Abstract
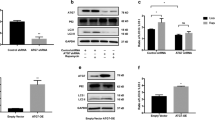
In order to identify the cellular factors involved in human melanogenesis, we carried out shRNA-mediated loss-of-function screening in conjunction with induction of melanogenesis by 1-oleoyl-2-acetyl-glycerol (OAG) in human melanoma cells using biochemical and visual assays. Gene targets of the shRNAs (that caused loss of OAG-induced melanogenesis) and their pathways, as determined by bioinformatics, revealed involvement of proteins that regulate cell stress response, mitochondrial functions, proliferation, and apoptosis. We demonstrate, for the first time, that the mitochondrial stress chaperone mortalin is crucial for melanogenesis. Upregulation of mortalin was closely associated with melanogenesis in in vitro cell-based assays and clinical samples of keloids with hyperpigmentation. Furthermore, its knockdown resulted in compromised melanogenesis. The data proposed mortalin as an important protein that may be targeted to manipulate pigmentation for cosmetic and related disease therapeutics.







Similar content being viewed by others
References
Abdel-Malek ZA, Kadekaro AL, Swope VB (2010) Stepping up melanocytes to the challenge of UV exposure. Pigment Cell Melanoma Res 23:171–186
Bagchi D, Sen CK, Bagchi M, Atalay M (2004) Anti-angiogenic, antioxidant, and anti-carcinogenic properties of a novel anthocyanin-rich berry extract formula. Biochemistry (Mosc) 69:75–80
Bellei B, Maresca V, Flori E, Pitisci A, Larue L, Picardo M (2010) p38 regulates pigmentation via proteasomal degradation of tyrosinase. J Biol Chem 285:7288–7299
Benathan M (1997) Opposite regulation of tyrosinase and glutathione peroxidase by intracellular thiols in human melanoma cells. Arch Dermatol Res 289:341–346
Bottinger L, Oeljeklaus S, Guiard B, Rospert S, Warscheid B, Becker T (2015) Mitochondrial heat shock protein (Hsp) 70 and Hsp10 cooperate in the formation of Hsp60 complexes. J Biol Chem 290:11611–11622
Cappello F, Marino Gammazza A, Palumbo Piccionello A, Campanella C, Pace A, Conway de Macario E, Macario AJ (2014) Hsp60 chaperonopathies and chaperonotherapy: targets and agents. Expert Opin Ther Targets 18:185–208
Chiasserini D, Tozzi A, de Iure A, Tantucci M, Susta F, Orvietani PL, Koya K, Binaglia L, Calabresi P (2011) Mortalin inhibition in experimental Parkinson’s disease. Mov Disord 26:1639–1647
Choi YK, Rho YK, Yoo KH, Lim YY, Li K, Kim BJ, Seo SJ, Kim MN, Hong CK, Kim DS (2010) Effects of vitamin C vs. multivitamin on melanogenesis: comparative study in vitro and in vivo. Int J Dermatol 49:218–226
Dadzie OE, Jablonski NG, Mahalingam M, Dupuy A, Petit A (2014) Skin cancer, photoprotection, and skin of color. J Am Acad Dermatol 71:586
Daiwile AP, Naoghare PK, Giripunje MD, Rao PD, Ghosh TK, Krishnamurthi K, Alimba CG, Sivanesan S (2015) Correlation of melanophore index with a battery of functional genomic stress indicators for measurement of environmental stress in aquatic ecosystem. Environ Toxicol Pharmacol 39:489–495
Daniele T, Hurbain I, Vago R, Casari G, Raposo G, Tacchetti C, Schiaffino MV (2014) Mitochondria and melanosomes establish physical contacts modulated by Mfn2 and involved in organelle biogenesis. Curr Biol 24:393–403
del Cerro S, Merino S, Martinez-de la Puente J, Lobato E, Ruiz-de-Castaneda R, Rivero-de Aguilar J, Martinez J, Morales J, Tomas G, Moreno J (2010) Carotenoid-based plumage colouration is associated with blood parasite richness and stress protein levels in blue tits (Cyanistes caeruleus). Oecologia 162:825–835
