Abstract
Background
Current knowledge suggests that proton pump inhibitors (PPIs) are associated with an increased risk of hepatic encephalopathy (HE) and spontaneous bacterial peritonitis (SBP). These conditions and PPI use are related to gut microbiota. The aim of this study is to research the changes in gut microbiota caused by PPI in patients with chronic liver disease.
Methods
From 198 Japanese patients, 31 patients in the PPI and non-PPI groups were matched using propensity score matching (PSM) based on age, sex, and Child–Turcotte–Pugh class. We investigated the gut microbial composition of stool samples using the Illumina MiSeq sequencing platform and compared them using linear discriminant analysis effect size and phylogenetic investigation of communities by reconstruction of unobserved states.
Results
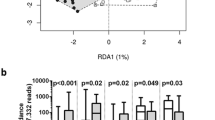
Before PSM, Child–Turcotte–Pugh score (p = 0.038), ascites (p = 0.049), encephalopathy (p = 0.023), and esophageal varices (p < 0.01) were significantly higher in the PPI group than in the non-PPI group. After PSM, six genera, consisting of Lactobacillus, Streptococcus, Selenomonas, Veillonella, Campylobacter, and Haemophilus were enriched in the PPI group. Eggerthella, Paraprevotella, Turicibacter, Dorea, Anaerotruncus, and Ruminococcus were less abundant in the PPI group. We identified five types of level 3 KEGG pathways predicted to be significantly different.
Conclusions
Part of microbial changes caused by PPI use was common to the changes by progression of liver cirrhosis. Increases in oral bacterial flora and decreases in autochthonous flora may produce the intestinal environment which tends to make the risk factor for HE or SBP.




Similar content being viewed by others
References
Tsai C-F, Chen M-H, Wang Y-P, Chu C-J, Huang Y-H, Lin H-C, et al. Proton pump inhibitors increase risk for hepatic encephalopathy in patients with cirrhosis in a population study. Gastroenterology. 2017;152:134–141 (W.B. Saunders)
Yu T, Tang Y, Jiang L, Zheng Y, Xiong W, Lin L. Proton pump inhibitor therapy and its association with spontaneous bacterial peritonitis incidence and mortality: a meta-analysis. Dig Liver Dis. 2016;48:353–359 (W.B. Saunders)
Naito Y, Kashiwagi K, Takagi T, Andoh A, Inoue R. Intestinal dysbiosis secondary to proton-pump inhibitor use. Digestion. 2018;8566:8195–8204
Minalyan A, Gabrielyan L, Scott D, Jacobs J, Pisegna JR. The gastric and intestinal microbiome: role of proton pump inhibitors. Curr Gastroenterol Rep. 2017;19:42
Fukui H. Gut microbiome-based therapeutics in liver cirrhosis: basic consideration for the next step. J Clin Transl Hepatol. 2017;5:249–260 (Xia & He Publishing Inc. (USA))
Bajaj JS, Acharya C, Fagan A, White MB, Gavis E, Heuman DM, et al. Proton pump inhibitor initiation and withdrawal affects gut microbiota and readmission risk in cirrhosis. Am J Gastroenterol. 2018. https://doi.org/10.1038/s41395-018-0085-9 (Nature Publishing Group)
Williamson E, Morley R, Lucas A, Carpenter J. Propensity scores: from naïve enthusiasm to intuitive understanding. Stat Methods Med Res. 2012;21:273–293 (SAGE Publications, London)
Bajaj JS, Cox IJ, Betrapally NS, Heuman DM, Schubert ML, Ratneswaran M, et al. Systems biology analysis of omeprazole therapy in cirrhosis demonstrates significant shifts in gut microbiota composition and function. AJP Gastrointest Liver Physiol. 2014;307:G951–G957 (American Physiological Society)
Jackson MA, Goodrich JK, Maxan M-E, Freedberg DE, Abrams JA, Poole AC, et al. Proton pump inhibitors alter the composition of the gut microbiota. Gut. 2016;65:749–756 (BMJ Publishing Group)
Tsuda A, Suda W, Morita H, Takanashi K, Takagi A, Koga Y, et al. Influence of proton-pump inhibitors on the luminal microbiota in the gastrointestinal tract. Clin Transl Gastroenterol. 2015;6:e89 (Nature Publishing Group)
Dam G, Vilstrup H, Watson H, Jepsen P. Proton pump inhibitors as a risk factor for hepatic encephalopathy and spontaneous bacterial peritonitis in patients with cirrhosis with ascites. Hepatology. 2016;64:1265–1272
Bian J, Wang A, Lin J, Wu L, Huang H, Wang S, et al. Association between proton pump inhibitors and hepatic encephalopathy: a meta-analysis. Medicine (Baltimore). 2017;96:e6723
Deshpande A, Pasupuleti V, Thota P, Pant C, Mapara S, Hassan S, et al. Acid-suppressive therapy is associated with spontaneous bacterial peritonitis in cirrhotic patients: a meta-analysis. J Gastroenterol Hepatol. 2013;28:235–242
Su T, Lai S, Lee A, He X, Chen S. Meta-analysis: proton pump inhibitors moderately increase the risk of small intestinal bacterial overgrowth. J Gastroenterol. 2018;53:27–36 (Springer, Japan)
