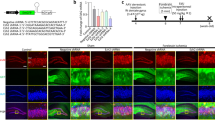
Abstract
Docosahexaenoic acid (DHA, 22:6n-3) is an essential omega-3 polyunsaturated fatty acid (PUFA) that is required for proper brain development and cerebral functions. While DHA deficiency in the brain was shown to be linked to the emergence of cerebral diseases, a dietary intake of omega-3 PUFA could prevent or attenuate neurologic disturbances linked with aging or neurodegenerative diseases. In this context, targeting the brain with DHA might offer great promise in developing new therapeutics for neurodegenerative diseases. We previously synthesized a stabilized form of DHA-containing lysophosphatidylcholine a major vector of DHA transportation to the brain, which is 1-acetyl,2-docoshexaenoyl-glycerophosphocholine, named AceDoPC®. Injection of AceDoPC® or DHA after experimental ischemic stroke showed that both molecules had neuroprotective effects but AceDoPC® was the most potent. This study aims to investigate the beneficial effects of DHA either unesterified or esterified within AceDoPC® on a model of neurogenesis in vitro, under physiological or pathological conditions. The effect of protectin DX (PDX, a double lipoxygenase product of DHA) was also tested. We cultured neural stem progenitor cells (NSPCs) derived from the adult mouse brain under normal or hypoxigenic (ischemic) conditions in vitro. Neurogenesis study of cell cultures with AceDoPC® showed enhanced neurogenesis compared to addition of unesterified DHA, PDX, or vehicle control, especially under pathological conditions. Our studies of the potential mechanisms involved in neuroprotection hinted that AceDoPC® neuroprotective and regenerative effects might be due in part to its anti-oxidative effects. These results indicate the potential for novel therapeutics against stroke that target the brain.







Similar content being viewed by others
References
Thies F, Garry JMC, Yaqoob P, Rerkasem K, Williams J, Shearman CP, Gallagher PJ, Calder PC, et al. (2003) Mechanisms of disease association of n-3 polyunsaturated fatty acids with stability of atherosclerotic plaques: a randomised controlled trial. Lancet 361:477–485 . doi: https://doi.org/10.1016/S0140-6736(03)12468-3
Colussi G, Catena C, Dialti V, Mos L, Sechi AL (2014) Effects of the consumption of fish meals on the carotid intimamedia thickness in patients with hypertension: a prospective study. J Atheroscler Thromb 941–956 . doi: DN/JST.JSTAGE/jat/22921 [pii]
Hashimoto M, Shahdat HM, Katakura M, Tanabe Y, Gamoh S, Miwa K, Shimada T, Shido O (2009) Effects of docosahexaenoic acid on in vitro amyloid beta peptide 25-35 fibrillation. Biochim Biophys Acta 1791:289–296. https://doi.org/10.1016/j.bbalip.2009.01.012
Chauveau F, Cho T-H, Perez M, Guichardant M, Riou A, Aguettaz P, Picq M, Lagarde M, et al. (2011) Brain-targeting form of docosahexaenoic acid for experimental stroke treatment: MRI evaluation and anti-oxidant impact. Curr Neurovasc Res 8:95–102 . doi: https://doi.org/10.2174/156720211795495349
Young G, Conquer J (2005) Omega-3 fatty acids and neuropsychiatric disorders. Reprod Nutr Dev 45:1–28
Peet M, Stokes C (2005) Omega-3 fatty acids in the treatment of psychiatric disorders. Drugs 65:1051–1059
Gao X, Chen H, Fung TT, Logroscino G, Schwarzschild MA, Hu FB, Ascherio A (2007) Prospective study of dietary pattern and risk of Parkinson disease. Am J Clin Nutr 86:1486–1494
