Abstract
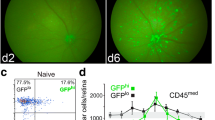
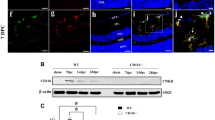
Neuroinflammatory diseases are characterized by blood-brain barrier disruption (BBB) and leukocyte infiltration. We investigated the involvement of monocyte recruitment in visual pathway damage provoked by primary optic neuritis (ON) induced by a microinjection of bacterial lipopolysaccharide (LPS) into the optic nerve from male Wistar rats. Increased Evans blue extravasation and cellularity were observed at 6 h post-LPS injection. In WT-GFPþ/WT chimeric rat optic nerves, the presence of GFP(+) neutrophils and GFP(+) monocytes, and in wild-type rat optic nerves, an increase in CD11b+CD45low and CD11b+CD45high cell number, were observed at 24 h post-LPS. Gamma-irradiation did not affect the increase in BBB permeability, but significantly lessened the decrease in pupil light reflex (PLR), and retinal ganglion cell (RGC) number induced by LPS. At 6 h post-LPS, an increase in chemokine (C-C motif) ligand 2 (CCL2) immunoreactivity co-localized with neutrophils (but not microglia/macrophages or astrocytes) was observed, while at 24 h post-injection, an increase in Iba-1-immunoreactivity and its co-localization with CCL2 became evident. The co-injection of LPS with bindarit (a CCL2 synthesis inhibitor) lessened the effect of LPS on PLR, and RGC loss. The treatment with etoposide or gadolinium chloride that significantly decreased peripheral monocyte (but not neutrophil or lymphocyte) percentage decreased the effect of LPS on PLR, and RGC number. Moreover, a negative correlation between PRL and monocyte (but not lymphocyte or neutrophil) percentage was observed at 7 days post-LPS. Taken together, these results support that monocytes are key players in the initial events that take place during primary ON.








Similar content being viewed by others
References
Toosy AT, Mason DF, Miller DH (2014) Optic neuritis. Lancet Neurol 13:83–99. https://doi.org/10.1016/S1474-4422(13)70259-X
Pau D, Al Zubidi N, Yalamanchili S, Plant GT, Lee AG (2011) Optic neuritis. Eye (Lond) 25:833–842. https://doi.org/10.1038/eye.2011.81
Kale N (2016) Optic neuritis as an early sign of multiple sclerosis. Eye Brain 8:195–202. https://doi.org/10.2147/EB.S54131
Aranda ML, Dorfman D, Sande PH, Rosenstein RE (2015) Experimental optic neuritis induced by the microinjection of lipopolysaccharide into the optic nerve. Exp Neurol 266:30–41. https://doi.org/10.1016/j.expneurol.2015.01.010
Jones KA, Maltby S, Plank MW, Kluge M, Nilsson M, Foster PS, Walker FR (2018) Peripheral immune cells infiltrate into sites of secondary neurodegeneration after ischemic stroke. Brain Behav Immun 67:299–307. https://doi.org/10.1016/j.bbi.2017.09.006
Kuo PC, Scofield BA, Yu IC, Chang FL, Ganea D, Yen JH (2016) Interferon-β modulates inflammatory response in cerebral ischemia. J Am Heart Assoc 5. doi: https://doi.org/10.1161/JAHA.115.002610.
