Abstract
The incidence of colorectal cancer (CRC) is lower in women than in men, and sex steroids can be considered contributing factors because oral contraception usage and estrogen replacement therapy are associated with decreased risk. Conversely, colorectal polyp development in familial adenomatous polyposis (FAP) begins during puberty. The objectives were to evaluate the relationship between the expression of these hormone receptors and adenoma-carcinoma progression, CRC stage and overall survival. We studied 120 A.C. Camargo Cancer Center patients diagnosed with either FAP-associated or spontaneous adenomatous polyps or CRC to determine the immunohistochemical expression levels of estrogen receptor (ER)-α, ER-β and the progesterone and androgen receptors (480 analyses). The ER-β expression levels differed between the groups: the group with FAP polyps had lower ER-β expression than that of the sporadic polyp group. With transformation of the sporadic polyps to cancer, there was a considerable decrease in ER-β expression (from 90% with strong expression to 80% with absent or weak expression) (p < 0.001). The ER-β expression was lower in T3/T4 tumors than in T1/T2 tumors (p = 0.015). The 5-year overall survival of CRC patients positively expressing ER-β exceeded that of patients without detectable expression levels (74.8% vs. 44.3%, respectively; p = 0.035). There was no significant expression of the androgen or progesterone receptor or ER-α among the groups. Differences in ER-β expression represent a potential mechanism through which estrogen might alter the susceptibility to colon cancer, thereby confirming the possibility of a protective role of estrogen against colorectal carcinogenesis.
Similar content being viewed by others
Introduction
Colorectal cancer (CRC) is a pathology with high incidence and mortality. The highest incidence rates are in Australia, New Zealand, Europe and North America, and the lowest rates are found in Africa and South-Central Asia. These geographic differences appear to be attributable to differences in dietary and environmental factors that are imposed on a background of genetically determined susceptibility [1].
Familial adenomatous polyposis (FAP) is an autosomal dominant disease characterized by the development of hundreds to thousands of adenomatous polyps in the colon and rectum, and it accounts for 1% of CRC cases. These polyps emerge in the second decade of life, which is during puberty, and increase in size and number during adolescence, which is at the peak of sex steroid production; these findings suggest that sex steroids may act as cofactors in the development of polyps and FAP-related colorectal carcinogenesis [2,3,4,5,6]. Conversely, the incidence of CRC is lower in women than men. Although the cause for this difference is not known, sex steroids may be considered a contributing factor because high parity, early age at first pregnancy, use of oral contraceptives and estrogen replacement therapy are associated with a decreased risk of sporadic CRC [7].
Giardiello et al. [8] described a patient from the placebo group of a 4-year primary chemoprevention clinical trial of sulindac who developed adenomatous polyps that were later eradicated following the administration of oral contraceptives, suggesting a likely interruption of the adenoma-carcinoma sequence in an FAP patient. However, the possibility of a functional interaction with sex hormone receptors in an APC gene mutation carrier has not been thoroughly investigated.
According to the Women’s Health Initiative (WHI) [9, 10], combined (estrogen and progesterone) hormone replacement therapy (HRT) is associated with a decreased risk of CRC (hazard ratio (HR), 0.56), and epidemiological studies have convincingly demonstrated this decreased risk even for the use of estrogen alone [11,12,13,14]. Grodstein et al. [15] showed that, in addition to having a lower CRC risk, women who used estrogen had a lower risk of large colorectal adenomas (≥1 cm) compared to that for women who had never used the hormone (HR, 0.74).
Previous studies were also conducted to analyze the presence of hormone receptors in the mucosal epithelium of the normal colon and that of sporadic CRC [16,17,18,19,20,21,22]; these studies suggested that the high titer of ER-β observed predominantly in normal tissue is most likely the dominant transcriptional mediator of the estrogen effect that links this effect with a better prognosis [7, 23]. However, no studies have been conducted to evaluate the immunohistochemical expression of estrogen receptor (ER)–α, ER-β and the progesterone and androgen receptors in FAP-associated and sporadic polyps and invasive carcinomas.