Belayev L, Marcheselli VL, Khoutorova L, Rodriguez de Turco EB, Busto R, Ginsberg MD, Bazan NG (2005) Docosahexaenoic acid complexed to albumin elicits high-grade ischemic neuroprotection. Stroke 36:118–123. https://doi.org/10.1161/01.STR.0000149620.74770.2e
Belayev L, Khoutorova L, Atkins KD, Bazan NG (2009) Robust docosahexaenoic acid-mediated neuroprotection in a rat model of transient, focal cerebral ischemia. Stroke 40:3121–3126. https://doi.org/10.1161/STROKEAHA.109.555979
Hong S-H, Belayev L, Khoutorova L, Obenaus A, Bazan NG (2014) Docosahexaenoic acid confers enduring neuroprotection in experimental stroke. J Neurol Sci 338:135–141. https://doi.org/10.1016/j.pestbp.2011.02.012.Investigations
Calon F, Cole G (2007) Neuroprotective action of omega-3 polyunsaturated fatty acids against neurodegenerative diseases: evidence from animal studies. Prostaglandins Leukot Essent Fatty Acids 77:287–293. https://doi.org/10.1016/j.plefa.2007.10.019
Brenna JT, Salem N, Sinclair AJ, Cunnane SC (2009) Alpha-linolenic acid supplementation and conversion to n-3 long-chain polyunsaturated fatty acids in humans. Prostaglandins Leukot Essent Fat Acids 80:85–91. https://doi.org/10.1016/j.plefa.2009.01.004
Thies F, Pillon C, Moliere P, Lagarde M, Lecerf J (1994) Preferential incorporation of sn-2 lysoPC DHA over unesterified DHA in the young rat brain. Am J Phys 267:R1273–R1279
Bernoud N, Fenart L, Molière P, Dehouck MP, Lagarde M, Cecchelli R, Lecerf J (1999) Preferential transfer of 2-docosahexaenoyl-1-lysophosphatidylcholine through an in vitro blood-brain barrier over unesterified docosahexaenoic acid. J Neurochem 72:338–345. https://doi.org/10.1046/j.1471-4159.1999.0720338.x
Lagarde M, Bernoud N, Thies F, Brossard N, Lemaitre-Delaunay D, Croset M, Lecerf J (2001) Lysophosphatidylcholine as a carrier of docosahexaenoic acid to target tissues. J Mol Neurosci 88:173–177
Nguyen LN, Ma D, Shui G, Wong P, Cazenave-Gassiot A, Zhang X, Wenk MR, Goh ELK, et al. (2014) Mfsd2a is a transporter for the essential omega-3 fatty acid docosahexaenoic acid. Nature 509:503–506 . doi: https://doi.org/10.1038/nature13241
Quek DQY, Nguyen LN, Fan H, Silver DL (2016) Structural insights into the transport mechanism of the human sodium-dependent Lysophosphatidylcholine transporter MFSD2A. J Biol Chem 291:9383–9394. https://doi.org/10.1074/jbc.M116.721035
Sugasini D, Thomas R, Yalagala PCR, Tai LM, Subbaiah PV (2017) Dietary docosahexaenoic acid (DHA) as lysophosphatidylcholine, but not as free acid, enriches brain DHA and improves memory in adult mice. Sci Rep 7:1–11. https://doi.org/10.1038/s41598-017-11766-0
Hachem M, Géloën A, Lo Van A, Fourmaux B, Fenart L, Gosselet F, Da Silva P, Breton G, et al. (2016) Efficient docosahexaenoic acid uptake by the brain from a structured phospholipid. Mol Neurobiol 53:3205–3215. doi: https://doi.org/10.1007/s12035-015-9228-9
Lagarde M, Hachem M, Bernoud-Hubac N, Picq M, Véricel E, Guichardant M (2015) Biological properties of a DHA-containing structured phospholipid (AceDoPC) to target the brain. Prostaglandins Leukot Essent Fat Acids 92:63–65. https://doi.org/10.1016/j.plefa.2014.01.005
Goldberg M, Choi W (1993) Combined oxygen and glucose deprivation in cortical cell culture: calcium-dependent and calcium-independent mechanisms of neuronal injury. J Neurosci 13:3510–3524
