Abstract
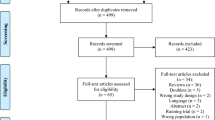
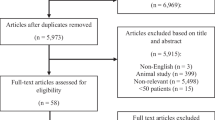
Our goal was to perform a systematic review of the literature on the use of ketamine in traumatic brain injury (TBI) and its effects on intracranial pressure (ICP). All articles from MEDLINE, BIOSIS, EMBASE, Global Health, HealthStar, Scopus, Cochrane Library, the International Clinical Trials Registry Platform (inception to November 2013), reference lists of relevant articles, and gray literature were searched. Two reviewers independently identified all manuscripts pertaining to the administration of ketamine in human TBI patients that recorded effects on ICP. Secondary outcomes of effect on cerebral perfusion pressure, mean arterial pressure, patient outcome, and adverse effects were recorded. Two reviewers independently extracted data including population characteristics and treatment characteristics. The strength of evidence was adjudicated using both the Oxford and GRADE methodology. Our search strategy produced a total 371 citations. Seven articles, six manuscripts and one meeting proceeding, were considered for the review with all utilizing ketamine, while documenting ICP in severe TBI patients. All studies were prospective studies. Five and two studies pertained to adults and pediatrics, respectively. Across all studies, of the 101 adult and 55 pediatric patients described, ICP did not increase in any of the studies during ketamine administration. Three studies reported a significant decrease in ICP with ketamine bolus. Cerebral perfusion pressure and mean blood pressure increased in two studies, leading to a decrease in vasopressors in one. No significant adverse events related to ketamine were recorded in any of the studies. Outcome data were poorly documented. There currently exists Oxford level 2b, GRADE C evidence to support that ketamine does not increase ICP in severe TBI patients that are sedated and ventilated, and in fact may lower it in selected cases.

Similar content being viewed by others
References
Aroni F, Iacovidou N, Dontas I, Pourzitaki C, Xanthos T. Pharmacological aspects and potential new clinical applications of ketamine: reevaluation of an old drug. J Clin Pharmacol. 2009;49:957–64.
Roberts DJ, Hall RI, Kramer AH, Roberston HL, Gallagher CN, Zygun DA. Sedation for critically ill adults with severe traumatic brain injury: a systematic review of randomized controlled trials. Crit Care Med. 2011;39(12):2743–51.
Sedev RS, Symmons DAD, Kindl K. Ketamine for rapid sequence induction in patients with head injury in the emergency department. Emerg Med Australas. 2006;18:37–44.
Himmelseher S, Durieux ME. Revising a dogma: ketamine for patients with neurological injury? Anesth Analg. 2005;101:524–34.
Wyte SR, Shapiro HM, Turner P, Harris AB. Ketamine-induced intracranial hypertension. Anesthesiology. 1972;36(2):174–6.
Shapiro HM, Wyte SR, Harris AB. Ketamine anesthesia in patients with intracranial pathology. Br J Anaesth. 1972;44:1200–4.
Gardner AE, Olson BE, Lichtiger M. Cerebrospinal-fluid pressure during dissociative anesthesia with ketamine. Anesthesiology. 1971;35(2):226–8.
List WF, Crumrine RS, Cascorbi HF, Weiss MH. Increased cerebrospinal fluid pressure after ketamine. Anesthesiology. 1972;36(1):93–4.
Visser E, Schug SA. The role of ketamine in pain management. Biomed Pharmacother. 2006;60:341–8.
Bar-Joseph G, Guilburd Y, Guilburd J. Ketamine effectively prevents intracranial pressure elevations during endotracheal suctioning and other distressing interventions in patients with severe traumatic brain injury. Crit Care Med. 2009;37(12):A402.
Bar-Joseph G, Guilburd Y, Tamir A, Guilburd J. Effectiveness of ketamine in decreasing intracranial pressure in children with intracranial hypertension. J Neurosurg Pediatr. 2009;4:40–6.
Boirgoin A, Albanese J, Leone M, Sampol-Manos E, Viviand X, Martin C. Effects of sufentanil or ketamine administered in target-controlled infusion on the cerebral hemodynamics of severely brain injured patients. Crit Care Med. 2005;33(5):1109–13.
