Abstract
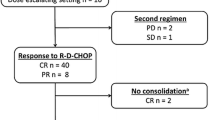
The feasibility and efficacy of cyclophosphamide, vincristine, doxorubicin, and methotrexate (CODOX-M)/ifosfamide, etoposide, and cytarabine (IVAC) therapy in Japanese adult patients with Burkitt lymphoma (BL) and B cell lymphoma, unclassifiable, with features intermediate between diffuse large B cell lymphoma and BL (intermediate DLBCL/BL) have never been reported. The effects of adding rituximab to CODOX-M/IVAC have not been published either. Fifteen consecutive patients with a median age of 39 years were treated with modified CODOX-M/IVAC regimen (particularly, reducing the dose of methotrexate to 3 g/m2) with or without rituximab at our institution. Although all patients developed grade 4 neutropenia and grade 3/4 thrombocytopenia/anemia, 93% had febrile neutropenia, 60% showed transaminase elevation, and 40% had mucositis/stomatitis (all grade 3), there were no treatment-related deaths. Two of nine patients treated with rituximab developed biphasic late-onset neutropenia. Thirteen patients (87%) showed complete responses. The remaining two patients had refractory disease; one had presented with peritoneal dissemination and complex chromosomal abnormalities, while the other had double IGH–MYC and IGH–BCL2 translocations. The estimated 5-year overall and progression-free survival were 87% each, with a median follow-up of 74 months. In conclusion, our modified CODOX-M/IVAC regimen is well tolerated and highly effective in Japanese adult patients with BL and intermediate DLBCL/BL, warranting a larger study for confirmation.

Similar content being viewed by others
References
Diebold J, Jaffe ES, Raphael M, Warnke RA. Burkitt lymphoma. In: Jaffe ES, Harris NL, Stein H, Vardiman JW, editors. World Health Organization classification of tumours. Pathology and genetics: tumours of hematopoietic and lymphoid tissues. Lyon, France: International Agency for Research on Cancer; 2001. p. 181–4.
Leoncini L, Raphel M, Stein H, Harris NL, Jaffe ES, Kluin PM. Burkitt lymphoma. In: Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, et al., editors. World Health Organization classification of tumours, WHO classification of tumours of hematopoietic and lymphoid tissues. Lyon, France: International Agency for Research on Cancer; 2008. p. 262–4.
Blum KA, Lozanski G, Byrd JC. Adult Burkitt leukemia and lymphoma. Blood. 2004;104:3009–20.
The Non-Hodgkin’s Lymphoma Classification Project. A clinical evaluation of the International Lymphoma Study Group classification of non-Hodgkin’s lymphoma. Blood. 1997; 89:3909–18.
Harris NL, Jaffe ES, Stein H, Banks PM, Chan JK, Cleary ML, et al. A revised European–American classification of lymphoid neoplasms: a proposal from the International Lymphoma Study Group. Blood. 1994;84:1361–92.
Kluin PM, Harris NL, Stein H, Leoncini L, Raphael M, Campo E, et al. B-cell lymphoma unclassifiable, with features intermediate between diffuse large B-cell lymphoma and Burkitt lymphoma. In: Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, et al., editors. World Health Organization classification of tumours, WHO classification of tumours of hematopoietic and lymphoid tissues. Lyon, France: International Agency for Research on Cancer; 2008. p. 265–6.
Smeland S, Blystad AK, Kvaløy SO, Ikonomou IM, Delabie J, Kvalheim G, et al. Treatment of Burkitt’s/Burkitt-like lymphoma in adolescents and adults: a 20-year experience from Norwegian Radium Hospital with use of three successive regimens. Ann Oncol. 2004;15:1072–8.
Dave SS, Fu K, Wright GW, Lam LT, Kluin P, Boerma EJ, et al. Molecular diagnosis of Burkitt’s lymphoma. N Engl J Med. 2006;354:2431–42.
Murphy SB, Bowman WP, Abromowitch M, Mirro J, Ochs J, Rivera G, et al. Results of treatment of advanced-stage Burkitt’s lymphoma and B cell (Sig+) acute lymphoblastic leukaemia with high-dose fractionated cyclophosphamide and co-ordinated high-dose methotrexate and cytarabine. J Clin Oncol. 1986;4:1732–9.
Patte C, Philip T, Rodary C, Zucker JM, Behrendt H, Gentet JC, et al. High survival rate in advanced-stage B-cell lymphomas and leukaemia’s without CNS involvement with a short intensive polychemotherapy: results from the French Pediatric Oncology Society of a randomized trial of 216 children. J Clin Oncol. 1991;9:123–32.
