Abstract
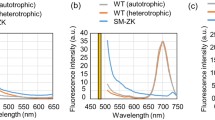
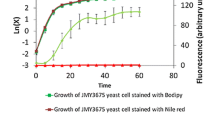
Herein, we combined metabolic evolution with fluorescence-activated cell sorting (FACS) of cells stained with the lipophilic dye BODIPY for isolation of SCO-overproducing strains of Yarrowia lipolytica. Metabolic evolution was implemented for enrichment of high SCO-accumulating mutant population which were then sorted by fluorescence signals using flow cytometry coupled with FACS. A mutant isolated by this approach exhibited 1.5- and 1.2-fold higher SCO titer and content, respectively, than the wild type under batch culture of sugarcane bagasse hydrolysate complex media. In addition, the mutant had whole-cell fatty acid composition different from that of the wild type with higher oleic and linoleic acids. Dual-stage fed-batch process applied to the mutant yielded high SCO titer of 49.7 g/L from hydrolysates, a fourfold improvement over batch process. This study highlights evolution-based in conjunction with fluorescence-based high-throughput screening as a powerful strategy for attaining high single-cell oil-accumulating phenotype in Y. lipolytica exploited for sustainable biodiesel and oleochemicals synthesis.





Similar content being viewed by others
References
Jin M, Slininger PJ, Dien BS, Waghmode S, Moser BR, Orjuela A, Sousa Lda C, Balan V (2015) Microbial lipid-based lignocellulosic biorefinery: feasibility and challenges. Trends Biotechnol 33(1):43–54
Koutinas AA, Chatzifragkou A, Kopsahelis N (2014) Design and techno-economic evaluation of microbial oil production as a renewable resource for biodiesel and oleo-chemical production. Fuel 116:566–577
Fontanille P, Kumar V, Christophe G, Nouaille R, Larroche C (2012) Bioconversion of volatile fatty acids into lipids by the oleaginous yeast Yarrowia lipolytica. Bioresour Technol 114:443–449
Back A, Rossignol T, Krier F, Nicaud JM, Dhulster P (2016a) High-throughput fermentation screening for the yeast Yarrowia lipolytica with real-time monitoring of biomass and lipid production. Microb Cell Factories 23(15):147
Ledesma-Amaro R, Nicaud JM (2016) Yarrowia lipolytica as a biotechnological chassis to produce usual and unusual fatty acids. Prog Lipid Res 61:40–50
Freitas C, Nobre B, Gouveia L, Roseiro J, Reis A, da Silva TL (2014) New at-line flow cytometric protocols for determining carotenoidcontent and cell viability during Rhodosporidium toruloides NCYC 921 batch growth. Process Biochem 49:554–562
Besmer MD, Weissbrodt DG, Kratochvil BE, Sigrist JA, Weyland MS, Hammes F (2014) The feasibility of automated online flow cytometry for in-situ monitoring of microbial dynamics in aquatic ecosystems. Front Microbiol 5:265. doi:10.3389/fmicb. 2014.00265
Back A, Rossignol T, Krier F, Nicaud JM, Dhulster P (2016b) High-throughput fermentation screening for the yeast Yarrowia lipolytica with real-time monitoring of biomass and lipid production. Microb Cell Factories 23(15):147
Hyka P, Lickova S, Pribyl P, Melzoch K, Kovar K (2013) Flow cytometry for the development of biotechnological processes with microalgae. Biotechnol Adv 31:2–16
Friedlander J, Tsakraklides V, Kamineni A, Greenhagen EH, Consiglio AL, MacEwen K, Crabtree DV, Afshar J, Nugent RL, Hamilton MA, Joe SA, South CR, Stephanopoulos G, Brevnova EE (2016) Engineering of a high lipid producing Yarrowia lipolytica strain. Biotechnol Biofuels 9:77
Velmurugan N, Sung M, Yim SS, Park MS, Yang JW, Jeong KJ (2013) Evaluation of intracellular lipid bodies in Chlamydomonas reinhardtii strains by flow cytometry. Bioresour Technol 138:30–37
Xie B, Stessman D, Hart JH, Dong H, Wang Y, Wright DA, Nikolau BJ, Spalding MH, Halverson LJ (2014) High-throughput fluorescence-activated cell sorting for lipid hyperaccumulating Chlamydomonas reinhardtii mutants. Plant Biotechnol J 12:872–882
Terashima M, Freeman ES, Jinkerson RE, Jonikas MC (2015) A fluorescence-activated cell sorting-based strategy for rapid isolation of high-lipid Chlamydomonas mutants. Plant J 81(1):147–159
Unrean P, Khajeeram S (2016) Optimization and techno-economic assessment of high-solid fed-batch saccharification and ethanol fermentation by Scheffersomyces stipitis and Saccharomyces cerevisiae consortium. Renew Energy 99:1062–1072
Schneiter R, Daum G (2006) Extraction of yeast lipids. Methods Mol Biol 313:41–45
Blazeck J, Hill A, Liu L, Knight R, Miller J, Pan A, Otoupal P, Alper HS (2014) Harnessing Yarrowia lipolytica lipogenesis to create a platform for lipid and biofuel production. Nat Commun 5:3131
Unrean P, Srienc F (2011) Metabolic networks evolve towards states of maximum entropy production. Metab Eng 13(6):666–673
Sitepu IR, Ignatia L, Franz AK, Wong DM, Faulina SA, Tsui M, Kanti A, Boundy-Mills K (2012) An improved high-throughput Nile red fluorescence assay for estimating intracellular lipids in a variety of yeast species. J Microbiol Methods 91(2):321–328
Silverman AM, Qiao K, Xu P, Stephanopoulos G (2016) Functional overexpression and characterization of lipogenesis-related genes in the oleaginous yeast Yarrowia lipolytica. Appl Microbiol Biotechnol 100(8):3781–3798
Wahlen BD, Morgan MR, McCurdy AT, Willis RM, Morgan MD, Dye DJ, Bugbee B, Wood BD, Seefeldt LC (2013) Biodiesel from microalgae, yeast, and bacteria: engine performance and exhaust emissions. Energy Fuel 27:220–228
Unrean P, Khajeeram S, Laoteng K (2016) Systematic optimization of fed-batch simultaneous saccharification and fermentation at high-solid loading based on enzymatic hydrolysis and dynamic metabolic modeling of Saccharomyces cerevisiae. Appl Microbiol Biotechnol 100(5):2459–2470
Qiao K, Imam Abidi SH, Liu H, Zhang H, Chakraborty S, Watson N, Kumaran Ajikumar P, Stephanopoulos G (2015) Engineering lipid overproduction in the oleaginous yeast Yarrowia lipolytica. Metab Eng 29:56–65
Liu L, Pan A, Spofford C, Zhou N, Alper HS (2015) An evolutionary metabolic engineering approach for enhancing lipogenesis in Yarrowia lipolytica. Metab Eng 29:36–45
Xu P, Qiao K, Stephanopoulos G (2017) Engineering oxidative stress defense pathways to build a robust lipid production platform in Yarrowia lipolytica. Biotechnol Bioeng 114(7):1521–1530
Acknowledgements
The authors would like to thank the National Center for Genetic Engineering and Biotechnology, Thailand, for the financial support of this project (Grant No. P-16-50341).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Unrean, P., Champreda, V. High-Throughput Screening and Dual Feeding Fed-Batch Strategy for Enhanced Single-Cell Oil Accumulation in Yarrowia lipolytica . Bioenerg. Res. 10, 1057–1065 (2017). https://doi.org/10.1007/s12155-017-9865-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12155-017-9865-0