bull 189(2):77–80
Schlitzer R (2018) Ocean Data View, https://odv.awi.de
Sheader M, Batten SD (1995) Comparative study of sympatric populations of two hyperiid amphipods, Primno johnsoni and P. evansi, from the eastern North Atlantic Ocean. Mar Biol 124:43–50
Smith EA (1902) VII. Mollusca. Report on the collections of natural history made in the Antarctic regions during the voyage of the “Southern Cross” 201-213, pls 24-25
Sromek L, Lasota R, Szymelfenig M, Wolowicz M (2015) Genetic evidence for the existence of two species of the “bipolar” pelagic mollusc Clione limacina. Am Malacol Bull 33(1):118–120
Stebbing TRR (1888) Report on the Amphipoda collected by H.M.S. Challenger during the years 1873-1876. Report on the scientific results of the voyage of H.M.S. challenger during the years 1873-76. Zoology 29:1–1737
Stephensen K (1925) Hyperiidea-Amphipoda (part 3: Lycaeopsidae, Pronoidae, Lycaeidae, Brachyscelidae, Oxycephalidae, Parascelidae, Platyscelidae). Report on the Danish oceanographical expeditions 1908-1910 to the Mediterranean and adjacent seas 2:151–252
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30(12):2725–2729
Vacchi M, La Mesa M (1995) The diet of the Antarctic fish Trematomus newnesi Boulenger, 1902 (Nototheniidae) from Terra Nova Bay, Ross Sea. Ant Sci 7(1):37–38
Vinogradov GM (1999) Amphipoda. In: Boltovskoy D (ed) South Atlantic zooplankton. Backhuys Publishers, Leiden, pp 1141–1240
Weigmann-Haass R (1989) Zur Taxonomie und Verbreitung der Gattung Hyperiella Bovallius 1887 im antarktischen Teil des Atlantik (Crustacea: Amphipoda: Hyperiidae). Senckenberg Biol 69:177–191
Zeidler W (2015) A review of the hyperiidean amphipod genus Hyperoche Bovallius, 1887 (Crustacea: Amphipoda: Hyperiidea: Hyperiidae), with the description of a new genus to accommodate H. shihi Gasca, 2005. Zootaxa 3905(2):151–192
Zeidler W, De Broyer C (2009) Catalogue of the Hyperiidean Amphipoda (Crustacea) of the Southern Ocean with distribution and ecological data. In: De Broyer C (ed) Census of Antarctic marine life: synopsis of the Amphipoda of the Southern Ocean. vol. 3. Bull l’Institut R Sci Nat Belgique, Biol 79:1–96
Zeidler W, De Broyer C (2014) Amphipoda: Hyperiidea. In: De Broyer C, Koubbi P, Griffiths HJ, Raymond B, Udekem d’Acoz C d’ et al (eds) Biogeographic Atlas of the Southern Ocean. Scientific Committee on Antarctic Research, Cambridge, pp 303–308
Acknowledgements
We greatly acknowledge Dr. Olaf Boebel as chief scientist during the expedition PS103, for the numerous sampling opportunities, as well as the captain and crew of R/V Polarstern for their skilful support. Samples were also obtained during the R/V Polarstern expedition PS82 (ANT-XXIX/9) to the Filchner area of the Weddell Sea, for which we are grateful to the chief scientist Dr. Rainer Knust, captain and crew as well as fellow scientists of the pelagic sampling team for providing amphipods. This publication is a result of the Polarstern Grant No. AWI_PS103_03 of the project “InterPelagic” and Grant No. AWI_PS82_03 of the Filchner expedition. A permit for sampling south of 60° S under the Antarctic Treaty was obtained from the German Environmental Agency (UBA) with the number II 2.8–94003-3/382. Special thanks go to Franz Schröter and Simon Schöbinger for their valuable support in sorting samples during PS103.
Funding
This work was supported by the German Science Foundation/Deutsche Forschungsgemeinschaft (DFG) in the framework of the Priority Programme 1158 on “Antarctic Research with comparative investigations in Arctic ice areas” by the grant HA 7627/1-1 to the first author.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national and/or institutional guidelines for the care and use of animals (invertebrates) were followed.
Sampling and field studies
The necessary permit for sampling and observational field studies have been obtained by the authors from the competent authorities and are mentioned in the acknowledgements.
Additional information
Communicated by A. Brandt
Rights and permissions
About this article
Cite this article
Havermans, C., Hagen, W., Zeidler, W. et al. A survival pack for escaping predation in the open ocean: amphipod – pteropod associations in the Southern Ocean. Mar Biodiv 49, 1361–1370 (2019). https://doi.org/10.1007/s12526-018-0916-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12526-018-0916-3