Abstract
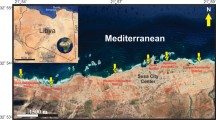
The Golfe d’Arguin offshore of northern Mauritania hosts a rare modern analogue for heterozoan carbonate production in a tropical marine setting. Dominated by ocean upwelling and with additional fertilisation by iron-rich aeolian dust, this naturally eutrophic marine environment lacks typical photozoan communities. A highly productive, tropical cosmopolitan biota dominated by molluscs and suspension-feeders such as bryozoans and balanids characterises the carbonate-rich surface sediments. Overall biodiversity is relatively low and the species present are tolerant against the eutrophic and low-light conditions, the strong hydrodynamic regime governed by ocean upwelling, and the unstable, soft-bottom seafloor with few hard substrata. Here, we describe an ectosymbiosis between the hermit crab Pseudopagurus granulimanus (Miers, 1881) and monospecific assemblages of the encrusting cheilostome bryozoan Acanthodesia commensale (Kirkpatrick and Metzelaar, 1922) that cohabits vacant gastropod shells. Nucleating on an empty gastropod shell, the bryozoan colonies form multilamellar skeletal crusts that produce spherical encrustations and extend the living chamber of the hermit crab through helicospiral tubular growth. This non-obligate mutualistic symbiosis illustrates the adaptive capabilities and benefits from a close partnership in a complex marine environment, driven by trophic conditions, high water energies and instable substratum. Sectioned bryoliths show that between 49 and 97 % of the solid volume of the specimens consists of bryozoan skeleton.



Similar content being viewed by others
References
Adey WH, MacIntyre IG (1973) Crustose coralline algae: a re-evaluation in the geological sciences. Geol Soc Am Bull 84:883–903
Amini ZZ, Adabi MH, Burrett CF, Quilty PG (2004) Bryozoandistribution and growth form associations as a tool in environmentalinterpretation, Tasmania, Australia. Sediment Geol 167:1–15.
Ardovini R, Cossignani T (2004) West African seashells (including Azores, Madeira and Canary Is.) Conchiglie dell’Africa Occidentale (incluse Azzorre, Madeira e Canarie). English-Italian edition. L’Informatore Piceno, Ancona
Campredon P (2000) Entre le Sahara et L’Atlantique—Le parc National du Banc d’Arguin—Mauritanie. FIBA, la Tour du Valat
Balson PS, Taylor PD (1982) Palaeobiology and systematics of large cyclostome bryozoans from the Pliocene Coralline Crag of Suffolk. Palaeontology 25(3):529–554
Boucher DH (1988) The biology of mutualism: ecology and evolution. Oxford University Press, Oxford
Cadée GC (2007) Balanuliths: free-living clusters of the barnacle Balanus crenatus. Palaios 22:680–681
Cook PL (1964) Polyzoa from West Africa. Notes on the genera Hippoporina Neviani, Hippoporella Canu, Cleidochasma Harmer and Hippoporidra Canu & Bassler (Cheilostomata, Ascophora). Bull Br Mus (Nat Hist) (Zool Ser) 12:1–35
Cook PL (1968a) Bryozoa (Polyzoa) from the coasts of tropical West Africa. Atlantide Rep 10:115–262
Cook PL (1968b) Polyzoa from West Africa. The Malacostega—Part 1. Bulletin of The British Museum (Natural History). Zoology 16:115–160
Cook PL (1985) Bryozoa from Ghana. K Mus Midden-Afr Zool Wet 238:1–315
Cuffey RJ and Johnson ME (1997) Bryozoan nodules built around andesite clasts from upper Pliocene of Baja California: palaeoecological implications and closure of the Panama Isthmus. In Johnson ME and Ledesma-Vázques J (eds.): Pliocene Carbonates and related Facies Flanking the Gulf of California, Baja California, Mexico. Geological Society of America. Special Paper 318:111–117
Douglas AE (1994) Symbiotic interactions. Oxford University Press, New York
Ehrenberg K (1931) Über Lebensspuren von Einsiedlerkrebsen. Palaebiologica 4:137–174
Eisele M, Frank N, Wienberg C, Titschack J, Mienis F, Beuck L, Tisnerat-Laborde N, Hebbeln, D (2013) From sediment baffling to mass wasting—Sedimentation patterns on a cold-water coral mound in the Banda Mound Province, Mauritania, submitted to Deep Sea Research II
