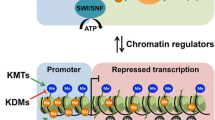
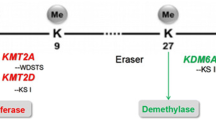
Abstract
Rubinstein–Taybi syndrome (RTS) is an incurable genetic disorder with combination of mental retardation and physical features including broad thumbs and toes, craniofacial abnormalities, and growth deficiency. While the autosomal dominant mode of transmission is limitedly known, the majority of cases are attributable to de novo mutations in RTS. The first identified gene associated with RTS is CREB-binding protein (CREBBP/CBP). Alterations of the epigenetic ‘histone code’ due to dysfunction of the CBP histone acetyltransferase activity deregulate gene transcriptions that are prominently linked to RTS pathogenesis. In this review, we discuss how CBP mutation contributes to modifications of histone and how histone deacetylase inhibitors are therapeutically applicable to epigenetic conditioning in RTS. Since most genetic mutations are irreversible and therapeutic approaches are limited, therapeutic targeting of reversible epigenetic components altered in RTS may be an ideal strategy. Expeditious further study on the role of the epigenetic mechanisms in RTS is encouraged to identify novel epigenetic markers and therapeutic targets to treat RTS.



Similar content being viewed by others
References
Abel, T., Nguyen, P. V., Barad, M., Deuel, T. A., Kandel, E. R., & Bourtchouladze, R. (1997). Genetic demonstration of a role for PKA in the late phase of LTP and in hippocampus-based long-term memory. Cell, 88(5), 615–626.
Abel, T., & Zukin, R. S. (2008). Epigenetic targets of HDAC inhibition in neurodegenerative and psychiatric disorders. Current Opinion in Pharmacology, 8(1), 57–64.
Alarcon, J. M., Malleret, G., Touzani, K., Vronskaya, S., Ishii, S., Kandel, E. R., et al. (2004). Chromatin acetylation, memory, and LTP are impaired in CBP ± mice: A model for the cognitive deficit in Rubinstein–Taybi syndrome and its amelioration. Neuron, 42(6), 947–959.
Atkins, C. M., Selcher, J. C., Petraitis, J. J., Trzaskos, J. M., & Sweatt, J. D. (1998). The MAPK cascade is required for mammalian associative learning. Nature Neuroscience, 1(7), 602–609.
Balasubramanyam, M., Koteswari, A. A., Kumar, R. S., Monickaraj, S. F., Maheswari, J. U., & Mohan, V. (2003). Curcumin-induced inhibition of cellular reactive oxygen species generation: Novel therapeutic implications. Journal of Biosciences, 28(6), 715–721.
Balci, S., Bostanci, S., Ekmekci, P., Cebeci, I., Bokesoy, I., Bartsch, O., et al. (2004). A 15-year-old boy with Rubinstein–Taybi syndrome associated with severe congenital malalignment of the toenails. Pediatric Dermatology, 21(1), 44–47.
Bannister, A. J., Schneider, R., & Kouzarides, T. (2002). Histone methylation: Dynamic or static? Cell, 109(7), 801–806.
Bartholdi, D., Roelfsema, J. H., Papadia, F., Breuning, M. H., Niedrist, D., Hennekam, R. C., et al. (2007). Genetic heterogeneity in Rubinstein–Taybi syndrome: Delineation of the phenotype of the first patients carrying mutations in EP300. Journal of Medical Genetics, 44(5), 327–333.
Bartsch, O., Wagner, A., Hinkel, G. K., Krebs, P., Stumm, M., Schmalenberger, B., et al. (1999). FISH studies in 45 patients with Rubinstein–Taybi syndrome: Deletions associated with polysplenia, hypoplastic left heart and death in infancy. European Journal of Human Genetics, 7(7), 748–756.
Beluffi, G., Pazzaglia, U. E., Fiori, P., Pricca, P., & Poznanski, A. K. (1987). Oto-palato-digital syndrome. Clinico-radiological study. Radiology Medica, 74(3), 176–184.
Bito, H., Deisseroth, K., & Tsien, R. W. (1996). CREB phosphorylation and dephosphorylation: A Ca(2+)- and stimulus duration-dependent switch for hippocampal gene expression. Cell, 87(7), 1203–1214.