Zhang M, Wang S, Mao L, Leak RK, Shi Y, Zhang W, Hu X, Sun B, et al. (2014) Omega-3 fatty acids protect the brain against ischemic injury by activating Nrf2 and upregulating heme oxygenase 1. J Neurosci 34:1903–1915 . doi: https://doi.org/10.1523/JNEUROSCI.4043-13.2014
Lo Van A, Sakayori N, Hachem M, Belkouch M, Picq M, Lagarde M, Osumi N, Bernoud-Hubac N (2016) Mechanisms of DHA transport to the brain and potential therapy to neurodegenerative diseases. Biochimie 130:163–167. https://doi.org/10.1016/j.biochi.2016.07.011
Bannenberg G, Serhan CN (2010) Specialized pro-resolving lipid mediators in the inflammatory response: an update. Biochim Biophys Acta 1801:1260–1273. https://doi.org/10.1016/j.bbalip.2010.08.002
Spite M, Clària J, Serhan CN (2014) Resolvins, specialized proresolving lipid mediators, and their potential roles in metabolic diseases. Cell Metab 19:21–36. https://doi.org/10.1016/j.cmet.2013.10.006
Chen P, Fenet B, Michaud S, Tomczyk N, Véricel E, Lagarde M, Guichardant M (2009) Full characterization of PDX, a neuroprotectin/protectin D1 isomer, which inhibits blood platelet aggregation. FEBS Lett 583:3478–3484. https://doi.org/10.1016/j.febslet.2009.10.004
Bazan NG, Marcheselli VL, Cole-Edwards K (2005) Brain response to injury and neurodegeneration: endogenous neuroprotective signaling. Ann N Y Acad Sci 1053:137–147. https://doi.org/10.1196/annals.1344.011
Serhan CN, Gotlinger K, Hong S, Lu Y, Siegelman J, Baer T, Yang R, Colgan SP, et al. (2006) Anti-inflammatory actions of neuroprotectin D1/protectin D1 and its natural stereoisomers: assignments of dihydroxy-containing docosatrienes. J Immunol 176:1848–1859
Levy BD (2010) Resolvins and protectins: natural pharmacophores for resolution biology. Prostaglandins Leukot Essent Fatty Acids 82:327–332. https://doi.org/10.1016/j.plefa.2010.02.003
Chen P, Véricel E, Lagarde M, Guichardant M (2011) Poxytrins, a class of oxygenated products from polyunsaturated fatty acids, potently inhibit blood platelet aggregation. FASEB J 25:382–388. https://doi.org/10.1096/fj.10-161836
Liu M, Boussetta T, Makni-Maalej K, Fay M, Driss F, El-Benna J, Lagarde M, Guichardant M (2014) Protectin DX, a double lipoxygenase product of DHA, inhibits both ROS production in human neutrophils and cyclooxygenase activities. Lipids 49:49–57. https://doi.org/10.1007/s11745-013-3863-6
Zhu M, Wang X, Hjorth E, Colas RA, Schroeder L, Granholm A-C, Serhan CN, Schultzberg M (2016) Pro-resolving lipid mediators improve neuronal survival and increase Aβ42 phagocytosis. Mol Neurobiol 53:2733–2749. https://doi.org/10.1159/000381474.A
Jung TW, Kim HC, Abd El-Aty AM, Jeong JH (2017) Protectin DX ameliorates palmitate-or high-fat diet-induced insulin resistance and inflammation through an AMPK-PPARα-dependent pathway in mice. Sci Rep 7:1–9. https://doi.org/10.1038/s41598-017-01603-9
Holte LL, Separovic F, Gawrisch K (1996) Nuclear magnetic resonance investigation of hydrocarbon chain packing in bilayers of polyunsaturated phospholipids. Lipids 31:S199–S203
Stubbs CD, Smith AD (1984) The modification of mammalian membrane polyunsaturated fatty acid composition in relation to membrane fluidity and function. Biochim Biophys Acta 779:89–137