The objective of this study was to evaluate the relationship between the expression of these hormone receptors and adenoma-carcinoma progression, CRC clinical and pathological stage and overall survival.
Materials and Methods
Study Subjects
This was a descriptive and analytical cohort study investigating the immunohistochemical expression of ER-α, ER-β and the progesterone and androgen receptors in patients with adenomatous polyps or invasive CRC, with or without a clinical FAP diagnosis. The study was approved by the ethics committee (1453/10).
The sample included all patients with FAP syndrome with cancer between the years 1993–2013 who were treated at the A.C. Camargo Cancer Center and had biological samples stored in the tissue bank; this population is rare because most of these patients are syndromic and underwent preventive CRC surgery with total removal of the colon; these cases determined the sample size. The other groups of patients with adenomatous polyps (FAP and sporadic) and sporadic cancer were defined as randomly treated during the same period. Thus, we evaluated 480 immunohistochemistry exams for ER-α and ER-β, progesterone and androgen receptors for 120 patients, who equally distributed between the following groups:
-
(A)
FAP patients with a histological diagnosis of adenomatous polyps with low-grade dysplasia;
-
(B)
FAP patients with a histological diagnosis of invasive colorectal adenocarcinoma;
-
(C)
patients with sporadic adenomatous polyps with low-grade dysplasia and no clinical diagnosis of FAP or the associated Lynch syndrome; and (D) patients with sporadic invasive colorectal adenocarcinoma and no clinical diagnosis of FAP or the associated Lynch syndrome.
The follow-up routine for patients with colorectal tumors treated at the institution included a clinical visit every three months during the first two years; the routine included laboratory tests, tumor marker evaluation, chest X-ray and abdominal ultrasound or cross-sectional imaging (tomography), alternately. From the third to the fifth year, this routine examination was performed every six months; afterward, this procedure was conducted annually. Proctosigmoidoscopy was repeated annually in FAP patients who underwent surgery.
Antibodies, Construction and Immunohistochemical Analysis
The immunohistochemistry was performed using two slides of the tissue microarray (TMA) blocks for each antibody. TMA sections from the array block were stained with the following primary antibodies: anti-ER (clone SPL Dako), anti-ER-β (clone 14C8, Genetex), anti-progesterone receptor (clone PgR 636, Dako) and anti-androgen receptor (clone AR 441, Neomarkes). Four-micrometer-thick sections were cut from the array block and were deparaffinized with xylene and dehydrated through a graded alcohol series. Microwave antigen retrieval was used for all of the antibodies: the slides were placed in 10 mM citrate buffer (pH 6) for 15 min. A standard peroxidase-conjugated streptavidin-biotin method was used to detect the staining reaction (Advance, HRP Link, DAKO, Carpinteria, USA). External positive control tissues included breast and prostate samples from normal tissue that were positive for the studied antibodies. For negative controls, the primary antibodies were omitted and substituted with normal serum antigen.
A scoring system was applied based on the intensity as recommended by Rüschoff et al. [24] using an optical microscope with objective magnification. The intensity was classified as strong when nuclear staining was observed at 5X magnification, moderate for staining observed at 10X and weak for staining that was only observed at 40X (Fig. 1).
The scoring was conducted independently by 2 pathologists: M.D.B. and M.M. The preparation for the immunohistochemistry and the scoring were conducted in a manner blinded to the other case characteristics. Discordant results were resolved by an additional joint review of the relevant sample.
Statistical Analysis
For statistical analysis, the software Statistical Package for Social Science (SPSS) version 20 was used. Survival curves were estimated using the Kaplan-Meier product-limit method, and significant differences between the survival curves were determined using the log-rank test. All variables associated with survival with p < 0.20 in univariate analysis were included in multivariate analysis using a Cox proportional hazard regression model. Correlation coefficients between all findings were estimated using Pearson’s chi-squared test or a chi-squared two-tailed corrected test (Fisher’s exact test) as indicated. The results were considered statistically significant at p < 0.05.