Nguyen HX, Hooshmand MJ, Saiwai H, Maddox J, Salehi LA, Nishi RA, Salazar D, Uchida N et al (2017) Systemic neutrophil depletion modulates the migration and fate of transplanted human neural stem cells to rescue functional repair. J Neurosci 37:9269–9287. https://doi.org/10.1523/JNEUROSCI.2785-16.2017
Nourshargh S, Alon R (2014) Leukocyte migration into inflamed tissues. Immunity 41:694–707. https://doi.org/10.1016/j.immuni.2014.10.008
Aubé B, Lévesque SA, Paré A, Chamma É, Kébir H, Gorina R, Lécuyer MA, Alvarez JI et al (2014) Neutrophils mediate blood-spinal cord barrier disruption in demyelinating neuroinflammatory diseases. J Immunol 193:2438–2454. https://doi.org/10.4049/jimmunol.1400401
Hendriks JJ, Teunissen CE, de Vries HE, Dijkstra CD (2005) Macrophages and neurodegeneration. Brain Res Brain Res Rev 48:185–195. https://doi.org/10.1016/j.brainresrev.2004.12.008
Mishra MK, Yong VW (2016) Myeloid cells - targets of medication in multiple sclerosis. Nat Rev Neurol 12:539–551. https://doi.org/10.1038/nrneurol.2016.110
Shindler KS, Ventura E, Dutt M, Rostami A (2008) Inflammatory demyelination induces axonal injury and retinal ganglion cell apoptosis in experimental optic neuritis. Exp Eye Res 87:208–213. https://doi.org/10.1016/j.exer.2008.05.017
Adamus G, Brown L, Andrew S, Meza-Romero R, Burrows GG, Vandenbark AA (2012) Neuroprotective effects of recombinant T-cell receptor ligand in autoimmune optic neuritis in HLA-DR2 mice. Invest Ophthalmol Vis Sci 53:406–412. https://doi.org/10.1167/iovs.11-8419
Brambilla R, Dvoriantchikova G, Barakat D, Ivanov D, Bethea JR, Shestopalov VI (2012) Transgenic inhibition of astroglial NF-κB protects from optic nerve damage and retinal ganglion cell loss in experimental optic neuritis. J Neuroinflammation 9:213. https://doi.org/10.1186/1742-2094-9-213
Dorfman D, Aranda ML, González Fleitas MF, Chianelli MS, Fernandez DC, Sande PH, Rosenstein RE (2014) Environmental enrichment protects the retina from early diabetic damage in adult rats. PLoS One 9:e101829. https://doi.org/10.1371/journal.pone.0101829
Fernandez DC, Sande PH, Chianelli MS, Aldana Marcos HJ, Rosenstein RE (2011) Induction of ischemic tolerance protects the retina from diabetic retinopathy. Am J Pathol 178:2264–2274. https://doi.org/10.1016/j.ajpath.2011.01.040
Nakata W, Nakai Y, Yoshida T, Sato M, Hatano K, Nagahara A, Fujita K, Uemura M et al (2015) Bone marrow-derived cells contribute to regeneration of injured prostate epithelium and stroma. Prostate 75:806–814. https://doi.org/10.1002/pros.22962
Kim SY, Cheon SY, Kim EJ, Lee JH, Kam EH, Kim JM, Park M, Koo BN (2017) Isoflurane postconditioning inhibits tPA-induced matrix metalloproteinases activation after hypoxic injury via low-density lipoprotein receptor-related protein and extracellular signal-regulated kinase pathway. Neurochem Res 42:1533–1542. https://doi.org/10.1007/s11064-017-2211-2
Su EJ, Fredriksson L, Geyer M, Folestad E, Cale J, Andrae J, Gao Y, Pietras K et al (2008) Activation of PDGF-CC by tissue plasminogen activator impairs blood-brain barrier integrity during ischemic stroke. Nat Med 14:731–737. https://doi.org/10.1038/nm1787
Dong MX, Hu QC, Shen P, Pan JX, Wei YD, Liu YY, Ren YF, Liang ZH et al (2016) Recombinant tissue plasminogen activator induces neurological side effects independent on thrombolysis in mechanical animal models of focal cerebral infarction: a systematic review and meta-analysis. PLoS One 11:e0158848. https://doi.org/10.1371/journal.pone.0158848
Fredriksson L, Lawrence DA, Medcalf RL (2017) tPA modulation of the blood-brain barrier: a unifying explanation for the pleiotropic effects of tPA in the CNS. Semin Thromb Hemost 43:154–168. https://doi.org/10.1055/s-0036-1586229