Hashimoto M, Hossain S, Shido O (2006) Docosahexaenoic acid but not eicosapentaenoic acid withstands dietary cholesterol-induced decreases in platelet membrane fluidity. Mol Cell Biochem 293:1–8
Yehuda S, Rabinovitz S, Mostofsky DI (1999) Essential fatty acids are mediators of brain biochemistry and cognitive functions. J Neurosci Res 56:565–570
Horrocks LA, Farooqui AA (2004) Docosahexaenoic acid in the diet: its importance in maintenance and restoration of neural membrane function. Prostaglandins Leukot Essent Fatty Acids 70:361–372
Sakayori N, Maekawa M, Numayama-Tsuruta K, Katura T, Moriya T, Osumi N (2011) Distinctive effects of arachidonic acid and docosahexaenoic acid on neural stem/progenitor cells. Genes Cells 16:778–790. https://doi.org/10.1111/j.1365-2443.2011.01527.x
Ming G, Song H (2011) Adult neurogenesis in the mammalian brain: significant answers and significant questions. Neuron 70:687–702. https://doi.org/10.1016/j.neuron.2011.05.001.Adult
Shivraj Sohur U, Emsley JG, Mitchell BD, Macklis JD (2006) Adult neurogenesis and cellular brain repair with neural progenitors, precursors and stem cells. Philos Trans R Soc B Biol Sci 361:1477–1497. https://doi.org/10.1098/rstb.2006.1887
Kawakita E, Hashimoto M, Shido O (2006) Docosahexaenoic acid promotes neurogenesis in vitro and in vivo. Neuroscience 139:991–997. https://doi.org/10.1016/j.neuroscience.2006.01.021
He C, Qu X, Cui L, Wang J, Kang JX (2009) Improved spatial learning performance of fat-1 mice is associated with enhanced neurogenesis and neuritogenesis by docosahexaenoic acid. Proc Natl Acad Sci U S A 106:11370–11375. https://doi.org/10.1073/pnas.0904835106
Reynolds BA, Weiss S (1992) Nervous system generation of neurons and astrocytes from isolated cells of the adult mammalian central nervous system. Science 255:1707–1710
Sakayori N, Kikkawa T, Tokuda H, Kiryu E, Yoshizaki K, Kawashima H, Yamada T, Arai H et al. (2016) Maternal dietary imbalance between omega-6 and omega-3 polyunsaturated fatty acids impairs neocortical development via epoxy metabolites. Stem Cells 34:470–482 . doi: https://doi.org/10.1002/stem.2246
Katakura M, Hashimoto M, Shahdat HM, Gamoh S, Okui T, Matsuzaki K, Shido O (2009) Docosahexaenoic acid promotes neuronal differentiation by regulating basic helix-loop-helix transcription factors and cell cycle in neural stem cells. Neuroscience 160:651–660. https://doi.org/10.1016/j.neuroscience.2009.02.057
Song JH, Fujimoto K, Miyazawa T (2000) Polyunsaturated (n-3) fatty acids susceptible to peroxidation are increased in plasma and tissue lipids of rats fed docosahexaenoic acid-containing oils. J Nutr 130:3028–3033
Mukherjee PK, Marcheselli VL, Serhan CN, Bazan NG (2004) Neuroprotectin D1: a docosahexaenoic acid-derived docosatriene protects human retinal pigment epithelial cells from oxidative stress. Proc Natl Acad Sci U S A 101:8491–8496. https://doi.org/10.1073/pnas.0402531101
Hiratsuka S, Ishihara K, Kitagawa T, Wada S, Yokogoshi H (2008) Effect of dietary docosahexaenoic acid connecting phospholipids on the lipid peroxidation of the brain in mice. J Nutr Sci Vitaminol (Tokyo) 54:501–506. https://doi.org/10.3177/jnsv.54.501
Song JH, Miyazawa T (2001) Enhanced level of n-3 fatty acid in membrane phospholipids induces lipid peroxidation in rats fed dietary docosahexaenoic acid oil. Atherosclerosis 155:9–18. https://doi.org/10.1016/S0021-9150(00)00523-2