Flor FD (1970) Biometrische Untersuchungen zur Autökologie ober-kretazischer Bryozoen. Dissertation, Fachbereich für Geowissenschaften, Universität Hamburg
Ginsburg RN, James NP (1974) Holocene carbonate sediments of continental shelves. In: Burke CA, Drake CL (eds) The geology of continental margins. Springer, Berlin, pp 137–155
Glynn PW (1974) Rolling stones among the Scleractinia: mobile coralliths in the Gulf of Panama. Proceedings of the 2nd International Coral Reef Synopsium 2:183–198
Gordon DP (1972) Biological relationships of an intertidal bryozoan population. J Nat Hist 6:503–514
Grischenko AV, Taylor PD, Mawatari SF (2002) A New Cheilostome Bryozoan with Gigantic Zooids from the North-West Pacific. Zool Sci 19:1279–1289
Hanebuth TJJ, Lantzsch H (2008) A Late Quaternary sedimentary shelf system under hyperarid conditions: unravelling climatic, oceanographic and sea-level controls (Golfe d’Arguin, Mauritania, NW Africa). Mar Geol 256:77–89
Hanebuth TJJ, Henrich R (2009) Recurrent decadal-scale dust events over Holocene western Africa and their control on canyon turbidite activity (Mauritania). Quat Sci Rev 28:261–270
Hanebuth TJJ, Mersmeyer H, Kudrass HR, Westphal H (subm. 09/2012) Aeolian to shallow-marine shelf architecture off a major desert since the Late Pleistocene (Northern Mauritania). Geomorphology
Hayton S, Nelson CS, Hood SD (1995) A skeletal assemblage classification system for non-tropical carbonate deposits based on New Zealand Cenozoic limestones. Sediment Geol 100(1–4):123–141
Hoffmann L (1988) The Banc d’Arguin: nature in harmony with man. WWF News, June/July: 10–11
Hayward PJ, Ryland JS (1979) Bristish ascophoran bryozoans. Synopses Br Fauna (New Ser) 14:1–312
IMROP (2013) Atlas maritime des zones vulnérables en Mauritanie—Un appui à la gestion écosystémique et équitable. IMROP, Nouhadibou, Mauritania (in press)
James DW, Foster MS, O’Sullivan J (2006) Bryoliths (Bryozoa) in the Gulf of California. Pac Sci 60:117–124
James NP (1997) The cool-water carbonate depositional realm, in James NP and Clarke JAD (eds.). Cool-Water Carbonates. SEPM Special Publication 56 pp. 1–20
Johnson NM, Officer CB, Opdyke ND, Woodard GD, Zeitler PK, Lindsay EH (1983) Rates of late Cenozoic tectonism in the Vallecito-Fish Creek Basin, western Imperial Valley, California. Geology 11:664–667
Karleskint G, Turner RL, and Small, JW (2009) Introduction to Marine Biology. Brooks/Cole. ISBN: 0495561975
Kellog CW (1976) Gastropods shells: a potential limiting resource for hermit crabs. J Exp Mar Biol Ecol 22:101–111
Kirkpatrick R, Metzelaar J (1922) On an instance of commensalism between hermit crabs and a polyzoon. Proc Zool Soc Lond 1922:983–990
Kidwell SM, Gyllenhaal ED (1998) Symbiosis, competition, and physical disturbance in the growth histories of Pliocene cheilostome bryoliths. Lethaia 31:221–239
Kissling DL (1973) Circumrotatory growth form in Recent and Silurian corals. In: Boardman RS, Cheetham AH, Oliver WA (eds) Animal colonies. Dowden, Hutchinson & Ross, Stroudsburg, pp 43–58
Koopmann B, Lees A, Piessens P, Sarnthein M (1979) Skeletal carbonate sands and wind-derived silty-marls off the Saharan coast: Baie du Lévrier, Arguin Platform, Mauritania: Meteor Forschungsergebnisse. Reihe C 30:15–57
Lagaaij R (1952) The Pliocene Bryozoa of the low countries and their bearing on the Marine Stratigraphy of the North Sea Region. Proefschrift Robert Lagaaij, Ernest van Aelst
Lagaaij R (1963) New additions to the bryozoan fauna of the Gulf of Mexico. Publ Inst Mar Sci, Tex 9:162–236
Lees A, Buller AT (1972) Modern temperate water and warm-water shelf carbonate sediments contrasted. Mar Geol 13:67–73
McKinney FK, Jackson JBC (1991) Bryozoan evolution. University of Chicago Press edition 1991