Boirgoin A, Albanese J, Wereszczynski N, Charbit M, Vialet R, Martin C. Safety of sedation with ketamine in severe head injury patients: comparison with fentanyl. Crit Care Med. 2003;31(3):711–7.
Albanese J, Arnaud S, Rey M, Thomachot L, Alliez B, Martin C. Ketamine decreases intracranial pressure and electroencephalographic activity in traumatic brain injury patients during propofol sedation. Anesthesiology. 1997;87:1328–34.
Kolenda H, Gremmelt A, Rading S, Braun U, Markakis E. Ketamine for analgosedative therapy in intensive care treatment of head-injured patients. Acta Neurochir (Wien). 1996;138:1193–9.
Schmittner MD, Vajkoczy SL, Horn P, Bertsch T, Quintel M, Vajkoczy P, et al. Effect of fentanyl and S(+)-ketamine on cerebral hemodynamics, gastrointestinal motility, and need of vasopressors in patients with intracranial pathologies a pilot study. J Neurosurg Anesthesiol. 2007;19:257–62.
Yurkewicz L, Weaver J, Bullock MR, Marshall MF. The effect of elective NMDA receptor antagonist traxoprodil in the treatment of traumatic brain injury. J Neurotrauma. 2005;22(12):1428–43.
Morris GF, Bullock R, Marshall SB, Marmarou A, Maas A, Marshall LF, et al. Failure of the competitive N-methyl-d-aspartate antagonist Selfotel (CGS 19755) in treatment of severe head injury: results of two phase III trials. J Neurosurg. 1999;91:737–43.
Stewart L, Bullock R, Teasdale GM, Wagstaff A. First observations of the safety and tolerability of a competitive antagonist to the glutamate NMDA receptor (CGS 19755) in patients with severe head injury. J Neurotrauma. 1999;16(9):843–50.
Higgins JPT, Green S, editors. Cochrane handbook for systematic reviews of interventions Version 5.1.0. http://handbook.cochrane.org/. Accessed 25 October 2013.
Moher D, Liberati A, Tetzlaff J, Altman DG, Group P. Preferred reporting items for systematic reviews and meta-analysis: the PRISMA statement. Ann Intern Med. 2009;151(4):264–9.
Phillips B, Ball C, Sackett D, Straus S, Haynes B, Dawes M. Oxford Centre for Evidence-Based Medicine Levels of Evidence Version 2009. http://www.cebm.net/?o=1025. Accessed October 2013.
Guyatt GH, Oxman AD, Vist G, Kunz R, Falck-Ytter Y, Alonso-Coello P, et al. Rating quality of evidence and strength of recommendations GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008;336:924–6.
Guyatt GH, Oxman AD, Kunz R, Vist GE, Falck-Ytter Y, Schünemann HJ, et al. Rating quality of evidence and strength of recommendations: what is “quality of evidence” and why is it important to clinicians? BMJ. 2008;336(7651):995–8.
Schünemann HJ, Oxman AD, Brozek J, Glasziou P, Jaeschke R, Vist GE, et al. Grading quality of evidence and strength of recommendations for diagnostic tests and strategies. BMJ. 2008;336(7653):1106–10.
Guyatt GH, Oxman AD, Kunz R, Jaeschke R, Helfand M, Liberati A, et al. Rating quality of evidence and strength of recommendations: incorporating considerations of resources use into grading recommendations. BMJ. 2008;336(7654):1170–3.
Guyatt GH, Oxman AD, Kunz R, Falck-Ytter Y, Vist GE, Liberati A, et al. Rating quality of evidence and strength of recommendations: going from evidence to recommendations. BMJ. 2008;336(7652):1049–51.
Jaeschke R, Guyatt GH, Dellinger P, Schünemann H, Levy MM, Kunz R, et al. Use of GRADE grid to reach decisions on clinical practice guidelines when consensus is elusive. BMJ. 2008;337:a744.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zeiler, F.A., Teitelbaum, J., West, M. et al. The Ketamine Effect on ICP in Traumatic Brain Injury. Neurocrit Care 21, 163–173 (2014). https://doi.org/10.1007/s12028-013-9950-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-013-9950-y