Lukiw WJ, Cui J-G, Marcheselli VL, Bodker M, Botkjaer A, Gotlinger K, Serhan CN, Bazan NG (2005) A role for docosahexaenoic acid-derived neuroprotectin D1 in neural cell survival and Alzheimer disease. J Clin Invest 115:2774–2783. https://doi.org/10.1172/JCI25420
Marcheselli VL, Hong S, Lukiw WJ, Tian XH, Gronert K, Musto A, Hardy M, Gimenez JM, et al. (2003) Novel docosanoids inhibit brain ischemia-reperfusion-mediated leukocyte infiltration and pro-inflammatory gene expression. J Biol Chem 278:43807–43817 . doi: https://doi.org/10.1074/jbc.M305841200
Begum G, Kintner D, Liu Y, Cramer SW, Sun D (2012) DHA inhibits ER ca 2+ release and ER stress in astrocytes following in vitro ischemia. J Neurochem 120:622–630. https://doi.org/10.1111/j.1471-4159.2011.07606.x
Katakura M, Hashimoto M, Okui T (2013) Omega-3 polyunsaturated fatty acids enhance neuronal differentiation in cultured rat neural stem cells. Stem Cells Int 2013:1–9. https://doi.org/10.1155/2013/490476
Qian X, Shen Q, Goderie SK, He W, Capela A, Davis AA, Temple S (2000) Timing of CNS cell generation. Neuron 28:69–80. https://doi.org/10.1016/S0896-6273(00)00086-6
Okada Y, Matsumoto A, Shimazaki T, Enoki R, Koizumi A, Ishii S, Itoyama Y, Sobue G, et al. (2008) Spatiotemporal recapitulation of central nervous system development by murine embryonic stem cell-derived neural stem/progenitor cells. Stem Cells 26:3086–3098 . doi: https://doi.org/10.1634/stemcells.2008-0293
Martinez M (1992) Tissue levels of polyunsaturated fatty acids during early human development. J Pediatr 120:129–138. https://doi.org/10.1016/S0022-3476(05)81247-8
Clandinin MT, Chappell JE, Leong S, Heim T, Swyer PR, Chance GW (1980) Intrauterine fatty acid accretion rates in human brain: implications for fatty acid requirements. Early Hum Dev 4:121–129. https://doi.org/10.1016/0378-3782(80)90015-8
Lauritzen L, Hansen HS, Jørgensen MH, Michaelsen KF (2001) The essentiality of long chain n-3 fatty acids in relation to development and function of the brain and retina. Prog Lipid Res 40:1–94. https://doi.org/10.1016/S0163-7827(00)00017-5
Hattiangady B, Shuai B, Cai J, Coksaygan T, Rao MS, Shetty AK (2007) Increased dentate neurogenesis after grafting of glial restricted progenitors or neural stem cells in the aging hippocampus. Stem Cells 25:2104–2117. https://doi.org/10.1634/stemcells.2006-0726
Ma DK, Ming GL, Song H (2005) Glial influences on neural stem cell development: cellular niches for adult neurogenesis. Curr Opin Neurobiol 15:514–520. https://doi.org/10.1016/j.conb.2005.08.003
Sairanen T, Ristimaki A, Paetau A, Kaste M, Lindsberg PJ, Karjalainen-lindsberg M (1998) Cyclooxygenase-2 is induced globally in infarcted human Brian. Ann Neurol 738–747
Tomimoto H, Shibata M, Ihara M, Akiguchi I, Ohtani R, Budka H (2002) A comparative study on the expression of cyclooxygenase and 5-lipoxygenase during cerebral ischemia in humans. Acta Neuropathol 104:601–607. https://doi.org/10.1007/s00401-002-0590-0
Ohtsuki T, Matsumoto M, Hayashi Y, Yamamoto K, Kitagawa K, Ogawa S, Yamamoto S, Kamada T (1995) Reperfusion induces 5-lipoxygenase translocation and leukotriene C4 production in ischemic brain. Am J Physiol Heart Circ Physiol 268:H1249–H1257
