Abstract
Purpose
To evaluate predictive factors associated with detectable prostate-specific antigen (PSA) and describe clinical recurrence (CR) and cancer-specific mortality (CSM) after robot-assisted radical prostatectomy (RARP).
Methods
The study included 2500 patients who were treated with RARP at a single institution between 2000 and 2016. All patients had clinically localized PCa. Patients were divided into two groups according to PSA value at 6 weeks after surgery: undetectable (n = 2271; PSA < 0.1 ng/dl) and persistently elevated (n = 229; PSA ≥ 0.1 ng/dl). The association between various covariates and: (1) detectable PSA and (2) CR was evaluated. Kaplan–Meier analyses estimated CR and CSM rates according to PSA persistence.
Results
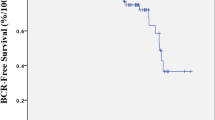
Inside the group of detectable PSA, 146 men (63.75%) received adjuvant treatments, 44 patients (19.21%) salvages therapies and 38 men (16.5%) experienced CR. Factors associated with aggressive disease predicted PSA persistence. Within patients with detectable PSA, pathologic stage ≥ pT3a (HR 2.71; p < 0.029) and to received adjuvant androgen deprivation therapy (ADT) due to bad prognosis tumors (HR 13.36; p < 0.001) were associated with CR. Overall 14 (0.56%) died of PCa. 5 and 10-year CSM rates were higher for patients with CR (9.6 and 23.7%, p < 0.001), and Gleason ≥ 8 (5.7 and 6.9%, p = 0.003).
Conclusions
A detectable PSA is affected by factors associated with aggressive prostate cancer. Within men with persistent PSA, those with higher pathologic stage and who received adjuvant ADT are more likely to have CR. Patients with CR, Gleason ≥ 8, and those who received adjuvant ADT must have a close monitoring due to the high rate of mortality.
Similar content being viewed by others
References
Arnold M, Karim-Kos HE, Coebergh JW, Byrnes G, Antilla A, Ferlay J, et al. Recent trends in incidence of five common cancers in 26 European countries since 1988: analysis of the European Cancer Observatory. Eur J Cancer. 2013;8:S0959–8049.
Stattin P, Holmberg E, Johansson JE, Holmberg L, Adolfsson J, Hugosson J. Outcomes in localized prostate cancer: national Prostate Cancer Register of Sweden follow-up study. J Natl Cancer Inst. 2010;102:950–8.
Edwards BK, Noone AM, Mariotto AB, Simard EP, Boscoe FP, Henley SJ, et al. Annual Report to the Nation on the status of cancer, 1975–2010, featuring prevalence of comorbidity and impact on survival among persons with lung, colorectal, breast, or prostate cancer. Cancer. 2014;120:1290–314.
Bill-Axelson A, Holmberg L, Ruutu M, Häggman M, Andersson SO, Bratell S, et al. Radical prostatectomy versus watchful waiting in early prostate cancer. N Engl J Med. 2005;352:1977.
Pound CR, Partin AW, Eisenberger MA, Chan DW, Pearson JD, Walsh PC. Natural history of progression after PSA elevation following radical prostatectomy. JAMA. 1999;281:1591.
Walz J, Chun FK, Klein EA, Reuther A, Saad F, Graefen M, et al. Nomogram predicting the probability of early recurrence after radical prostatectomy for prostate cancer. J Urol. 2009;181:601.
Ficarra V, Novara G, Artibani W, Cestari A, Galfano A, Graefen M, et al. Retropubic, laparoscopic, and robot-assisted radical prostatectomy: a systematic review and cumulative analysis of comparatives studies. Eur Urol. 2009;55:1037–63.
Coelho RF, Rocco B, Patel MB. Retropubic, laparoscopic, and robot-assisted radical prostatectomy: a critical review of outcomes reported by high-volume centers. J Endourol. 2010;24:2003–15.
Suardi N, Ficarra V, Willemsen P, De Wil P, Gallina A, De Naeyer G, Schatteman P, Montorsi F, Carpentier P, Mottrie A. Long-term biochemical recurrence rates after robot-assisted radical prostatectomy: analysis of a single-center series of patients with a minimum follow-up of 5 years. Urology. 2012;79:133–8.
Sooriakumaran P, Haendler L, Nyberg T, Gronberg H, Nilsson A, Carlsson S, Hosseini A, Adding C, Jonsson M, Ploumidis A, Egevad L, Steineck G, Wiklund P. Biochemical recurrence after robot-assisted radical prostatectomy in a European single-centre cohort with a minimum follow-up time of 5 years. Eur Urol. 2012;62:768–74.