Reijerkerk A, Kooij G, van der Pol SM, Leyen T, van Het Hof B, Couraud PO, Vivien D, Dijkstra CD et al (2008) Tissue-type plasminogen activator is a regulator of monocyte diapedesis through the brain endothelial barrier. J Immunol 181:3567–3574. https://doi.org/10.4049/jimmunol.181.5.3567
Yepes M, Sandkvist M, Moore EG, Bugge TH, Strickland DK, Lawrence DA (2003) Tissue-type plasminogen activator induces opening of the blood-brain barrier via the LDL receptor-related protein. J Clin Invest 112:1533–1540. https://doi.org/10.1172/JCI19212
Puliti M, von Hunolstein C, Bistoni F, Castronari R, Orefici G, Tissi L (2002) Role of macrophages in experimental group B streptococcal arthritis. Cell Microbiol 4:691–700. https://doi.org/10.1046/j.1462-5822.2002.00223.x
Pendino KJ, Meidhof TM, Heck DE, Laskin JD, Laskin DL (1995) Inhibition of macrophages with gadolinium chloride abrogates ozone-induced pulmonary injury and inflammatory mediator production. Am J Respir Cell Mol Biol 13:125–132. https://doi.org/10.1165/ajrcmb.13.2.7542894
Prinz M, Priller J (2017) The role of peripheral immune cells in the CNS in steady state and disease. Nat Neurosci 20:136–144. https://doi.org/10.1038/nn.4475
Hu P, Pollard J, Hunt N, Chan-Ling T (1998) Microvascular and cellular responses in the retina of rats with acute experimental allergic encephalomyelitis (EAE). Brain Pathol 8:487–498. https://doi.org/10.1111/j.1750-3639.1998.tb00169
Park HJ, Shin JY, Kim HN, Oh SH, Song SK, Lee PH (2015) Mesenchymal stem cells stabilize the blood-brain barrier through regulation of astrocytes. Stem Cell Res Ther 6:187. https://doi.org/10.1186/s13287-015-0180-4
Liu Y, Xie X, Xia LP, Lv H, Lou F, Ren Y, He ZY, Luo XG (2017) Peripheral immune tolerance alleviates the intracranial lipopolysaccharide injection-induced neuroinflammation and protects the dopaminergic neurons from neuroinflammation-related neurotoxicity. J Neuroinflammation 14:223. https://doi.org/10.1186/s12974-017-0994-3
Brown DA, Sawchenko PE (2007) Time course and distribution of inflammatory and neurodegenerative events suggest structural bases for the pathogenesis of experimental autoimmune encephalomyelitis. J Comp Neurol 502:236–260. https://doi.org/10.1002/cne.21307
Banks WA, Erickson MA (2010) The blood-brain barrier and immune function and dysfunction. Neurobiol Dis 37:26–32. https://doi.org/10.1016/j.nbd.2009.07.031
Jaworowicz DJ Jr, Korytko PJ, Singh Lakhman S, Boje KM (1998) Nitric oxide and prostaglandin E2 formation parallels blood-brain barrier disruption in an experimental rat model of bacterial meningitis. Brain Res Bull 46:541–546
Aranda ML, González Fleitas MF, De Laurentiis A, Keller Sarmiento MI, Chianelli M, Sande PH, Dorfman D, Rosenstein RE (2016) Neuroprotective effect of melatonin in experimental optic neuritis in rats. J Pineal Res 60:360–372. https://doi.org/10.1111/jpi.12318
Aranda ML, González Fleitas MF, Dieguez HH, Milne GA, Devouassoux JD, Keller Sarmiento MI, Chianelli M, Sande PH et al (2019) Therapeutic benefit of environmental enrichment on optic neuritis. Neuropharmacology 145:87–98. https://doi.org/10.1016/j.neuropharm.2017.12.017
Siao CJ, Fernandez SR, Tsirka SE (2003) Cell type-specific roles for tissue plasminogen activator released by neurons or microglia after excitotoxic injury. J Neurosci 23:3234–3242. https://doi.org/10.1523/JNEUROSCI.23-08-03234.2003
Villarán RF, de Pablos RM, Argüelles S, Espinosa-Oliva AM, Tomás-Camardiel M, Herrera AJ, Cano J, Machado A (2009) The intranigral injection of tissue plasminogen activator induced blood-brain barrier disruption, inflammatory process and degeneration of the dopaminergic system of the rat. Neurotoxicology 30:403–413. https://doi.org/10.1016/j.neuro.2009.02.011
Baron A, Montagne A, Cassé F, Launay S, Maubert E, Ali C, Vivien D (2010) NR2D-containing NMDA receptors mediate tissue plasminogen activator-promoted neuronal excitotoxicity. Cell Death Differ 17:860–871. https://doi.org/10.1038/cdd.2009.172
