Abstract
Background
Traumatic brain injury (TBI) is the leading cause of death and long-term disability among injured children. Early feeding has been shown to improve outcomes in adults, with some similar evidence in children with severe TBI. We aimed to examine the current practice of initiation of enteral nutrition in children with TBI and to evaluate the risk factors associated with delayed initiation of enteral nutrition.
Methods
This retrospective, multicenter study used the Pediatric Trauma Assessment and Management Database including all children with head trauma discharged from five pediatric intensive care units (PICU) at pediatric trauma centers between January 1, 2013 and December 31, 2013. We compared demographics, injury and procedure data, time to initiation of nutrition, and injury and illness severity scores between patients who received enteral nutrition early (≤ 48 h) and late (> 48 h). Fisher’s exact and Mann–Whitney U tests compared discrete and continuous variables. Univariate and multivariable analyses evaluated variables associated with delayed initiation of feeding. Outcomes of interest included mortality, complications, ventilator days, hospital and ICU length of stay, and functional status at ICU discharge.
Results
In the 416 patients in the study, the overall mortality was 2.6%. The majority of patients (83%; range 69–88% between five sites, p = 0.0008) received enteral nutrition within 48 h of PICU admission. Lower Glasgow Coma Scale scores and higher Injury Severity Score (ISS) were independently associated with delayed initiation of enteral nutrition. Delayed enteral nutrition was independently associated with worse functional status at PICU discharge (p = 0.02) but was not associated with mortality or increased length of stay.
Conclusions
Children with severe TBI and higher ISS were more likely to have delayed initiation of enteral nutrition. Delayed enteral nutrition was an independent risk factor for worse functional status at ICU discharge for the entire cohort, but not for the severe TBI group.
Similar content being viewed by others
Introduction
Traumatic brain injury (TBI) is the leading cause of death and long-term disability among injured children [1]. There has been a coordinated effort to develop evidence-based guidelines for the management of adults and children with severe TBI over the last decade [2]. Earlier feeding has been shown to improve outcomes in adults with severe TBI [3, 4], and there is limited evidence that earlier enteral nutrition also improves outcomes in critically ill and severely brain-injured children [5,6,7,8,9]. The aim of our study is to examine the current practice of initiation of enteral nutrition in children with TBI and to evaluate the risk factors associated with delayed initiation of enteral nutrition.
Methods
This was a retrospective, multicenter study using the Pediatric Trauma Assessment and Management (PTAM) Database. This database was created by merging institutional trauma registries and the Virtual Pediatric Systems, LLC database (VPS) for all children discharged from five participating pediatric intensive care units (PICU) at pediatric trauma centers between January 1, 2013 and December 31, 2013 with International Classification of Disease, 9th edition—Clinical Modification (ICD9-CM) codes 800–959.9 and/or identified as a trauma patient by the VPS participant profile page. Children 0–18 years of age with head injury were included. Head injury was defined by ICD9-CM diagnosis codes 800–804 or 850.0–859.9 to capture all concussion, intracranial injury, and skull fractures. All children had to have a head Abbreviated Injury Score of two or greater to exclude patients without a significant head injury who may have a low Glasgow Coma Scale (GCS) score for other reasons (i.e., medications, etc.). Patients who died < 48 h from admission and those with incomplete information on enteral nutrition were excluded. The PTAM database had institutional review board approval from all institutions involved, and all data were de-identified from the patient and study site.
Data Source
Data collected included site, age, sex, race, weight, height, enteral feeds (yes/no, if yes, then also date/time of initiation), parenteral feeds (yes/no, if yes, then also date/time of initiation), PICU admission date, PICU discharge date, hospital discharge date, emergency department (ED) and admit GCS scores, injury and illness severity scores (i.e., Injury Severity Score (ISS), Pediatric Risk of Mortality (PRISM3), Pediatric Index of Mortality (PIM2)), ICD9-CM primary external cause of injury codes (E-codes), ICD9-CM diagnoses and procedure codes, duration of mechanical and noninvasive ventilation, and initiation of a bowel regimen.