Liss MA, Lusch A, Morales B, Beheshti N, Skarecky D, Narula N, Osann K, Ahlering TE. Robot-assisted radical prostatectomy: 5-year oncological and biochemical outcomes. J Urol. 2012;188:2205–10.
De Boo L, Pintilie M, Yip P, Baniel J, Fleshner N, Margel D. Time from first detectable PSA following radical prostatectomy to biochemical recurrence: a competing risk analysis. Can Urol Assoc J. 2015;9:E14–21.
Audenet F, Seringe E, Drouin SJ, Comperat E, Cussenot O, Bitker MO, Rouprêt M. Persistently elevated prostate-specific antigen at six weeks after radical prostatectomy helps in early identification of patients who are likely to recur. World J Urol. 2012;30:239–44.
Moreira DM, Presti JC, Aronson WJ, Terris MK, Kane CJ, Amling CL, Freedland SJ. Definition and preoperative predictors of persistently elevated prostate specific antigen after radical prostatectomy: results from the SEARCH database. BJU Int. 2009;105:1541–7.
Eisenberg ML, Davies BJ, Cooperberg MR, Cowan JE, Carroll PR. Prognostic implications of an undetectable ultrasensitive prostate specific antigen level after radical prostatectomy. Eur Urol. 2010;57:622–30.
Kumar A, Samavedi S, Mouraviev V, Bates AS, Coelho RF, Rocco B, et al. Predictive factor and oncological outcomes of persistently elevated prostate-specific antigen in patients following robot-assisted radical prostatectomy. J Robot Surg. 2017;11(1):37–45.
Bianchi L, Nini A, Bianchi M, Gandaglia G, Fossati N, Suardi N, Moschini M, Dell’Oglio P, Schiavina R, Montorsi F, Briganti A. The role of prostate-specific antigen persistence after radical prostatectomy for the prediction of clinical progression and cancer-specific mortality in node-positive prostate cancer patients. Eur Urol. 2016;69:1142–8.
Partin AW, Oesterling JE. The clinical usefulness of prostate specific antigen: update 1994. J Urol. 1994;152:1358–68.
Freedland SJ, Humphreys EB, Mangold LA, Eisenberg M, Dorey FJ, Walsh PC, et al. Risk of prostate cancer-specific mortality following biochemical recurrence after radical prostatectomy. JAMA. 2005;294:433–9.
D’Amico AV, Chen MH, Roehl KA, Catalona WJ. Identifying patients at risk for significant versus clinically insignificant postoperative prostate specific antigen failure. J Clin Oncol. 2005;23:4975–9.
Rogers CG, Khan MA, Miller MC. Natural history of disease progression in patients who fail to achieve an undetectable prostate specific antigen level after undergoing radical prostatectomy. Cancer. 2004;101:2549–56.
Naselli A, Introini C, Andreatta R, Spina B, Truini M, Puppo P. Prognostic factors of persistently detectable PSA after radical prostatectomy. Int J Urol. 2009;16:82–6.
Stish BJ, Pisansky TM, Harmsen WS, Davis BJ, Tzou KS, Choo R, Buskirk SJ. Metastasis-free and survival outcomes with early salvage radiotherapy in men with detectable prostate-specific antigen after prostatectomy for prostate cancer. J Clin Oncol. 2016;34:3864–71.
Messing EM, Manola J, Yao J, Kiernan M, Crawford D, Wilding G, et al. Inmediate versus deferred androgen deprivation treatment in patients with node-positive prostate cancer after radical prostatectomy and pelvic lymphadenectomy. Lancet Oncol. 2006;7:472–9.
Schumacher MC, Burkhard FC, Thalmann GN, Fleischmann A, Studer UE. Good outcome for patients with few lymph node metastases after radical retropubic prostatectomy. Eur Urol. 2008;54:344–52.
Author information
Authors and Affiliations
Contributions
SG-B: project development, data collection and manuscript writing. FR: project development. IN-S: statistical analysis. VS: data collection. RS-S: data collection. EB: editing. MG: editing. XC: project development and manuscript writing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
CEPAR: Comité d’Evaluation des Protocoles et d’Aide à la Recherche (Protocol Evaluation Committee and for Research Assistance) committee approved the study.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
García-Barreras, S., Rozet, F., Nunes-Silva, I. et al. Predictive factors and the important role of detectable prostate-specific antigen for detection of clinical recurrence and cancer-specific mortality following robot-assisted radical prostatectomy. Clin Transl Oncol 20, 1004–1010 (2018). https://doi.org/10.1007/s12094-017-1812-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-017-1812-1