Tsirka SE, Gualandris A, Amaral DG, Strickland S (1995) Excitotoxin-induced neuronal degeneration and seizure are mediated by tissue plasminogen activator. Nature 377:340–344. https://doi.org/10.1038/377340a0
You Y, Gupta VK, Li JC, Klistorner A, Graham SL (2013) Optic neuropathies: characteristic features and mechanisms of retinal ganglion cell loss. Rev Neurosci 24:301–321. https://doi.org/10.1515/revneuro-2013-0003
Howell GR, Soto I, Zhu X, Ryan M, Macalinao DG, Sousa GL, Caddle LB, MacNicoll KH et al (2012) Radiation treatment inhibits monocyte entry into the optic nerve head and prevents neuronal damage in a mouse model of glaucoma. J Clin Invest 122:1246–1261. https://doi.org/10.1172/JCI61135
Mahad DJ, Ransohoff RM (2003) The role of (MCP-1) CCL2 and CCR2 in multiple sclerosis and experimental autoimmune encephalomyelitis (EAE). Semin Immunol 15:23–32. https://doi.org/10.1016/S1044-5323(02)00125-2
Hickman SE, El Khoury J (2010) Mechanisms of mononuclear phagocyte recruitment in Alzheimer’s disease. CNS Neurol Disord Drug Targets 9:168–173. https://doi.org/10.2174/187152710791011982
Babcock AA, Kuziel WA, Rivest S, Owens T (2003) Chemokine expression by glial cells directs leukocytes to sites of axonal injury in the CNS. J Neurosci 23:7922–7930. https://doi.org/10.1523/JNEUROSCI.23-21-07922.2003
Fuentes ME, Durham SK, Swerdel MR, Lewin AC, Barton DS, Megill JR, Bravo R, Lira SA (1995) Controlled recruitment of monocytes and macrophages to specific organs through transgenic expression of monocyte chemoattractant protein-1. J Immunol 155:5769–5776
Ge S, Shrestha B, Paul D, Keating C, Cone R, Guglielmotti A, Pachter JS (2012) The CCL2 synthesis inhibitor bindarit targets cells of the neurovascular unit, and suppresses experimental autoimmune encephalomyelitis. J Neuroinflammation 9:171. https://doi.org/10.1186/1742-2094-9-171
Yadav A, Saini V, Avora S (2010) MCP-1: chemoattractant with a role beyond immunity: a review. Clin Chim Acta 411:1570–1579. https://doi.org/10.1016/j.cca.2010.07.006
Zhang L, Ma P, Guan Q, Meng L, Su L, Wang L, Yuan B (2018) Effect of chemokine CC ligand 2 (CCL2) on α-synuclein-induced microglia proliferation and neuronal apoptosis. Mol Med Rep. https://doi.org/10.3892/mmr.2018.9468
Chang SL, Huang W, Mao X, Sarkar S (2017) NLRP12 inflammasome expression in the rat brain in response to LPS during morphine tolerance. Brain Sci 7(2):14. https://doi.org/10.3390/brainsci7020014
Davis RL, Stevens CW, Thomas Curtis J (2017) The opioid antagonist, β-funaltrexamine, inhibits lipopolysaccharide-induced neuroinflammation and reduces sickness behavior in mice. Physiol Behav 173:52–60. https://doi.org/10.1016/j.physbeh.2017.01.037
Le Thuc O, Cansell C, Bourourou M, Denis RG, Stobbe K, Devaux N, Guyon A, Cazareth J et al (2016) Central CCL2 signaling onto MCH neurons mediates metabolic and behavioral adaptation to inflammation. EMBO Rep 17:1738–1752. https://doi.org/10.15252/embr.201541499
Van Hove I, Lefevere E, De Groef L, Sergeys J, Salinas-Navarro M, Libert C, Vandenbroucke R, Moons L (2016) MMP-3 deficiency alleviates endotoxin-induced acute inflammation in the posterior eye segment. Int J Mol Sci 17. doi:https://doi.org/10.3390/ijms17111825
Arruda-Silva F, Bianchetto-Aguilera F, Gasperini S, Polletti S, Cosentino E, Tamassia N, Cassatella MA (2017) Human neutrophils produce CCL23 in response to various TLR-agonists and TNFα. Front Cell Infect Microbiol 7:176. https://doi.org/10.3389/fcimb.2017.00176
Huang DR, Wang J, Kivisakk P, Rollins BJ, Ransohoff RM (2001) Absence of monocyte chemoattractant protein 1 in mice leads to decreased local macrophage recruitment and antigen-specific T helper cell type 1 immune response in experimental autoimmune encephalomyelitis. J Exp Med 193:713–726. https://doi.org/10.1084/jem.193.6.713
Adamus G, Manczak M, Machnicki M (2001) Expression of CC chemokines and their receptors in the eye in autoimmune anterior uveitis associated with EAE. Invest Ophthalmol Vis Sci 42:2894–2903