Blough, R. I., Petrij, F., Dauwerse, J. G., Milatovich-Cherry, A., Weiss, L., Saal, H. M., et al. (2000). Variation in microdeletions of the cyclic AMP-responsive element-binding protein gene at chromosome band 16p13.3 in the Rubinstein–Taybi syndrome. American Journal of Medical Genetics, 90(1), 29–34.
Bourtchouladze, R., Lidge, R., Catapano, R., Stanley, J., Gossweiler, S., Romashko, D., et al. (2003). A mouse model of Rubinstein–Taybi syndrome: Defective long-term memory is ameliorated by inhibitors of phosphodiesterase 4. Proceedings of the National Academy of Sciences of the United States of America, 100(18), 10518–10522.
Caglayan, A. O., Lechno, S., Gumus, H., Bartsch, O., & Fryns, J. P. (2011). A boy with classical Rubinstein–Taybi syndrome but no detectable mutation in the CREBBP and EP300 genes. Genetic Counseling, 22(4), 341–346.
Camelo, S., Iglesias, A. H., Hwang, D., Due, B., Ryu, H., Smith, K., et al. (2005). Transcriptional therapy with the histone deacetylase inhibitor trichostatin A ameliorates experimental autoimmune encephalomyelitis. Journal of Neuroimmunology, 164(1–2), 10–21.
Cantani, A., & Gagliesi, D. (1998). Rubinstein–Taybi syndrome. Review of 732 cases and analysis of the typical traits. European Review for Medical and Pharmacological Sciences, 2(2), 81–87.
Coupry, I., Monnet, L., Attia, A. A., Taine, L., Lacombe, D., & Arveiler, B. (2004). Analysis of CBP (CREBBP) gene deletions in Rubinstein–Taybi syndrome patients using real-time quantitative PCR. Human Mutation, 23(3), 278–284.
Coupry, I., Roudaut, C., Stef, M., Delrue, M. A., Marche, M., Burgelin, I., et al. (2002). Molecular analysis of the CBP gene in 60 patients with Rubinstein–Taybi syndrome. Journal of Medical Genetics, 39(6), 415–421.
Demeer, B., Andrieux, J., Receveur, A., Morin, G., Petit, F., Julia, S., et al. (2013). Duplication 16p13.3 and the CREBBP gene: Confirmation of the phenotype. European Journal of Medical Genetics, 56(1), 26–31.
Devaskar, S. U., & Raychaudhuri, S. (2007). Epigenetics–a science of heritable biological adaptation. Pediatric Research, 61(5 Pt 2), 1R–4R.
Ferrante, R. J., Ryu, H., Kubilus, J. K., D’Mello, S., Sugars, K. L., Lee, J., et al. (2004). Chemotherapy for the brain: The antitumor antibiotic mithramycin prolongs survival in a mouse model of Huntington’s disease. Journal of Neuroscience, 24(46), 10335–10342.
Gardian, G., Browne, S. E., Choi, D. K., Klivenyi, P., Gregorio, J., Kubilus, J. K., et al. (2005). Neuroprotective effects of phenylbutyrate in the N171-82Q transgenic mouse model of Huntington’s disease. Journal of Biological Chemistry, 280(1), 556–563.
Ghosh, A. K., Nagpal, V., Covington, J. W., Michaels, M. A., & Vaughan, D. E. (2012). Molecular basis of cardiac endothelial-to-mesenchymal transition (EndMT): Differential expression of microRNAs during EndMT. Cellular Signalling, 24(5), 1031–1036.
Giles, R. H., Petrij, F., Dauwerse, H. G., den Hollander, A. I., Lushnikova, T., van Ommen, G. J., et al. (1997). Construction of a 1.2-Mb contig surrounding, and molecular analysis of, the human CREB-binding protein (CBP/CREBBP) gene on chromosome 16p13.3. Genomics, 42(1), 96–114.
Giussani, C., Selicorni, A., Fossati, C., Ingelmo, P., Canonico, F., Landi, A., et al. (2012). The association of neural axis and craniovertebral junction anomalies with scoliosis in Rubinstein–Taybi syndrome. Childs Nervous System, 28(12), 2163–2168.