Denat L, Kadekaro AL, Marrot L, Leachman SA, Abdel-Malek ZA (2014) Melanocytes as instigators and victims of oxidative stress. J Invest Dermatol 134:1512–1518
Deocaris CC, Kaul SC, Wadhwa R (2009) The versatile stress protein mortalin as a chaperone therapeutic agent. Protein Pept Lett 16:517–529
Deocaris CC, Lu WJ, Kaul SC, Wadhwa R (2013) Druggability of mortalin for cancer and neuro-degenerative disorders. Curr Pharm Des 19:418–429
Divi RL, Haverkos KJ, Humsi JA, Shockley ME, Thamire C, Nagashima K, Olivero OA, Poirier MC (2007) Morphological and molecular course of mitochondrial pathology in cultured human cells exposed long-term to Zidovudine. Environ Mol Mutagen 48:179–189
Double KL (2006) Functional effects of neuromelanin and synthetic melanin in model systems. J Neural Transm 113:751–756
Franceschini A, Szklarczyk D, Frankild S, Kuhn M, Simonovic M, Roth A, Lin J, Minguez P, Bork P, von Mering C, Jensen LJ (2013) STRING v9.1: protein-protein interaction networks, with increased coverage and integration. Nucleic Acids Res 41:D808–815
Friedmann PS, Wren FE, Matthews JN (1990) Ultraviolet stimulated melanogenesis by human melanocytes is augmented by di-acyl glycerol but not TPA. J Cell Physiol 142:334–341
Gaweda-Walerych K, Zekanowski C (2013) Integrated pathways of parkin control over mitochondrial maintenance—relevance to Parkinson’s disease pathogenesis. Acta Neurobiol Exp (Wars) 73:199–224
Gerlach M, Double KL, Youdim MB, Riederer P (2006) Potential sources of increased iron in the substantia nigra of parkinsonian patients. J Neural Transm Supp l133-142
Giuliano S, Ohanna M, Ballotti R, Bertolotto C (2011) Advances in melanoma senescence and potential clinical application. Pigment Cell Melanoma Res 24:295–308
Gordon PR, Gilchrest BA (1989) Human melanogenesis is stimulated by diacylglycerol. J Invest Dermatol 93:700–702
Guercio-Hauer C, Macfarlane DF, Deleo VA (1994) Photodamage, photoaging and photoprotection of the skin. Am Fam Physician 50(327–332):334
Gupta S, Knowlton AA (2002) Cytosolic heat shock protein 60, hypoxia, and apoptosis. Circulation 106:2727–2733
Hu DN, Wakamatsu K, Ito S, McCormick SA (2009) Comparison of eumelanin and pheomelanin content between cultured uveal melanoma cells and normal uveal melanocytes. Melanoma Res 19:75–79
Jablonski NG (2012) The evolution of human skin colouration and its relevance to health in the modern world. J R Coll Physicians Edinb 42:58–63
Jablonski NG, Chaplin G (2000) The evolution of human skin coloration. J Hum Evol 39:57–106
Jablonski NG, Chaplin G (2010) Colloquium paper: human skin pigmentation as an adaptation to UV radiation. Proc Natl Acad Sci U S A 107(Suppl 2):8962–8968
Jimenez-Cervantes C, Martinez-Esparza M, Perez C, Daum N, Solano F, Garcia-Borron JC (2001) Inhibition of melanogenesis in response to oxidative stress: transient downregulation of melanocyte differentiation markers and possible involvement of microphthalmia transcription factor. J Cell Sci 114:2335–2344
Jin J, Li GJ, Davis J, Zhu D, Wang Y, Pan C, Zhang J (2007) Identification of novel proteins associated with both alpha-synuclein and DJ-1. Mol Cell Proteomics 6:845–859
Kaul SC, Deocaris CC, Wadhwa R (2007) Three faces of mortalin: a housekeeper, guardian and killer. Exp Gerontol 42:263–274
Kim HE, Lee SG (2013) Induction of ATP synthase beta by H2O2 induces melanogenesis by activating PAH and cAMP/CREB/MITF signaling in melanoma cells. Int J Biochem Cell Biol 45:1217–1222