Schwenn MR, Blattner SR, Lynch E, Weinstein HJ. HiC-COM: a 2-month intensive chemotherapy regimen for children with stage III and IV Burkitt’s lymphoma and B-cell acute lymphoblastic leukemia. J Clin Oncol. 1991;9:133–8.
Reiter A, Schrappe M, Ludwig WD, Lampert F, Harbott J, Henze G, et al. Favourable outcome of B-cell acute lymphoblastic leukaemia in childhood: a report of three consecutive studies of the BFM group. Blood. 1992;80:2471–8.
Bernstein JI, Coleman CN, Strickler JG, Dorfman RF, Rosenberg SA. Combined modality therapy for adults with small noncleaved cell lymphoma (Burkitt’s and Non-Burkitt’s Types). J Clin Oncol. 1986;4:847–58.
van Imhoff GW, van der Holt B, MacKenzie MA, Ossenkoppele GJ, Wijermans PW, Kramer MH, et al. Short intensive sequential therapy followed by autologous stem cell transplantation in adult Burkitt, Burkitt-like and lymphoblastic lymphoma. Leukemia. 2005;19:945–52.
Braziel RM, Arber DA, Slovak ML, Gulley ML, Spier C, Kjeldsberg C, et al. The Burkitt-like lymphomas: a Southwest Oncology Group study delineating phenotypic, genotypic, and clinical features. Blood. 2001;97:3713–20.
Magrath I, Adde M, Shad A, Venzon D, Seibel N, Gootenberg J, et al. Adult and children with small non-cleaved-cell lymphoma have a similar excellent outcome when treated with the same chemotherapy regimen. J Clin Oncol. 1996;14:925–34.
Mead GM, Sydes MR, Walewski J, Grigg A, Hatton CS, Norbert P, et al. An international evaluation of CODOX-M and CODOX-M alternating with IVAC in adult Burkitt’s lymphoma: results of United Kingdom Lymphoma Group LY06 study. Ann Oncol. 2002;13:1264–74.
LaCasce A, Howard O, Lib S, Fisher D, Weng A, Neuberg D, et al. Modified Magrath regimens for adults with Burkitt and Burkitt-like lymphomas: preserved efficacy with decreased toxicity. Leuk Lymphoma. 2004;45:761–7.
Mead GM, Barrans SL, Qian W, Walewski J, Radford JA, Wolf M, et al. A prospective clinicopathologic study of dose-modified CODOX-M/IVAC in patients with sporadic Burkitt lymphoma defined using cytogenetic and immunophenotypic criteria (MRC/NCRI LY 10 trial). Blood. 2008;112:2248–60.
Coiffier B, Lepage E, Briere J, Herbrecht R, Tilly H, Bouabdallah R, et al. CHOP chemotherapy plus rituximab compared with CHOP alone in elderly patients with diffuse large B-cell lymphoma. N Engl J Med. 2002;346:235–42.
Pfreundschuh M, Trumper L, Osterborg A, Pettengell R, Trneny M, Imrie K, et al. CHOP-like chemotherapy plus rituximab versus CHOP-like chemotherapy alone in young patients with good-prognosis diffuse large B-cell lymphoma; a randomized controlled trial by the MabThera International Trial (MInT) Group. Lancet Oncol. 2006;7:379–91.
Hiddemann W, Kneba M, Dreyling M, Schmitz N, Lengfelder E, Schmits R, et al. Frontline therapy with rituximab added to the combination of cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP) significantly improves the outcome for the patients with advanced-stage follicular lymphoma compared with therapy with CHOP alone; results of a prospective randomized study of the German Low-Grade Lymphoma Study Group. Blood. 2005;106:3725–32.
Nitta E, Izutsu K, Sato T, Ota Y, Takeuchi K, Kamijo A, et al. A high incidence of late-onset neutropenia following rituximab-containing chemotherapy as a primary treatment of CD20-positive B-cell lymphoma: a single-institution study. Ann Oncol. 2007;18:364–9.
Hui CK, Cheung WW, Zhang HY, Au WY, Yueng YH, Leung AY, et al. Kinetics and risk of de novo hepatitis B infection in HBsAg-negative patients undergoing cytotoxic chemotherapy. Gastroenterology. 2006;131:59–68.
Yokoyama H, Watanabe T, Maruyama D, Kim SW, Kobayashi Y, Tobinai K. Progressive multifocal leukoencephalopathy in a patient with B-cell lymphoma during rituximab-containing chemotherapy: case report and review of the literature. Int J Hematol. 2008;88:443–7.
Carson KR, Evens AM, Richey EA, Habermann TM, Focosi D, Seymour JF, et al. Progressive multifocal leukoencephalopathy after rituximab therapy in HIV-negative patients: a report of 57 cases from the Research on Adverse Drug Events and Reports (RADAR) project. Blood. 2009;113:4834–40.