Definitions
GCS scores 13–15, 9–12, and < 9 were used to classify severity of TBI as mild, moderate, and severe, respectively. If both the ED and PICU admission GCS scores were available, the lowest was used to classify the severity of TBI. Early initiation of enteral feeds was defined as initiation at or before 48 h from PICU admission. We chose this cutoff based on the Trauma Quality Improvement Program [10] recommendation to initiate enteral nutrition within 24–48 h. Delayed initiation was defined as initiation more than 48 h from PICU admission. Initiation of feeds was captured as any oral diet or tube feeds (at any rate). In the case of tube feeds, these may be given by a tube directly into the patient’s gastrointestinal system (stomach, small bowel). No measure of advancement or lack thereof was captured. Diagnoses and procedure categories were categorized for specific covariates of interest based on a priori hypotheses that they may affect enteral nutrition initiation timing. These included abdominal or pelvic injury (ICD9-CM codes 863–868), abdominal procedures (ICD9-CM procedure codes 42–54), and child abuse (ICD9-CM codes 995.50, 995.54, 995.55 and 995.59). A bowel regimen was defined as any medication given to facilitate or stimulate a bowel movement and/or prevent or treat constipation. To qualify as a bowel regimen, the medications had to be ordered and given on a scheduled basis, at least once per day.
Outcomes
Outcomes included mortality, hospital complications, ICU and hospital length of stay, ventilator days, and functional status at ICU discharge. Hospital complications included infections, constipation, and abdominal compartment syndrome. Hospital-acquired infections included pneumonia, urinary tract infections, surgical site infections, central line-associated blood stream infections, ventilator-associated pneumonia, and clostridium difficile infection. The Pediatric Overall Performance Category (POPC) and Pediatric Cerebral Performance Category (PCPC) scores were used to assess functional status at ICU discharge. Developed in the early 1990s, the POPC and PCPC scales have been validated to assess the outcomes at PICU discharge with excellent interrater reliability [11] and with good correlation with long-term functional status [12]. The scores are calculated at baseline to represent pre-injury status and at discharge from the PICU. For this study, the POPC and PCPC scores were dichotomized by combining categories 1 and 2 (normal and mild disability) and categories 3–5 (moderate disability to coma/vegetative state). This was modeled after similar studies in order to minimize the risk of type II error which can occur when comparing outcomes with categories containing very few patients [3, 7].
Statistical Analysis
Percentiles and counts were reported for categorical data. Chi-square or Fisher’s exact tests were used for comparisons. Median and interquartile range (IQR) were reported for continuous variables due to the asymmetric nature of the data, and a Mann–Whitney test was used to compare the data. The main outcome was the time to initiation of enteral nutrition compared between patients with admission GCS < 9 and ≥ 9. Also, of interest was early versus delayed enteral nutrition (≤ 48 h and > 48 h). Stepwise logistic regression with alpha = 0.10 was created for the main outcome of interest, delayed feeding, using the variables hypothesized to be associated with early feeding, namely age, weight, diagnosis of abdominal/pelvic injury, abdominal procedure, child abuse diagnosis, use of mechanical ventilation, intracranial pressure (ICP) monitoring, injury and ISS, PRISM3, PIM2, GCS, and pupillary reaction. For other outcomes listed previously, stepwise logistic or linear regression was used adjusting the final models for the variables above and for delayed feeding.