Kim TJ, Cho MK, Lee JS, Whang KU, Jin SY, Hoshino T (2003) The expression of melanogenic proteins in Korean skin after ultraviolet irradiation. J Dermatol 30:665–672
Kim ES, Shin JH, Seok SH, Kim JB, Chang H, Park SJ, Jo YK, Choi ES, Park JS, Yeom MH, Lim CS, Cho DH (2013) Autophagy mediates anti-melanogenic activity of 3′-ODI in B16F1 melanoma cells. Biochem Biophys Res Commun 442:165–170
Kostyuk VA, Potapovich AI, Cesareo E, Brescia S, Guerra L, Valacchi G, Pecorelli A, Deeva IB, Raskovic D, De Luca C, Pastore S, Korkina LG (2010) Dysfunction of glutathione S-transferase leads to excess 4-hydroxy-2-nonenal and H(2)O(2) and impaired cytokine pattern in cultured keratinocytes and blood of vitiligo patients. Antioxid Redox Signal 13:607–620
Lee JM (1998) Inhibition of p53-dependent apoptosis by the KIT tyrosine kinase: regulation of mitochondrial permeability transition and reactive oxygen species generation. Oncogene 17:1653–1662
Lee B, Ahn Y, Kang SM, Park Y, Jeon YJ, Rho JM, Kim SW (2015) Stoichiometric expression of mtHsp40 and mtHsp70 modulates mitochondrial morphology and cristae structure via Opa1L cleavage. Mol Biol Cell 26:2156–2167
Lu WJ, Lee NP, Kaul SC, Lan F, Poon RT, Wadhwa R, Luk JM (2011) Induction of mutant p53-dependent apoptosis in human hepatocellular carcinoma by targeting stress protein mortalin. Int J Cancer 129:1806–1814
Maresca V, Flori E, Briganti S, Mastrofrancesco A, Fabbri C, Mileo AM, Paggi MG, Picardo M (2008) Correlation between melanogenic and catalase activity in in vitro human melanocytes: a synergic strategy against oxidative stress. Pigment Cell Melanoma Res 21:200–205
Marrot L, Meunier JR (2008) Skin DNA photodamage and its biological consequences. J Am Acad Dermatol 58:S139–148
Marrot L, Belaidi JP, Chaubo C, Meunier JR, Perez P, Agapakis-Causse C (1998) An in vitro strategy to evaluate the phototoxicity of solar UV at the molecular and cellular level: application to photoprotection assessment. Eur J Dermatol 8:403–412
Miller AJ, Tsao H (2010) New insights into pigmentary pathways and skin cancer. Br J Dermatol 162:22–28
Nasti TH, Timares L (2015) MC1R, eumelanin and pheomelanin: their role in determining the susceptibility to skin cancer. Photochem Photobiol 91:188–200
Nishiura H, Kumagai J, Kashino G, Okada T, Tano K, Watanabe M (2012) The bystander effect is a novel mechanism of UVA-induced melanogenesis. Photochem Photobiol 88:389–397
Pan T, Zhu J, Hwu WJ, Jankovic J (2012) The role of alpha-synuclein in melanin synthesis in melanoma and dopaminergic neuronal cells. PLoS One 7, e45183
Panich U, Tangsupa-a-nan V, Onkoksoong T, Kongtaphan K, Kasetsinsombat K, Akarasereenont P, Wongkajornsilp A (2011) Inhibition of UVA-mediated melanogenesis by ascorbic acid through modulation of antioxidant defense and nitric oxide system. Arch Pharm Res 34:811–820
Park SH, Kim DS, Kim WG, Ryoo IJ, Lee DH, Huh CH, Youn SW, Yoo ID, Park KC (2004) Terrein: a new melanogenesis inhibitor and its mechanism. Cell Mol Life Sci 61:2878–2885
Park SJ, Shin JH, Jeong JI, Song JH, Jo YK, Kim ES, Lee EH, Hwang JJ, Lee EK, Chung SJ, Koh JY, Jo DG, Cho DH (2014) Down-regulation of mortalin exacerbates Abeta-mediated mitochondrial fragmentation and dysfunction. J Biol Chem 289:2195–2204
Pavel S, van Nieuwpoort F, van der Meulen H, Out C, Pizinger K, Cetkovska P, Smit NP, Koerten HK (2004) Disturbed melanin synthesis and chronic oxidative stress in dysplastic naevi. Eur J Cancer 40:1423–1430