McLaughlin P (2012) Pseudopagurus granulimanus (Miers, 1881). In: Lemaitre R, McLaughlin P, (2012). World Paguroidea & Lomisoidea database. Accessed through: World Register of Marine Species at http://www.marinespecies.org/
Maranon E, Holligan PM (1999) Photosynthetic parameters of phytoplankton from 50°N to 50°S in the Atlantic Ocean. Mar Ecol Prog Ser 176:191–203
Michel J, Westphal H, Hanebuth TJJ (2009) Sediment partitioning and winnowing in a mixed eolian–marine system (Mauritanian shelf). Geo-Mar Lett 29:221–232
Michel J, Mateu-Vicens G, Westphal H (2011a) Modern heterozoan carbonates from a eutrophic tropical shelf (Mauritania). J Sediment Res 8:641–655
Michel J, Westphal H, Cosel V (2011b) The Mollusk Fauna of soft sediments from the tropical, upwelling- influenced shelf of Mauritania (Northwestern Africa). Palaios 26(7):447–460
Moen FE, Svensen E (2004) Marine fish & invertebrates of Northern Europe. AquaPress. ISBN: 0-9544060-2-8
Moran NA (2006) Symbiosis. Curr Biol 24(20):866–871
Morris PA, Soule DF, Soule JD (1989) Bryozoans, Hermit Crabs, and Gastropods: life strategies can affect the fossil record. Bull South Calif Acad Sci 88(2):45–60
Nelson CS, Keane SL, Head PS (1988a) Non-tropical carbonate deposits on the modern New Zealand shelf. Sediment Geol 60:71–94
Nelson CS, Hyden FM, Keane SL, Leask WL, Gordon DP (1988b) Application of bryozoan zoarial growth- form studies in facies analysis of non-tropical carbonate deposits in New Zealand. Sediment Geol 60:301–322
Ould Mahfoudh M, Dobrovine B, Ould Dedah S (1991) Hydrologique des eaux superficilles du Banc d’Arguin, Département Ressources et Environment et Laboratoire d’Hydrologie et Environment, Centre National de Recherches Oceanographiques et des Peches; http://hdl.handle.net/1834/518
Palmer TJ, Hancock CD (1973) Symbiotic relationships between ectoprocts and gastropods, and between ectoprocts and hermit crabs in the French Jurassic. Palaeontology 16:563–566
Peryt TM (1983) Cotaed grains. Springer, Berlin
Piessens P (1979) Influence relative des sources de production et du transport dans la formation de facies calcaires biogéniques: les sédiments quaternaires de la plate-forme d’Arguin (Mauritanie) et comparaison avec des régions cotières européennes (Irlande et Hébrides). Dissertation, Université Catholique de Louvain
Reguant S, Fernandez J, Rodriguez-Fernandez J, and Serra-Kiel J (1991) Bryozoan biofacies, zoarial forms and sedimentary environments in the Tertiary of Spain. In Bigey FP (ed.): Bryozoaires actuels et fossiles 361–370. Bulletin de la Société Sciences Naturelle d’Ouest France, Memoires, H.S. 1
Reymond CE, Mateu-Vicens G, Westphal H (2013) More than latitude: controlling factors shaping foraminiferal assemblages from the Golfe d’Arguin, Mauritania. Submitted to Estuarine, Coastal and Shelf Science
Rider J, Enrico R (1979) Structural and functional adaptations of mobile anascan ectoproct colonies (ectoproctaliths). In: Larwood EP, Abbott MB (eds) Advances in Bryozoology. Academic, London, pp 297–320
Roff G (2007) Corals on the move: morphological and reproductive strategies of reef flat coralliths. Coral Reefs 27:343–344
Ryland JS (1976) Physiology and ecology of marine bryozoans. In Advances in Marine Biology, 14:285–443. Academic, London
Smith DC (1992) The symbiotic condition. Symbiosis 14:3–15
Smith AM, Key MM Jr, Gordon DP (2006) Skeletal mineralogy of bryozoans: taxonomic and temporal patterns. Earth Sci Rev 78(3–4):287–306
Stachowitsch M (1980) The epibiotic and endolithic species associated with the gastropod shells inhabited by the hermit crabs Paguristes oculatus and Paguristes cuanensis. PSZNI Mar Ecol 1:73–101, 1980. 4386
Taylor PD (1976) Multilamellar growth in two Jurassic cyclostomatous Bryozoa. Palaeontology 19:293–306
Taylor PD (1991) Observations on symbiotic associations of bryozoans and hermit crabs from the Otago Shelf of New Zealand. In Bigey FP (ed.): Bryozoaires actuels et fossils, 487–495. Bullétin de la Societé des Sciences Naturelle de l’Quest de la France, Momoires, H.S. 1