Results
Clinical and Histopathological Results
For the 120 assessed patients, the median age was 49 years (19–80 years), 48% were males, and 52% were females. For patients with invasive cancer, the most frequent clinical complaint was bleeding (32% of cases). The most frequent site of primary tumor was the colon (70%), followed by the rectum (30%). Moderately differentiated adenocarcinoma represented 56.7% of the cases. In FAP patients, the median age for disease diagnosis was 31 years, and that for cancer diagnosis was 38 years. The median follow-up time of cancer patients in the study was 61 (4–239) months. Table 1 shows clinical and pathological profiles according to the expression of ER-β.
Immunohistochemical Results
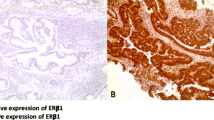
ER-β expression was significantly different between groups (Fig. 2). Compared with the sporadic polyp group, the FAP polyp group had lower expression levels of these receptors. In the sporadic group, CRC transformation was accompanied by a considerable decrease in the high ER-β expression observed in sporadic polyps, where 90% of the samples strongly expressed ER-β and 100% exhibited some degree of expression. This finding contrasts with that for the sporadic cancer group, where 80% exhibited a low or complete lack of ER-β expression (40% negative, 40% weak expression, 20% moderate expression and 0% strong expression; p < 0.001).
Patients with T3/T4 tumors demonstrated a significant absence in ER-β expression compared to that of patients with T1/T2 tumors (T1/T2: 5.8% negative, 94.2% positive vs. T3/T4: 37.2% negative, 62.8% positive; p = 0.015) (Fig. 3). There was no significant difference in expression with regard to lymph node status (N0: 25% negative, 75% positive vs. N+: 33.3% negative, 66.7% positive; p = 0.483) and metastatic status (M0: 30.2% negative, 69.8% positive vs. M+: 14.2% negative, 85.8% positive; p = 0.380).
There was no expression of androgen and progesterone receptors among the groups, as well as no significant expression of ER-α, which was present in only 3 individuals of the sporadic polyps group.
Overall Survival Results
The 5-year overall survival of all patients with CRC was 65.3% (Table 2). The highest survival rate was observed in patients showing some degree of ER-β expression compared to that in patients with no expression (74.8% vs. 44.3%, p = 0.035) (Fig. 4). Univariate analysis showed significantly better overall survival for the following characteristics: positive for ER-β, younger than 50 years of age, absence of lymphatic embolization and Clinical Stage I/II (Table 2).
Multivariate analysis was performed for the variables clinical stage, lymphatic embolization, expression of ER-β and the presence of FAP syndrome to determine the characteristics with a greater risk of death (p < 0.2, log rank). From the multivariate Cox regression model, clinical stage III/IV (HR 2.9, 95% CI = 1.15–7.43, p = 0.023), presence of lymphatic embolization (HR 2.6, 95% CI = 1.03–6.78, p = 0.043) and absence of ER expression (HR 3, 95% CI = 1.24–7.46, p = 0.015) showed a significantly higher risk of death (Table 3).
Discussion
In this study, analysis of the expression of the studied hormonal receptors (androgen, progesterone and estrogen α and β) showed dominant expression levels of ER-β in adenomatous polyps, and these levels varied in sporadic CRC and FAP patients. This fact, associated with the underexpression or absence of expression of the other sex hormone receptors, supports the hypothesis that estrogen signaling through ER-β plays a role in colorectal carcinogenesis. Biological evidence supports this association and has been observed in other studies, where an overexpression of ER-β was detected in normal colonic mucosa compared to that in sporadic CRC [15,16,17,18,19,20,21]; these results also apply to our findings in adenomas.