Porameesanaporn Y, Uthaisang-Tanechpongtamb W, Jarintanan F, Jongrungruangchok S, Thanomsub Wongsatayanon B (2013) Terrein induces apoptosis in HeLa human cervical carcinoma cells through p53 and ERK regulation. Oncol Rep 29:1600–1608
Qu M, Zhou Z, Xu S, Chen C, Yu Z, Wang D (2011) Mortalin overexpression attenuates beta-amyloid-induced neurotoxicity in SH-SY5Y cells. Brain Res 1368:336–345
Qu M, Zhou Z, Chen C, Li M, Pei L, Yang J, Wang Y, Li L, Liu C, Zhang G, Yu Z, Wang D (2012) Inhibition of mitochondrial permeability transition pore opening is involved in the protective effects of mortalin overexpression against beta-amyloid-induced apoptosis in SH-SY5Y cells. Neurosci Res 72:94–102
Rosania GR (2005) Mitochondria give cells a tan. Chem Biol 12:412–413
Salem MM, Shalbaf M, Gibbons NC, Chavan B, Thornton JM, Schallreuter KU (2009) Enhanced DNA binding capacity on up-regulated epidermal wild-type p53 in vitiligo by H2O2-mediated oxidation: a possible repair mechanism for DNA damage. FASEB J 23:3790–3807
Schallreuter KU, Behrens-Williams S, Khaliq TP, Picksley SM, Peters EM, Marles LK, Westerhof W, Miehe B, Fanghanel J (2003) Increased epidermal functioning wild-type p53 expression in vitiligo. Exp Dermatol 12:268–277
Schiller D, Cheng YC, Liu Q, Walter W, Craig EA (2008) Residues of Tim44 involved in both association with the translocon of the inner mitochondrial membrane and regulation of mitochondrial Hsp70 tethering. Mol Cell Biol 28:4424–4433
Shih YY, Lee H, Nakagawara A, Juan HF, Jeng YM, Tsay YG, Lin DT, Hsieh FJ, Pan CY, Hsu WM, Liao YF (2011) Nuclear GRP75 binds retinoic acid receptors to promote neuronal differentiation of neuroblastoma. PLoS One 6, e26236
Shin JM, Kim MY, Sohn KC, Jung SY, Lee HE, Lim JW, Kim S, Lee YH, Im M, Seo YJ, Kim CD, Lee JH, Lee Y, Yoon TJ (2014) Nrf2 negatively regulates melanogenesis by modulating PI3K/Akt signaling. PLoS One 9, e96035
Singh B, Gupta RS (2009) Conserved inserts in the Hsp60 (GroEL) and Hsp70 (DnaK) proteins are essential for cellular growth. Mol Genet Genomics 281:361–373
Smit NP, van Nieuwpoort FA, Marrot L, Out C, Poorthuis B, van Pelt H, Meunier JR, Pavel S (2008) Increased melanogenesis is a risk factor for oxidative DNA damage—study on cultured melanocytes and atypical nevus cells. Photochem Photobiol 84:550–555
Snyder JR, Hall A, Ni-Komatsu L, Khersonsky SM, Chang YT, Orlow SJ (2005) Dissection of melanogenesis with small molecules identifies prohibitin as a regulator. Chem Biol 12:477–484
Stepien K (2010) The role of melanocytes in protection against photooxidative stress Krystyna Stepien. Postepy Biochem 56:290–297
Szklarczyk D, Franceschini A, Wyder S, Forslund K, Heller D, Huerta-Cepas J, Simonovic M, Roth A, Santos A, Tsafou KP, Kuhn M, Bork P, Jensen LJ, von Mering C (2014) STRING v10: protein-protein interaction networks, integrated over the tree of life. Nucleic Acids Res 43:D447–452
Thomas PD, Kishi H, Cao H, Ota M, Yamashita T, Singh S, Jimbow K (1999) Selective incorporation and specific cytocidal effect as the cellular basis for the antimelanoma action of sulphur containing tyrosine analogs. J Invest Dermatol 113:928–934
Usui K, Ikeda T, Horibe Y, Nakao M, Hoshino T, Mizushima T (2015) Identification of HSP70-inducing activity in Arnica montana extract and purification and characterization of HSP70-inducers. J Dermatol Sci 78:67–75
Vachtenheim J, Borovansky J (2010) "Transcription physiology" of pigment formation in melanocytes: central role of MITF. Exp Dermatol 19:617–627