Ennishi D, Terui Y, Yokoyama M, Mishima Y, Takahashi S, Takeuchi K, et al. Increased incidence of interstitial pneumonia by CHOP combined with rituximab. Int J Hematol. 2008;87:393–7.
Thomas DA, Faderl S, O’Brien S, Bueso-Ramos C, Cortes J, Garcia-Manero G, et al. Chemoimmunotherapy with hyper-CVAD plus rituximab for the treatment of adult Burkitt and Burkitt-type lymphoma or acute lymphoblastic leukemia. Cancer. 2006;106:1569–80.
Abramson JS, Barnes JA, Toomey CE, Jacobsen ED, Armand P, Takvorian T, et al. Rituximab added to CODOX-M/IVAC is highly effective in HIV-negative and HIV-positive Burkitt lymphoma. Blood. 2008; 112:abstract 3595.
Mohamedbhai SG, Lowry L, Goldstone AH, Linch DC, Ardeshna KM. Rituximab in combination with CODOX-M/IVAC: toxicity and efficacy in 17 adults with non-HIV related B-cell non-Hodgkin lymphomas with >95% proliferation index. Br J Haematol. 2009;145(1):36.
Barnes JA, LaCasce AS, Feng Y, Toomey C, Neuberg D, Hochberg EP, et al. Rituximab added to CODOX-M/IVAC has no clear benefit compared to CODOX-M/IVAC alone in adult patients with Burkitt lymphoma. Blood. 2009; 114:abstract 1667.
Sekiguchi N, Kobayashi Y, Yokota Y, Kusumoto S, Tanimoto K, Watanabe T, et al. Follicular lymphoma subgrouping by fluorescence in situ hybridization analysis. Cancer Sci. 2005;96:77–82.
Maeshima AM, Omatsu M, Nomoto J, Maruyama D, Kim SW, Watanabe T, et al. Diffuse large B-cell lymphoma after transformation from low-grade follicular lymphoma: morphological, immunohistochemical, and FISH analyses. Cancer Sci. 2008;99:1760–8.
Carbone PP, Kaplan HS, Musshoff K, Smithers DW, Tubiana M. Report of the committee on Hodgkin’s disease staging classification. Cancer Res. 1971;31:1860–1.
Cheson BD, Horning SJ, Coiffier B, Shipp MA, Fisher RI, Connors JM, et al. Report of an international workshop to standardize response criteria for non-Hodgkin’s lymphoma. J Clin Oncol. 1999;17:1244–53.
Cheson BD, Pfistner B, Juweid ME, Gascoyne RD, Specht L, Horning SJ, et al. Revised response criteria for malignant lymphoma. J Clin Oncol. 2007;25:579–86.
NCI Common Terminology Criteria for Adverse Events (CTCAE) v 3.0. (Cited 9 Aug 2006) Available from http://ctep.cancer.gov/protocolDevelopment/electronic_applications/docs/ctcaev3.pdf.
The International Non-Hodgkin’s Lymphoma Prognostic Factors Project: a predictive model for aggressive non-Hodgkin’s lymphoma. N Engl J Med. 1993; 329:987–94.
Weintraub M, Adde MA, Venzon DJ, Shad AT, Horak ID, Neely JE, et al. Severe atypical neuropathy associated with administration of hematopoietic colony-stimulating factors and vincristine. J Clin Oncol. 1996;14:935–40.
Nomura Y, Karube K, Suzuki R, Ying G, Takeshita M, Hirose S, et al. High-grade mature B-cell lymphoma with Burkitt-like morphology: results of a clinicopathological study of 72 Japanese patients. Cancer Sci. 2008;99:246–52.
Johnson NA, Savage KJ, Ludkovski O, Ben-Neriah S, Woods R, Steidl C, et al. Lymphomas with concurrent BCL2 and MYC translocations: the critical factors associated with survival. Blood. 2009;114:2273–9.
Acknowledgments
This work was supported in part by a Grant-in-Aid for Cancer Research from the Ministry of Health, Labour and Welfare of Japan (to T.W. and K.T.).
Conflict of interest
None declared.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Maruyama, D., Watanabe, T., Maeshima, A.M. et al. Modified cyclophosphamide, vincristine, doxorubicin, and methotrexate (CODOX-M)/ifosfamide, etoposide, and cytarabine (IVAC) therapy with or without rituximab in Japanese adult patients with Burkitt lymphoma (BL) and B cell lymphoma, unclassifiable, with features intermediate between diffuse large B cell lymphoma and BL. Int J Hematol 92, 732–743 (2010). https://doi.org/10.1007/s12185-010-0728-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-010-0728-0