We observed lower ER-β expression levels in FAP polyps than in sporadic polyps. This finding, when associated with studies in an experimental model of APC-deficient mice, which had a loss of ER expression in colorectal tumors [25], may suggest that these receptors act as inhibitory modifiers of APC-dependent colon tumorigenesis. Cho et al. [26] demonstrated that oophorectomy resulted in a significant increase in the number of intestinal adenomas in mice using an animal model of APC-dependent CRC. Currently, little is known about estrogen signaling in FAP patients, but these findings favor a new line of investigation regarding whether estrogen actions can contribute to or cooperate with the tumor suppressive function of the APC gene. A double-blind, randomized, 4-year primary chemoprevention study investigated sulindac usage in genotypically affected individuals (APC gene mutation) who were initially not phenotypically affected; the results for the placebo group showed complete eradication of polyps with the occasional administration of an oral contraceptive to a patient who had developed polyps [8]. That same study demonstrated no changes in prostaglandin levels in the colonic mucosa, which makes anti-inflammatory drug administration an unlikely cause of this effect. Flexible proctosigmoidoscopy was performed for follow-up every four months for 48 months, and a recurrence and increase in the prevalence of polyps was observed after suspension of the oral contraceptive.
We observed significantly lower ER-β expression levels in the CRC groups than in the sporadic polyp group, with an increased loss of expression related to more advanced T stages (TNM [27]) (T3/T4 tumors vs. T1/T2 tumors). This finding might explain why we did not observe lower expression in the FAP cancer group compared to that in the FAP polyp group because the FAP cancer group was 56.7% T1/T2 patients, while the sporadic cancer group mainly had T3/T4 patients (Fig. 3). Other studies [28, 29] also reported a relationship between the loss of ER-β expression in CRC with Dukes’ advanced stage and a higher degree of tumor differentiation. Therefore, in the groups with polyps (FAP and sporadic), we only included patients with low-grade dysplasia because polyps with high-grade dysplasia could have a similar behavior to the cancer groups. These data indicate that the presence of ER-β has an important role in colorectal carcinogenesis as a marker of tumor progression and could explain the protective effect of estrogen against the development of CRC, as well as the worsened prognosis for patients with tumors lacking ER-β expression.
Among the numerous types of cancer, breast cancer has a model of hormonal carcinogenesis that is the most thoroughly described and understood, which has allowed the discovery of important prognostic markers and the establishment of anti-hormone therapies [30]. Subsequently, there has been an effort to understand the mechanism through which estrogen and other steroid hormones could act in carcinogenesis pathways. Estrogens control a wide range of vital physiological processes that are chiefly mediated by the activities of ER-α and ER-β, which are closely related to the nuclear receptors of ligand-dependent transcription factors and are at the promoters of estrogen-sensitive genes [31,32,33]. ERs may act as transcription cofactors through their interaction with other DNA-binding proteins, and they also mediate non-specific estrogen-induced physiological effects through genomic mechanisms of action [34, 35].
It has been reported that the administration of oral estrogen during HRT has reduced the risk of CRC development by approximately 44% and lessens the recurrence of neoplastic polyps after surgical removal of colon tumors [9, 10, 36, 37]. Thus, to evaluate one effect of the presence of ER-β expression, we assessed overall survival in terms of certain clinical pathological variables and the expression of ER-β. We observed a better overall survival in individuals with CRC associated with positive expression of ER-β (p = 0.035), and the loss of ER-β expression remained an independent prognostic factor of a higher risk of death (HR 3, 95% CI = 1.24–7.46, p = 0.015) (Tables 2 and 3).
In summary, our study suggests that ER-β expression might mediate the chemopreventive effects of estrogens in the development of polyps and CRC. Based on these findings, new questions arise, and further studies might be useful in the search for chemopreventive treatments for patients with FAP. Moreover, these data also suggest the need for greater surveillance and screening of polyps and CRC in individuals who receive anti-estrogen hormone treatment (tamoxifen/anastrozole) due to other diseases, as well as in patients who have undergone oophorectomy, are nulliparous, are in early menopause or have risk of hereditary breast or CRC syndrome. As in breast cancer, in CRC, the level of hormone receptor expression, ER-β in particular, is related to patient prognosis, supporting the role of ER-β as an important marker of tumor progression and a possible target for chemoprevention in patients at risk for CRC.