Results
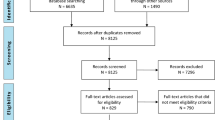
Of the 692 patients in the PTAM database, 416 patients were included in the study (Fig. 1). The overall mortality was 2.6% (11/416), all of whom had a GCS < 9 (i.e., mortality was 10% [11/107] in the GCS < 9 group). The majority of patients (83%) received enteral nutrition within 48 h of PICU admission. Demographic and anthropometric factors were not different between patients who were fed early and late (Table 1). The proportion of patients receiving enteral nutrition within 48 h of PICU admission varied from 70 to 88% between the five sites (p = 0.0008). Patients who had delayed initiation of enteral feedings had worse GCS scores, longer periods of mechanical ventilation and higher injury and illness severity scores. These patients also had more abdominopelvic injury diagnoses and procedures. A bowel regimen was more frequently ordered in patients with delayed initiation of enteral nutrition (Table 1). A sub-analysis of the 58 patients who were excluded due to incomplete feeding information showed them to be similar to the early feeding group with respect to GCS, ISS, and mechanical ventilation.
A total of 107 (26%) of the total population had severe TBI (GCS < 9). Initiation of enteral feeds was more often delayed in patients with severe TBI compared to mild or moderate TBI; 52% of patients with severe TBI had delayed feeding, compared to 4% with mild or moderate TBI, (p < 0.0001). Time to feeding initiation was significantly different between the two groups, with a median time to initiation of 47.4 h (IQR 21.9–73) among patients with severe TBI versus 10.7 h (3.9–18.5) among patients with mild or moderate TBI (p < 0.0001) (Table 2). A small number (4%) of patients received parenteral nutrition (15% vs 0.6% in GCS < 9 and GCS ≥ 9 groups, respectively, p < 0.0001).
The following factors were found to be associated with delayed initiation of enteral nutrition on univariate analysis: abdominal/pelvic injury diagnoses, abdominal procedures, mechanical ventilation, ICP monitoring, higher injury and illness severity scores, lower GCS score, and non-reactive pupils (Table 3). Lower GCS and higher ISS remained independently associated with delayed initiation of enteral nutrition in a multivariable analysis. Though abdominal/pelvic injury diagnoses were highly correlated with ISS, these injuries were not independently associated with delayed enteral feeding after multivariable analysis. Demographic and anthropometric parameters were not associated with timing of initiation of enteral nutrition. In the sub-analysis of patients with GCS < 9, only a higher ISS was independently associated with delayed initiation of enteral nutrition (Supplemental Table 1).
Patients with early initiation of enteral feeds, overall, had better outcomes compared to patients with delayed initiation. Among the GCS < 9 group, in univariate analyses, patients who received early feeding had less disability at discharge (lower POPC and PCPC scores) and shorter PICU length of stay (Table 4). Early feeding was also not associated with increased complication rates such as infections, abdominal compartment syndrome, and constipation. After adjusting for age, site, abdominal injury diagnosis, abdominal procedures, GCS, mechanical ventilation, injury and illness severity scores, and pupillary reactivity, however, delayed enteral nutrition was independently associated with worse functional status (POPC score) at discharge (p = 0.02) (Supplemental Table 2). In the subgroup of patients with GCS < 9, delayed initiation of enteral nutrition was not independently associated with any of the outcomes of interest (Supplemental Table 3).
A separate analysis of the data using 72 h as cutoff for early enteral nutrition yielded similar results for outcomes and factors associated with delayed enteral nutrition (data not shown). We also analyzed patients with moderate and severe TBI as a single cohort. Although we did not find any outcome benefit to early feeding, there was a trend toward significance for delayed feeding being independently associated with increased mortality (OR = 5.7 (95% CI 0.9–35.7), p = 0.06).
Discussion
In this retrospective multi-institutional study of five pediatric trauma centers evaluating timing of initiation of enteral feeds among injured children with TBI, we were able to describe feeding initiation, factors associated with delayed initiation of enteral nutrition, and outcomes associated with delayed feeding. We found that overall, most patients had enteral nutrition initiated within 48 h of admission. A lower GCS score and higher ISS were independently associated with delayed initiation of enteral nutrition. However, consistent with the Brain Trauma Foundation guidelines for pediatric severe TBI to initiate enteral nutrition within 72 h from admission, 48% of patients in our study with severe TBI were enterally fed within 48 h and 65% in < 72 h. This reflects the acceptance of earlier initiation of enteral nutrition in this patient population [13].