Ciceri P, Rabuffetti M, Monopoli A, Nicosia S (2001) Production of leukotrienes in a model of focal cerebral ischaemia in the rat. Br J Pharmacol 133:1323–1329. https://doi.org/10.1038/sj.bjp.0704189
Usui M, Asano T, Takakura K, Model O, Instruments M, Ka-Y (1987) Identification and quantitative analysis of hydroxy-eicosatetraenoic acids in rat brains exposed to regional ischemia. Stroke 18:490–494
Sun L, Xu Y-W, Han J, Liang H, Wang N, Cheng Y (2015) 12/15-lipoxygenase metabolites of arachidonic acid activate PPARγ: A possible neuroprotective effect in ischemic brain. J Lipid Res 56:502–514. https://doi.org/10.1194/jlr.M053058
Yagami T, Koma H, Yamamoto Y (2016) Pathophysiological roles of cyclooxygenases and prostaglandins in the central nervous system. Mol Neurobiol 53:4754–4771. https://doi.org/10.1007/s12035-015-9355-3
Lin H-J, Chen S-T, Wu H-Y, Hsu H-C, Chen M-F, Lee Y-T, Wu K-Y, Chien K-L (2015) Urinary biomarkers of oxidative and nitrosative stress and the risk for incident stroke: a nested case-control study from a community-based cohort. Int J Cardiol 183:214–220. https://doi.org/10.1016/j.ijcard.2015.01.043
Tyurin VA, Tyurina YY, Borisenko GG, Sokolova T V, Ritov VB, Quinn PJ, Rose M, Kochanek P, et al. (2000) Oxidative stress following traumatic brain injury in rats: quantitation of biomarkers and detection of free radical intermediates. J Neurochem 75:2178–2189 . doi: https://doi.org/10.1046/j.1471-4159.2000.0752178.x
Brault S, Martinez-Bermudez AK, Marrache AM, Gobeil F, Hou X, Beauchamp M, Quiniou C, Almazan G, et al. (2003) Selective neuromicrovascular endothelial cell death by 8-iso-prostaglandin f2: possible role in ischemic brain injury. Stroke 34:776–782 . doi: https://doi.org/10.1161/01.STR.0000055763.76479.E6
Yokose J, Ishizuka T, Yoshida T, Aoki J, Koyanagi Y, Yawo H (2011) Lineage analysis of newly generated neurons in organotypic culture of rat hippocampus. Neurosci Res 69:223–233. https://doi.org/10.1016/j.neures.2010.11.010
Dayer AG, Ford AA, Cleaver KM, Yassaee M, Cameron HA (2003) Short-term and long-term survival of new neurons in the rat dentate gyrus. J Comp Neurol 460:563–572. https://doi.org/10.1002/cne.10675
Sasaki T, Kitagawa K, Yagita Y, Sugiura S, Omura-Matsuoka E, Tanaka S, Matsushita K, Okano H, et al. (2006) Bcl2 enhances survival of newborn neurons in the normal and ischemic hippocampus. J Neurosci Res 84:1187–1196 . doi: https://doi.org/10.1002/jnr.21036
Pan HC, Kao TK, Ou YC, Yang DY, Yen YJ, Wang CC, Chuang YH, Liao SL, et al. (2009) Protective effect of docosahexaenoic acid against brain injury in ischemic rats. J Nutr Biochem 20:715–725 . doi: https://doi.org/10.1016/j.jnutbio.2008.06.014
Acknowledgments
This work was supported by the Japan Society for the Promotion of Science and Core-to-Core Program, LISA Carnot institute, National Institute of Applied Sciences-Lyon and the French Ministry of Education and Research. We thank the Functional Lipidomics Platform for lipid analyses.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(DOCX 1233 kb)
Rights and permissions
About this article
Cite this article
Lo Van, A., Sakayori, N., Hachem, M. et al. Targeting the Brain with a Neuroprotective Omega-3 Fatty Acid to Enhance Neurogenesis in Hypoxic Condition in Culture. Mol Neurobiol 56, 986–999 (2019). https://doi.org/10.1007/s12035-018-1139-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-018-1139-0