References
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D (2011) Global cancer statistics. CA Cancer J Clin 61:69–90
Gebert JF, Dupon C, Kadmon M, Hahn M, Herfarth C, von Knebel DM, Schackert HK (1999) Combined molecular and clinical approaches for the identification of families with familial adenomatous polyposis coli. Ann Surg 229:350–361. doi:10.1097/00000658-199903000-00008
Goss KH, Groden J (2000) Biology of the adenomatous polyposis coli tumor suppressor. J Clin Oncol 18:1967–1979. doi:10.1200/JCO.2000.18.9.1967
Wong N, Lasko D, Rabelo R, Pinsky L, Gordon PH, Foulkes W (2001) Genetic counseling and interpretation of genetic tests in familial adenomatous polyposis and hereditary nonpolyposis colorectal cancer. Dis Colon rectum 44:271–279. doi:10.1007/BF02234304
Vasen HF, Möslein G, Alonso A, Aretz S, Bernstein I, Bertario L, Blanco I, Bülow S, Burn J, Capella G, Colas C, Engel C, Frayling I, Friedl W, Hes FJ, Hodgson S, Järvinen H, Mecklin JP, Møller P, Myrhøi T, Nagengast FM, Parc Y, Phillips R, Clark SK, de Leon MP, Renkonen-Sinisalo L, Sampson JR, Stormorken A, Tejpar S, Thomas HJ, Wijnen J (2008) Guidelines for the clinical management of familial adenomatous polyposis (FAP). Gut 57:704–713. doi:10.1136/gut.2007.136127
Nieuwenhuis MH, Vasen HF (2007) Correlations between mutation site in APC and phenotype of familial adenomatous polyposis (FAP): a review of the literature. Crit Rev Oncol Hematol 61:153–161. doi:10.1016/j.critrevonc.2006.07.004
Kennelly R, Kavanagh DO, Hogan AM, Winter DC (2008) Oestrogen and the colon: potential mechanisms for cancer prevention. Lancet Oncol 9:385–391. doi:10.1016/S1470-2045(08)70100-1
Giardiello FM, Hylind LM, Trimbath JD, Hamilton SR, Romans KE, Cruz-Correa M, Corretti MC, Offerhaus JA, Yang VW (2005) Oral contraceptives and polyp regression in familial adenomatous polyposis. Gastroenterology 128:1077–1080
Chlebowski RT, Wactawski-Wende J, Ritenbaugh C, Hubbell FA, Ascensao J, Rodabough RJ, Rosenberg CA, Taylor VM, Harris R, Chen C, Adams-Campbell LL, White E, Investigators W's HI (2004) Estrogen plus progestin and colorectal cancer in postmenopausal women. N Engl J Med 350:991–1004. doi:10.1056/NEJMoa032071
Simon MS, Chlebowski RT, Wactawski-Wende J, Johnson KC, Muskovitz A, Kato I, Young A, Hubbell FA, Prentice RL (2012) Estrogen plus progestin and colorectal cancer incidence and mortality. J Clin Oncol 30:3983–3990. doi:10.1200/JCO.2012.42.7732
Anderson GL, Limacher M, Assaf AR, Bassford T, Beresford SA, Black H, Bonds D, Brunner R, Brzyski R, Caan B, Chlebowski R, Curb D, Gass M, Hays J, Heiss G, Hendrix S, Howard BV, Hsia J, Hubbell A, Jackson R, Johnson KC, Judd H, Kotchen JM, Kuller L, LaCroix AZ, Lane D, Langer RD, Lasser N, Lewis CE, Manson J, Margolis K, Ockene J, O’Sullivan MJ, Phillips L, Prentice RL, Ritenbaugh C, Robbins J, Rossouw JE, Sarto G, Stefanick ML (2004) Effects of conjugated equine estrogen in postmenopausal women with hysterectomy: the Women's Health Initiative randomized controlled trial. JAMA 291:1701–1712. doi:10.1001/jama.291.14.1701
Rennert G, Rennert HS, Pinchev M, Lavie O, Gruber SB (2009) Use of hormone replacement therapy and the risk of colorectal cancer. J Clin Oncol 27:4542–4547. doi:10.1200/JCO.2009.22.0764