Children with severe TBI (GCS < 9) were more likely to experience delayed initiation of enteral nutrition. This delay may be related to their physiologic instability and the various medical and surgical interventions necessary in the initial 48–72 h of PICU admission. Investigators have identified various factors associated with delayed initiation of enteral nutrition such as illness severity scores, noninvasive and invasive mechanical ventilation [14], surgical procedures, and planned extubation [15, 16]. Additionally, the level of vasoactive support in severe TBI may impact the initiation of enteral nutrition. Limitations of care or progression to brain death may have influenced decision making. Some reasons for delay in feeding may be modifiable, such as provider decisions based on a combination of factors mentioned above in addition to other clinical factors such as absence of bowel sounds, abdominal distension. Early use of naso-jejunal feeding might allow safe initiation and rapid advancement of enteral nutrition. It requires shorter interruptions for surgical procedures and has been shown to be well tolerated even when bowel sounds are absent [17]. Some sites were more likely to initiate enteral nutrition earlier than others. This suggests potential for quality improvement initiatives aimed to promote early initiation of enteral nutrition in brain-injured patients. For example, other studies have found that early involvement of a dietitian was associated with early initiation of enteral nutrition [13].
In our study, early enteral feeding appears safe in children with TBI even with coexisting abdominal injury. Early feeding was not associated with complications of increased infections, abdominal compartment syndrome, or constipation. Although significant univariate factors, abdominal injury, and abdominal procedures were not independent risk factors for delayed initiation of enteral nutrition. This may be related to the small numbers of these diagnoses and procedures; however, it may also reflect movements on behalf of pediatric surgeons to endorse enhanced recovery after surgery protocols and principles even after abdominal surgery [18,19,20].
Our findings on outcomes associated with delayed initiation of enteral nutrition are consistent with findings of other studies [7, 9], in that, early enteral feeding is associated with improved outcome. Our study includes multiply injured children of all ages and injury severity as typically admitted to PICUs. In comparison with previous studies [7,8,9], our study included injury and illness severity as covariates in the multivariable analysis, removing an important confounder of outcomes. We did not find a mortality difference in our analysis adjusted for illness/injury severity in contrast to two previous studies [8, 9]. Unlike the study by Meinert et al. [9], we chose to include patients with GCS = 3 as these patients constituted almost 60% of the cohort with GCS < 9. The lack of mortality benefit could be related to inclusion of these patients. It could also be the result of over-fitting the multivariable model as we adjusted for several markers of injury and illness severity. Moreover, mortality is a rare outcome in the pediatric population requiring very large sample size to show a mortality benefit associated with various interventions.
Severe brain injury in children can result in permanent disabilities with far-reaching consequences for the rest of their lives. Thus, functional outcome measures, such as POPC and PCPC, are equally important when assessing pediatric outcomes [21, 22]. Delayed enteral nutrition was an independent risk factor for worse functional status at ICU discharge for the overall cohort of children with TBI in our study, with an OR = 2.6 of being discharged with moderate/severe disability in the delayed feeding group. The benefit of early feeding on functional outcome was no longer present when limiting analysis to those with severe TBI. Larger, prospective studies, looking at the barriers to initiation of enteral nutrition as discussed above, will be needed to fully evaluate the risks and benefits of enteral nutrition in this most severely brain-injured group.
There are many hypotheses about why early feeding is important in pediatric TBI. For one, TBI is associated with a hypermetabolic state resulting in significantly higher protein and calorie requirements [23, 24]. It is also postulated that early nutrition attenuates the post-traumatic stress response and improves early immunological function [3]. This may in part explain why patients with delayed enteral nutrition had more infections compared to those fed early in our study, despite it not reaching statistical significance.
Limitations
The retrospective nature of our study of limited sample size only allows us to show association rather than causation. Although we had data on time to initiation of enteral nutrition, we did not have information on modalities of enteral feeding, advancement of volume of enteral feeds, or time to achieve goal calorie and protein intakes, which have been shown to impact outcomes [3, 4]. We also cannot rule out contribution of other factors that could not be evaluated such as vasoactive support, end of life care limitations. Additionally, we were unable to assess long-term outcomes.