Wadhwa R, Kaul SC, Ikawa Y, Sugimoto Y (1993) Identification of a novel member of mouse hsp70 family. Its association with cellular mortal phenotype. J Biol Chem 268:6615–6621
Wadhwa R, Takano S, Kaur K, Aida S, Yaguchi T, Kaul Z, Hirano T, Taira K, Kaul SC (2005) Identification and characterization of molecular interactions between mortalin/mtHsp70 and HSP60. Biochem J 391:185–119
Widodo N, Kaur K, Shrestha BG, Takagi Y, Ishii T, Wadhwa R, Kaul SC (2007) Selective killing of cancer cells by leaf extract of Ashwagandha: identification of a tumor-inhibitory factor and the first molecular insights to its effect. Clin Cancer Res 13:2298–2306
Williams D, Jung DW, Khersonsky SM, Heidary N, Chang YT, Orlow SJ (2004) Identification of compounds that bind mitochondrial F1F0 ATPase by screening a triazine library for correction of albinism. Chem Biol 11:1251–1259
Xu F, Luo JH, Jin JH (2008) Alpha-synuclein interacted proteins: the relevance with the pathogenesis of Parkinson’s disease. Zhejiang Da Xue Xue Bao Yi Xue Ban 37:524–530
Yamashita Y, Hoshino T, Matsuda M, Kobayashi C, Tominaga A, Nakamura Y, Nakashima K, Yokomizo K, Ikeda T, Mineda K, Maji D, Niwano Y, Mizushima T (2010) HSP70 inducers from Chinese herbs and their effect on melanin production. Exp Dermatol 19:e340–342
Yamashita Y, Ikeda T, Matsuda M, Maji D, Hoshino T, Mizushima T (2012) Purification and characterization of HSP-inducers from Eupatorium lindleyanum. Biochem Pharmacol 83:909–922
Zhu JY, Vereshchagina N, Sreekumar V, Burbulla LF, Costa AC, Daub KJ, Woitalla D, Martins LM, Kruger R, Rasse TM (2013) Knockdown of Hsc70-5/mortalin induces loss of synaptic mitochondria in a Drosophila Parkinson’s disease model. PLoS One 8, e83714
Zucca FA, Basso E, Cupaioli FA, Ferrari E, Sulzer D, Casella L, Zecca L (2014) Neuromelanin of the human substantia nigra: an update. Neurotox Res 25:13–23
Acknowledgments
We thank T. Nakamoto, R. Singh, T. Yaguchi, and N. Shah for their help.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflict of interest.
Author Contributions
RW and SCK conceived and coordinated the study and wrote the paper. RW, DP, RG, NW, NN, LL, and HA performed the experiments. RW, COY, NA, CM, and SCK analyzed the data. All authors reviewed the results and approved the final version of the manuscript.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
Schematic diagram showing loss-of-function screening for genes involved in melanogenesis pathway. Cells were transfected with shRNAs. Transfected cells were selected in puromycin-supplemented medium and then subjected to OAG treatment. Melanin content, tyrosinase activity, and melanosome staining were performed in parallel. The shRNAs that caused reduction in all the three parameters were selected for the next round. In total, four rounds of screening were done. (GIF 14 kb)
Supplementary Fig. 2
Effect of mitochondrial inhibitors and OAG on ROS and mitochondrial membrane potential. H2O2 was used as a positive control for oxidative stress. It caused increase in ROS and collapse of mitochondrial membrane potential (loss of red staining). Creatine and OAG caused similar, although somewhat milder effect as revealed by increase in ROS staining and loss of red staining. (GIF 15 kb)
Rights and permissions
About this article
Cite this article
Wadhwa, R., Priyandoko, D., Gao, R. et al. Stress chaperone mortalin regulates human melanogenesis. Cell Stress and Chaperones 21, 631–644 (2016). https://doi.org/10.1007/s12192-016-0688-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-016-0688-2