Green J, Czanner G, Reeves G, Watson J, Wise L, Roddam A, Beral V (2012) Menopausal hormone therapy and risk of gastrointestinal cancer: nested case-control study within a prospective cohort, and meta-analysis. Int J Cancer 130:2387–2396. doi:10.1002/ijc.26236
Lin KJ, Cheung WY, Lai JY, Giovannucci EL (2012) The effect of estrogen vs. combined estrogen-progestogen therapy on the risk of colorectal cancer. Int J Cancer 130:419–430. doi:10.1002/ijc.26026
Grodstein F, Newcomb PA, Stampfer MJ (1999) Postmenopausal hormone therapy and the risk of colorectal cancer: a review and meta-analysis. Am J Med 106:574–582. doi:10.1016/S0002-9343(99)00063-7
Kouzmenko AP, Takeyama K, Kawasaki Y, Akiyama T, Kato S (2008) Ligand-dependent interaction between estrogen receptor alpha and adenomatous polyposis coli. Genes Cells 13:723–730. doi:10.1111/j.1365-2443.2008.01200.x
Dawson PM, Shousha S, Blair SD, Carter GD, Jones J, Alaghband-Zadeh J, Theodorou NA (1990) Oestrogen receptors in colorectal carcinoma. J Clin Pathol 43:149–151. doi:10.1136/jcp.43.2.149
Hendrickse CW, Jones CE, Donovan IA, Neoptolemos JP, Baker PR (1993) Oestrogen and progesterone receptors in colorectal cancer and human colonic cancer cell lines. Br J Surg 80:636–640. doi:10.1002/bjs.1800800531
Di Leo A, Messa C, Russo F, Misciagna G, Guerra V, Taveri R, Leo S (1994) Prognostic value of cytosolic estrogen receptors in human colorectal carcinoma and surrounding mucosa. Preliminary results. Dig Dis Sci 39:2038–2042. doi:10.1007/BF02088144
Meggouh F, Lointier P, Pezet D, Saez S (1991) Status of sex steroid hormone receptors in large bowel cancer. Cancer 67:1964–1970. doi:10.1002/1097-0142(19910401)67:7<1964::AID-CNCR2820670724>3.0.CO;2-S
Slattery ML, Sweeney C, Murtaugh M, Ma KN, Caan BJ, Potter JD, Wolff R (2006) Associations between vitamin D, vitamin D receptor gene and the androgen receptor gene with colon and rectal cancer. Int J Cancer 118:3140–3146. doi:10.1002/ijc.21791
Slattery ML, Sweeney C, Murtaugh M, Ma KN, Wolff RK, Potter JD, Caan BJ, Samowitz W (2005) Associations between ERalpha, ERbeta, and AR genotypes and colon and rectal cancer. Cancer Epidemiol Biomark Prev 14:2936–2942. doi:10.1158/1055-9965.EPI-05-0514
Mostafaie N, Kállay E, Sauerzapf E, Bonner E, Kriwanek S, Cross HS, Huber KR, Krugluger W (2009) Correlated downregulation of estrogen receptor beta and the circadian clock gene Per1 in human colorectal cancer. Mol Carcinog 48:642–647. doi:10.1002/mc.20510
Rüschoff J, Dietel M, Baretton G, Arbogast S, Walch A, Monges G, Chenard MP, Penault-Llorca F, Nagelmeier I, Schlake W, Höfler H, Kreipe HH (2010) HER2 diagnostics in gastric cancer-guideline validation and development of standardized immunohistochemical testing. Virchows Arch 457:299–307. doi:10.1007/s00428-010-0952-2
Foley EF, Jazaeri AA, Shupnik MA, Jazaeri O, Rice LW (2000) Selective loss of estrogen receptor beta in malignant human colon. Cancer Res 60:245–248
Cho NL, Javid SH, Carothers AM, Redston M, Bertagnolli MM (2007) Estrogen receptors α and β are inhibitory modifiers of Apc-dependent tumorigenesis in the proximal colon of min/+ mice. Cancer Res 67:2366–2372. doi:10.1158/0008-5472.CAN-06-3026