Strengths
This retrospective study using the PTAM database is the first study, to our knowledge, to assess the factors associated with delayed initiation of enteral nutrition in children with TBI. While evaluating the impact of delayed enteral nutrition on outcomes, we were able to adjust for important variables that are known to impact outcomes in these patients, such as GCS, injury and illness severity, pupillary reactivity, and child abuse status. Additionally, the study is multi-institutional, including data from five pediatric trauma centers around the nation. It is likely that the management of these TBI patients reflects practices at other pediatric trauma centers around the country. While conclusions drawn from a retrospective study like ours are dependent on the accuracy and availability of data, there is high interrater reliability for the data collected in the trauma registry and the VPS database. There was minimal missingness among the variables analyzed, with only 8% of patients being excluded due to incomplete data, which helps mitigate confounding. Prospective studies will be needed to better assess risk factors for delayed initiation of enteral nutrition as well as the impact of nutrition on outcomes in children with severe TBI.
Conclusion
Early enteral feeding is common in critically ill children after TBI. Children with severe TBI (GCS < 9) and higher ISS were more likely to have delayed initiation of enteral nutrition. Abdominal injury and procedures were associated with delayed enteral nutrition in unadjusted analysis, though not after adjustment for injury/illness severity. Delayed enteral nutrition was also an independent risk factor for worse functional status at ICU discharge in the entire cohort, but not in the severe TBI cohort.
References
Langlois JA. Traumatic brain injury in the United States: assessing outcomes in children, summary and recommendations. Atlanta: National Center for Injury Prevention and Control, Center for Disease Control and Prevention; 2001.
Kochanek PM, Carney N, Adelson P, et al. Guidelines for the acute medical management of severe traumatic brain injury in infants, children and adolescents—second edition. Chapter 16. Glucose and nutrition. Pediatr Crit Care Med. 2012;13(1(Suppl)):S68–71.
Hartl R, Gerber LM, Ni Q, Ghajar J. Effect of early nutrition on deaths due to severe traumatic brain injury. J Neurosurg. 2008;109:50–6.
Perel P, Yanagawa T, Bunn F, Roberts I, Wentz R, Pieno A. Nutritional support for head-injured patients [Review of the data base utritional support for head-injured patients]. Cochrane Database Syst Rev. 2006. https://doi.org/10.1002/14651858.cd001530.pub2.
Mehta NM, Bechard LJ, Zurakowski D, et al. Adequate enteral protein intake is inversely associated with 60-d mortality in critically ill children: a multicenter, prospective, cohort study. Am J Clin Nutr. 2015;102(199–206):10.
Mikhailov TA, Kuhn EM, Manzi J, et al. Early enteral nutrition is associated with lower mortality in critically ill children. JPEN J Parenter Enteral Nutr. 2014;38:459–66.
Taha AA, Badr L, Westlake C, Dee V, Mudit M, Tiras KL. Effect of early nutritional support on intensive care unit length of stay and neurological status at discharge in children with severe traumatic brain injury. J Neurosci Nurs. 2011;43(6):291–7.
Vavilala MS, Kernic MA, Wang J, et al. Pediatric Guideline Adherence and Outcomes Study: acute care clinical indicators associated with discharge outcomes in children with severe traumatic brain injury. Crit Care Med. 2014;42:2258–66.
Meinert E, Bell MJ, Buttram S, Kochanek PM, Balasubramani GK, Wisniewski SR, et al. Initiating nutritional support before 72 hours is associated with favorable outcome after severe traumatic brain injury in children: a secondary analysis of a randomized, controlled trial of therapeutic hypothermia. Pediatr Crit Care Med. 2018;19(4):345–52.
ACS TQIP. Best practices in management of traumatic brain injury. Jan 2015.
Fiser DH. Assessing the outcome of pediatric intensive care. J Pediatr. 1992;121(1):68–74.