Grossman, S. R. (2001). p300/CBP/p53 interaction and regulation of the p53 response. European Journal of Biochemistry, 268(10), 2773–2778.
Grossman, S. R., Deato, M. E., Brignone, C., Chan, H. M., Kung, A. L., Tagami, H., et al. (2003). Polyubiquitination of p53 by a ubiquitin ligase activity of p300. Science, 300(5617), 342–344.
Grossman, S. R., Perez, M., Kung, A. L., Joseph, M., Mansur, C., Xiao, Z. X., et al. (1998). p300/MDM2 complexes participate in MDM2-mediated p53 degradation. Molecular Cell, 2(4), 405–415.
Gu, W., & Roeder, R. G. (1997). Activation of p53 sequence-specific DNA binding by acetylation of the p53 C-terminal domain. Cell, 90(4), 595–606.
Haettig, J., Stefanko, D. P., Multani, M. L., Figueroa, D. X., McQuown, S. C., & Wood, M. A. (2011). HDAC inhibition modulates hippocampus-dependent long-term memory for object location in a CBP-dependent manner. Learning and Memory, 18(2), 71–79.
Hallam, T. M., & Bourtchouladze, R. (2006). Rubinstein–Taybi syndrome: Molecular findings and therapeutic approaches to improve cognitive dysfunction. Cellular and Molecular Life Sciences, 63(15), 1725–1735.
Harth, W., & Linse, R. (2001). Dermatological stigmata in Rubinstein–Taybi syndrome. Hautarzt, 52(10 Pt 2), 977–979.
Hennekam, R. C. (2006). Rubinstein–Taybi syndrome. European Journal of Human Genetics, 14(9), 981–985.
Hennekam, R. C., Tilanus, M., Hamel, B. C., Voshart-van Heeren, H., Mariman, E. C., van Beersum, S. E., et al. (1993). Deletion at chromosome 16p13.3 as a cause of Rubinstein–Taybi syndrome: Clinical aspects. American Journal of Human Genetics, 52(2), 255–262.
Hou, J. W. (2005). Rubinstein–Taybi syndrome: Clinical and molecular cytogenetic studies. Acta Paediatrica Taiwanica, 46(3), 143–148.
Iyer, N. G., Ozdag, H., & Caldas, C. (2004). p300/CBP and cancer. Oncogene, 23(24), 4225–4231.
Kalkhoven, E., Roelfsema, J. H., Teunissen, H., den Boer, A., Ariyurek, Y., Zantema, A., et al. (2003). Loss of CBP acetyltransferase activity by PHD finger mutations in Rubinstein–Taybi syndrome. Human Molecular Genetics, 12(4), 441–450.
Kalkhoven, E., Teunissen, H., Houweling, A., Verrijzer, C. P., & Zantema, A. (2002). The PHD type zinc finger is an integral part of the CBP acetyltransferase domain. Molecular and Cellular Biology, 22(7), 1961–1970.
Kawai, H., Nie, L., Wiederschain, D., & Yuan, Z. M. (2001). Dual role of p300 in the regulation of p53 stability. Journal of Biological Chemistry, 276(49), 45928–45932.
Kazantsev, A. G., & Thompson, L. M. (2008). Therapeutic application of histone deacetylase inhibitors for central nervous system disorders. Nature Reviews. Drug Discovery, 7(10), 854–868.
Kim, M. S., Akhtar, M. W., Adachi, M., Mahgoub, M., Bassel-Duby, R., Kavalali, E. T., et al. (2012). An essential role for histone deacetylase four in synaptic plasticity and memory formation. Journal of Neuroscience, 32(32), 10879–10886.
Korzus, E., Rosenfeld, M. G., & Mayford, M. (2004). CBP histone acetyltransferase activity is a critical component of memory consolidation. Neuron, 42(6), 961–972.
Kumar, S., Suthar, R., Panigrahi, I., & Marwaha, R. K. (2012). Rubinstein–Taybi syndrome: Clinical profile of 11 patients and review of literature. Indian Society of Human Genetics, 18(2), 161–166.