Edge SB, Byrd DR, Compton CC, Fritz AG, Greene FL, Trotti A (2009) American joint committee on cancer staging manual, 7th edn. Springer, New York
Jassam N, Bell SM, Speirs V, Quirke P (2005) Loss of expression of oestrogen receptor beta in colon cancer and its association with dukes’ staging. Oncol Rep 14:17–21
Konstantinopoulos PA, Kominea A, Vandoros G, Sykiotis GP, Andricopoulos P, Varakis I, Sotiropoulou-Bonikou G, Papavassiliou AG (2003) Oestrogen receptor beta (ERbeta) is abundantly expressed in normal colonic mucosa, but declines in colon adenocarcinoma paralleling the tumour’s dedifferentiation. Eur J Cancer 39:1251–1258. doi:10.1016/S0959-8049(03)00239-9
Jordan VC (2007) New insights into the metabolism of tamoxifen and its role in the treatment and prevention of breast cancer. Steroids 72:829–842. doi:10.1016/j.steroids.2007.07.009
Kato S, Sato T, Watanabe T, Takemasa S, Masuhiro Y, Ohtake F, Matsumoto T (2005) Function of nuclear sex hormone receptors in gene regulation. Cancer Chemother Pharmacol 56:4–9. doi:10.1007/s00280-005-0102-8
Ohtake F, Takeyama K, Matsumoto T, Kitagawa H, Yamamoto Y, Nohara K, Tohyama C, Krust A, Mimura J, Chambon P, Yanagisawa J, Fujii-Kuriyama Y, Kato S (2003) Modulation of oestrogen receptor signalling by association with the activated dioxin receptor. Nature 423:545–550. doi:10.1038/nature01606
Métivier R, Reid G, Gannon F (2006) Transcription in four dimensions: nuclear receptor-directed initiation of gene expression. EMBO Rep 7:161–167. doi:10.1038/sj.embor.7400626
Björnström L, Sjöberg M (2005) Mechanisms of estrogen receptor signaling: convergence of genomic and nongenomic actions on target genes. Mol Endocrinol 19:833–842. doi:10.1210/me.2004-0486
Vasudevan N, Pfaff DW (2007) Membrane-initiated actions of estrogens in neuroendocrinology: emerging principles. Endocr Rev 28:1–19. doi:10.1210/er.2005-0021
Nanda K, Bastian LA, Hasselblad V, Simel DL (1999) Hormone replacement therapy and the risk of colorectal cancer: a meta-analysis. Obstet Gynecol 93:880–888. doi:10.1016/S0029-7844(98)00424-4
Slattery ML, Potter JD, Curtin K, Edwards S, Ma KN, Anderson K, Schaffer D, Samowitz WS (2001) Estrogens reduce and withdrawal of estrogens increase risk of microsatellite instability-positive colon cancer. Cancer Res 61:126–130
Acknowledgments
We thank Dr. Renata Maymi Takahashi, Bruna Catin and Mariana Petaccia de Macedo, who was also a coworker in this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Grants
This study was supported by grants from the State of São Paulo Research Foundation (FAPESP - 1453/10 - http://www.fapesp.br/en). The funders had no role in the study design, data collection and analysis, decision to publish or preparation of the manuscript.
Conflict of Interest
The authors declare that there is no conflict of interest.
Rights and permissions
About this article
Cite this article
Stevanato Filho, P.R., Aguiar Júnior, S., Begnami, M.D. et al. Estrogen Receptor β as a Prognostic Marker of Tumor Progression in Colorectal Cancer with Familial Adenomatous Polyposis and Sporadic Polyps. Pathol. Oncol. Res. 24, 533–540 (2018). https://doi.org/10.1007/s12253-017-0268-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-017-0268-5