Fiser DH, Tilford JM, Roberson PK. Relationship of illness severity and length of stay to functional outcomes in the pediatric intensive care unit: a multi-institutional study. Crit Care Med. 2000;28(4):1173–9.
Malakouti A, Sookplung P, Siriussawakul A, Philip S, et al. Nutrition support and deficiencies in children with severe traumatic brain injury. Pediatr Crit Care Med. 2012;13(1):e18–24. https://doi.org/10.1097/PCC.0b013e31820aba1f.
Canarie MF, Barry S, Carroll CL, Hassinger A, Kandil S, Li S, Pinto M, Valentine SL, Faustino EV. Northeast pediatric critical care research consortium. risk factors for delayed enteral nutrition in critically ill children. Pediatr Crit Care Med. 2015;16(8):e283–9.
Keehn A, O’Brien C, Mazurak V, Brunet-Wood K, Joffe A, et al. Epidemiology of interruptions to nutrition support in critically ill children in the pediatric intensive care unit. JPEN J Parenter Enteral Nutr. 2015;39(2):211–7.
Mehta NM, McAleer D, Hamilton S, Naples E, Leavitt K, et al. Challenges to optimal enteral nutrition in a multidisciplinary pediatric intensive care unit. JPEN J Parenter Enteral Nutr. 2010;34(1):38–45.
Grahm TW, Zadrozny DB, Harrington T. The benefits of early jejunal hyperalimentation in the head-injured patient. Neurosurgery. 1989;25:729–35.
Sangkhathat S, Patrapinyokul S, Tadyathikom K. Early enteral feeding after closure of colostomy in pediatric patients. J Pediatr Surg. 2003;38(10):1516–9.
Shinnick JK, Short HL, Heiss KF, Santore MT, Blakely ML, Raval MV. Enhancing recovery in pediatric surgery: a review of the literature. J Surg Res. 2016;202(1):165–76.
Vrecenak JD, Mattei P. Fast-track management is safe and effective after bowel resection in children with Crohn’s disease. J Pediatr Surg. 2014;49(1):99–102 discussion 102–3.
Gabbe BJ, Simpson PM, Sutherland AM, Palmer CS, Williamson OD, et al. Functional and health-related quality of life outcomes after pediatric trauma. J Trauma. 2011;70(6):1532–8.
Stylianos S, Ford HR. Outcomes in pediatric trauma care. Semin Pediatr Surg. 2008;17(2):110–5.
Phillips R, Ott L, Young B, Walsh J. Nutritional support and measured energy expenditure of the child and adolescent with head injury. J Neurosurg. 1987;67:846Y851.
Rhoney DH, Parker D, Formea CM, Yap C, Coplin WM. Tolerability of bolus versus continuous gastric feeding in brain-injured patients. Neurol Res. 2002;24:613Y620.
Funding
This project was, in part, supported by a 2014 Childress Foundation grant. Dr. Katherine T Flynn-O’Brien received fellowship support from the National Institute of Child Health and Human Development (T32-HD057822) during the preparation of this paper. VPS data were provided from the VPS, LLC. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health, the Childress Institute, or VPS, LLC.
Author information
Authors and Affiliations
Contributions
Dr. Balakrishnan conceptualized the study, interpreted the data analysis, wrote the first draft and edited the manuscript. Dr. Flynn-O’Brien created the PTAM database used for the study and was involved in data interpretation and manuscript edits. Dr. Simpson and Ms. Dasgupta provided statistical data analysis and edited the manuscript. Dr. Hanson conceptualized the study, interpreted the data analysis and edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest relevant to this article to disclose.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Balakrishnan, B., Flynn-O’Brien, K.T., Simpson, P.M. et al. Enteral Nutrition Initiation in Children Admitted to Pediatric Intensive Care Units After Traumatic Brain Injury. Neurocrit Care 30, 193–200 (2019). https://doi.org/10.1007/s12028-018-0597-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-018-0597-6