Taylor PD (1994) Evolutionary palaeoecology of symbioses between bryozoans and hermit crabs. Hist Biol 9:147–205
Taylor PD (2005) Bryozoans and palaeoenvironmental interpretation. J Palaeontol Soc India 50(2):1–11
Taylor PD, Monks N (1997) A new cheilostome bryozoan genus pseudoplanktonic on molluscs and algae. Invertebr Biol 116(1):39–51
Taylor PD, Schindler KS (2004) A new Eocene species of the hermit-crab symbiont Hippoporidra (Bryozoa) from the Ocala Limestone of Florida. J Paleontol 78:790–794
Taylor PD, Schembri PJ, Cook PL (1989) Symbiotic associations between hermit crabs and bryozoans from the Otago region, southeastern New Zealand. J Nat Hist 23:1059–1085
Vermaat JE, Beijer JAJ, Gijlstra R, Hootsmans MJM, Philippart CJM (1993) Leaf dynamics and standing stocks of intertidal Zostera noltii Hornem, and Cymodocea nodosa (Ucira) Ascherson on the Banc d’Arguin (Mauritania). Hydrobiology 258:59–72
Williams JD, McDermott JJ (2004) Hermit crab biocoenoses: a worldwide review of the diversity and natural history of hermit crab associates. J Exp Mar Biol Ecol 305:1–128
Winker CD, Kidwell SM (1996) Stratigraphy of marine rift basin: Neogene of the western Salton Trough, California. In Abbott PL and Cooper JD (eds.): Field Conference Guide 1996. Pacific Section AAPG Guide Book 73, Pacific SEPM Book 80:295–336
Winston JE (1982) Marine bryozoans (Ectoprocta) of the Indian River area (Florida). Bull Am Mus Nat Hist 173:99–176
Winston JA, Heimberg BF (1986) Bryozoans from Bali, Lombok, and Komodo. Am Mus Novit 2847(99):1–49
Westphal H, Freiwald A, Hanebuth TJJ, Eisele M, Gürs K, Heindel K, Michel J and Reumont J (2007) Report and preliminary results of Poseidon cruise 346—MACUMA: integrating carbonates, siliciclastics and deep-water reefs for understanding a complex environment, Las Palmas (Spain)–Las Palmas (Spain), 28.12.2006–15.1.2007. Reports of the Dept. of Geosciences, University of Bremen, Germany
Westphal H, Beuck L, Braun S, Freiwald A, Hanebuth TJJ, Hetzinger S, Klicpera A, Kudrass H, Lantzsch H, Lundälv T, Mateu-Vicens G, Preto N, Reumont J, Schilling S, Taviani M and Wienberg C (2013) Report of Cruise Maria S. Merian 16/3—Phaeton—Paleoceanographic and paleo-climatic record on the Mauritanian shelf. Oct. 13—Nov. 20, 2010, Bremerhaven (Allemagne)—Mindelo (Cap Verde). Maria S. Merian-Berichte, Leibniz-ZMT, Bremen, Germany
Westphal H, Halfar J, Freiwald A (2010) Heterozoan carbonates in subtropical to tropical settings in the present and in the past. Int J Earth Sci 99:153–159
Wolff WJ, Van Der Land J, Nienhuis PH, De Wilde PAWJ (1993) The functioning of the ecosystem of the Banc d’Arguin, Mauritania: a review. Hydrobiologia 258:211–222
Acknowledgments
Participants and crew of the “Maria S. Merian” Cruise MSM 16 leg 3 are gratefully acknowledged for providing support and sample background data. Claire Reymond and Julien Michel (both ZMT-Bremen, Germany) are acknowledged for stimulating discussions and comments on an earlier version of this manuscript. Susan M. Kidwell and an anonymous reviewer provided thought-provoking reviews for which we are grateful. The project was funded through the DFG-Research Center/Cluster of Excellence “The Ocean in the Earth System”, project SD-2 and is part of the PhD thesis of André Klicpera.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Suppl. 1
Bivalve shells and bryoliths collected by dredging during the cruise of the R/V Al Awam (IMROP) in late 2012. (JPEG 1224 kb)
Rights and permissions
About this article
Cite this article
Klicpera, A., Taylor, P.D. & Westphal, H. Bryoliths constructed by bryozoans in symbiotic associations with hermit crabs in a tropical heterozoan carbonate system, Golfe d’Arguin, Mauritania. Mar Biodiv 43, 429–444 (2013). https://doi.org/10.1007/s12526-013-0173-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12526-013-0173-4