Geissmann F, Jung S, Littman DR (2003) Blood monocytes consist of two principal subsets with distinct migratory properties. Immunity 19:71–82. https://doi.org/10.1016/S1074-7613(03)00174-2
Serbina NV, Pamer EG (2006) Monocyte emigration from bone marrow during bacterial infection requires signals mediated by chemokine receptor CCR2. Nat Immunol 7:311–317. https://doi.org/10.1038/ni1309
Tacke F, Alvarez D, Kaplan TJ, Jakubzick C, Spanbroek R, Llodra J, Garin A, Liu J et al (2007) Monocyte subsets differentially employ CCR2, CCR5, and CX3CR1 to accumulate within atherosclerotic plaques. J Clin Invest 117:185–194. https://doi.org/10.1172/JCI28549
Mirolo M, Fabbri M, Sironi M, Vecchi A, Guglielmotti A, Mangano G, Biondi G, Locati M et al (2008) Impact of the anti-inflammatory agent bindariton the chemokinome: selective inhibition of the monocytechemoattractant proteins. Eur Cytokine Netw 19:119–122. https://doi.org/10.1684/ecn.2008.0133
Bhatia M, Ramath RD, Chevali L, Guglielmotti A (2005) Treatment with bindarit, ablocker of MCP-1 synthesis, protects mice against acute pancreatitis. Am J Physiol Gastrointest Liver Physiol 288:G1259–G1265. https://doi.org/10.1152/ajpgi.00435
Grassia G, Maddaluno M, Guglielmotti A, Mangano G, Biondi G, Maffia P, Ialenti A (2009) The anti-inflammatory agent bindarit inhibits neointima formation in both rats and hyperlipodaemic mice. Cardiovasc Res 84:485–493. https://doi.org/10.1093/cvr/cvp238
Guglielmotti A, Aquilini L, D’Onofrio F, Rosignoli MT, Milanese C, Pinza M (1998) Bindarit prolongs survival and reduces renal damage in NZB/W lupus mice. Clin Exp Rheumatol 16:149–154
Guglielmotti A, D’Onofrio E, Coletta I, Aquilini L, Milanese C, Pinza M (2002) Amelioration of rat adjuvant arthritis by therapeutic treatment with bindarit, an inhibitor of MCP-1 and TNF-alpha production. Inflamm Res 51:252–258
Lin J, Zhu X, Chade A, Jordan KL, Lavi R, Daghini E, Gibson ME, Guglielmotti A et al (2009) Monocyte chemoattractant proteins mediate microvascular dysfunction in swine renovascular hypertension. Arterioscler Thromb Vasc Biol 29:1810–1816. https://doi.org/10.1161/ATVBAHA.109.190546
Huitinga N, van Rooijen CJ, De Groot CJ, Uitdehaag BM, Dijkstra CD (1990) Suppression of experimental allergic encephalomyelitis in Lewis rats after elimination of macrophages. J Exp Med 172:1025–1033. https://doi.org/10.1084/jem.172.4.1025
Funding
This work was supported by grants from the Agencia Nacional de Promoción Científica y Tecnológica (PICT 1563, PICT 2731), The University of Buenos Aires (20020130100564), and Consejo Nacional de Investigaciones Científicas y Técnicas (PIP 0707), Argentina. The funding organizations have no role in the design or conduct of this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
All animal procedures were in strict accordance with the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research. The ethic committee of the School of Medicine, University of Buenos Aires (Institutional Committee for the Care and Use of Laboratory Animals, (CICUAL)) approved this study, and all efforts were made to minimize animal suffering.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Fig. 9
Representative photomicrographs of consensual PLR (left panel) and Brn3a-immunostaining in flat-mounted retinas (right panel) from vehicle, etoposide- or gadolinium chloride (GdCl3)-treated animals, whose optic nerves were injected with vehicle or LPS (PNG 2605 kb)
Rights and permissions
About this article
Cite this article
Aranda, M.L., Guerrieri, D., Piñero, G. et al. Critical Role of Monocyte Recruitment in Optic Nerve Damage Induced by Experimental Optic Neuritis. Mol Neurobiol 56, 7458–7472 (2019). https://doi.org/10.1007/s12035-019-1608-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-019-1608-0