Giamarellos-Bourboulis EJ, Pyleris E, Barbatzas C, Pistiki A, Pimentel M. Small intestinal bacterial overgrowth is associated with irritable bowel syndrome and is independent of proton pump inhibitor usage. BMC Gastroenterol. 2016;16:67
Fujiwara Y, Watanabe T, Muraki M, Yamagami H, Tanigawa T, Shiba M, et al. Association between chronic use of proton pump inhibitors and small-intestinal bacterial overgrowth assessed using lactulose hydrogen breath tests. Hepatogastroenterology. 2015;62:268–272
Imhann F, Bonder MJ, Vich Vila A, Fu J, Mujagic Z, Vork L, et al. Proton pump inhibitors affect the gut microbiome. Gut. 2016;65:740–748 (BMJ Publishing Group)
Otsuka T, Sugimoto M, Inoue R, Ohno M, Ban H, Nishida A, et al. Influence of potassium-competitive acid blocker on the gut microbiome of Helicobacter pylori-negative healthy individuals. Gut. 2017;66:1723–1725 (BMJ Publishing Group)
Chen Y, Yang F, Lu H, Wang B, Chen Y, Lei D, et al. Characterization of fecal microbial communities in patients with liver cirrhosis. Hepatology. 2011;54:562–572 (Wiley Subscription Services, Inc., A Wiley Company)
Inoue T, Nakayama J, Moriya K, Kawaratani H, Momoda R, Ito K, et al. Gut dysbiosis associated with hepatitis C virus infection. Clin Infect Dis. 2018;67:869–877
Duncan SH, Louis P, Flint HJ. Cultivable bacterial diversity from the human colon. Lett Appl Microbiol. 2007;44:343–350 (Wiley/Blackwell (10.1111))
Leone V, Gibbons SM, Martinez K, Hutchison AL, Huang EY, Cham CM, et al. Effects of diurnal variation of gut microbes and high-fat feeding on host circadian clock function and metabolism. Cell Host Microbe. 2015;17:681–689 (NIH Public Access)
Kakiyama G, Pandak WM, Gillevet PM, Hylemon PB, Heuman DM, Daita K, et al. Modulation of the fecal bile acid profile by gut microbiota in cirrhosis. J Hepatol. 2013;58:949–955 (NIH Public Access)
Zhang Z, Zhai H, Geng J, Yu R, Ren H, Fan H, et al. Large-scale survey of gut microbiota associated with MHE via 16S rRNA-based pyrosequencing. Am J Gastroenterol. 2013;108:1601–1611 (Nature Publishing Group)
Ahluwalia V, Betrapally NS, Hylemon PB, White MB, Gillevet PM, Unser AB, et al. Impaired gut-liver-brain axis in patients with cirrhosis. Sci Rep. 2016;6:26800 (Nature Publishing Group)
Bajaj JS. The role of microbiota in hepatic encephalopathy. Gut Microbes. 2014;5:397–403 (Taylor and Francis)
Bajaj JS, Ridlon JM, Hylemon PB, Thacker LR, Heuman DM, Smith S, et al. Linkage of gut microbiome with cognition in hepatic encephalopathy. Am J Physiol Gastrointest Liver Physiol. 2012;302:G168–G175 (American Physiological Society)
Takagi T, Naito Y, Inoue R, Kashiwagi S, Uchiyama K, Mizushima K, et al. The influence of long-term use of proton pump inhibitors on the gut microbiota: an age-sex-matched case-control study. J Clin Biochem Nutr. 2018;62:100–105 (The Society for Free Radical Research Japan)
Llorente C, Jepsen P, Inamine T, Wang L, Bluemel S, Wang HJ, et al. Gastric acid suppression promotes alcoholic liver disease by inducing overgrowth of intestinal Enterococcus. Nat Commun. 2017;8:1–14 (Springer, US)
Qin N, Yang F, Li A, Prifti E, Chen Y, Shao L, et al. Alterations of the human gut microbiome in liver cirrhosis. Nature. 2014;513:59–64 (Nature Publishing Group)
Bajaj JS, Heuman DM, Hylemon PB, Sanyal AJ, White MB, Monteith P, et al. The cirrhosis dysbiosis ratio defines changes in the gut microbiome associated with cirrhosis and its complications. J Hepatol. 2014;60:940–947 (NIH Public Access)
Aly AM, Adel A, El-Gendy AO, Essam TM, Aziz RK. Gut microbiome alterations in patients with stage 4 hepatitis C. Gut Pathog. 2016;8:42 (BioMed Central)
Acknowledgements
We thank Ms. Akina Ooishi at Department of Gastroenterology and Hepatology, Nagoya University Graduate School of Medicine for technical assistance to DNA isolate and 16S rRNA gene sequence.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This study was approved by the Research Ethics Committee of Nagoya University Hospital on August 30, 2016 (Protocol number 2015-0420). In accordance with the Declaration of Helsinki, written informed consent was obtained from all patients before registration. This study is registered in University Hospital Medical Information Network Clinical Trials Registry (UMIN ID: 000020269).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yamamoto, K., Ishigami, M., Honda, T. et al. Influence of proton pump inhibitors on microbiota in chronic liver disease patients. Hepatol Int 13, 234–244 (2019). https://doi.org/10.1007/s12072-019-09932-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12072-019-09932-9