Kuwabara, T., Hsieh, J., Nakashima, K., Warashina, M., Taira, K., & Gage, F. H. (2005). The NRSE smRNA specifies the fate of adult hippocampal neural stem cells. Symposium on Nucleic Acids Chemistry, 49, 87–88.
Labenne, M., Noir, A., Amsallem, D., Bertrand, A. M., Menget, A., & Burguet, A. (1990). Rubinstein–Taybi syndrome in four cases. Pediatrie, 45(7–8), 471–475.
Lee, J., Hagerty, S., Cormier, K. A., Kim, J., Kung, A. L., Ferrante, R. J., et al. (2008). Monoallele deletion of CBP leads to pericentromeric heterochromatin condensation through ESET expression and histone H3 (K9) methylation. Human Molecular Genetics, 17(12), 1774–1782.
Lee, J., Hwang, Y. J., Shin, J.-Y., Lee, W.-C., Wie, J., Kim, K. Y., et al. (2013). Epigenetic regulation of cholinergic receptor M1 (CHRM1) by histone H3K9me3 impairs Ca2+ signaling in Huntington’s disease. Acta Neuropathologica, 125, 727–739.
Marangi, G., Leuzzi, V., Orteschi, D., Grimaldi, M. E., Lecce, R., Neri, G., et al. (2008). Duplication of the Rubinstein–Taybi region on 16p13.3 is associated with a distinctive phenotype. American Journal of Medical Genetics. Part A, 146A(18), 2313–2317.
Marks, P. A., Richon, V. M., & Rifkind, R. A. (2000). Histone deacetylase inhibitors: Inducers of differentiation or apoptosis of transformed cells. Journal of the National Cancer Institute, 92(15), 1210–1216.
Miller, R. W., & Rubinstein, J. H. (1995). Tumors in Rubinstein–Taybi syndrome. American Journal of Medical Genetics, 56(1), 112–115.
Mizuguchi, Y., Specht, S., Lunz, J. G, 3rd, Isse, K., Corbitt, N., Takizawa, T., et al. (2012). Cooperation of p300 and PCAF in the control of microRNA 200c/141 transcription and epithelial characteristics. PLoS ONE, 7(2), e32449.
Oike, Y., Takakura, N., Hata, A., Kaname, T., Akizuki, M., Yamaguchi, Y., et al. (1999). Mice homozygous for a truncated form of CREB-binding protein exhibit defects in hematopoiesis and vasculo-angiogenesis. Blood, 93(9), 2771–2779.
O’Neil, D. W., Canada, R. T., Clark, M. V., & Lowe, J. W. (1989). Rubinstein–Taybi syndrome: Case report. Pediatric Dentistry, 11(2), 158–160.
Petrij, F., Giles, R. H., Dauwerse, H. G., Saris, J. J., Hennekam, R. C., Masuno, M., et al. (1995). Rubinstein–Taybi syndrome caused by mutations in the transcriptional co-activator CBP. Nature, 376(6538), 348–351.
Roelfsema, J. H., & Peters, D. J. (2007). Rubinstein–Taybi syndrome: Clinical and molecular overview. Expert Reviews in Molecular Medicine, 9(23), 1–16.
Rouaux, C., Jokic, N., Mbebi, C., Boutillier, S., Loeffler, J. P., & Boutillier, A. L. (2003). Critical loss of CBP/p300 histone acetylase activity by caspase-6 during neurodegeneration. EMBO Journal, 22(24), 6537–6549.
Rubinstein, J. H., & Taybi, H. (1963). Broad thumbs and toes and facial abnormalities. A possible mental retardation syndrome. American Journal of Diseases of Children, 105, 588–608.
Ryu, H., Lee, J., Hagerty, S. W., Soh, B. Y., McAlpin, S. E., Cormier, K. A., et al. (2006). ESET/SETDB1 gene expression and histone H3 (K9) trimethylation in Huntington’s disease. Proceedings of the National Academy of Sciences of the United States of America, 103(50), 19176–19181.
Ryu, H., Smith, K., Camelo, S. I., Carreras, I., Lee, J., et al. (2005). Sodium phenylbutyrate prolongs survival and regulates expression of anti-apoptotic genes in transgenic amyotrophic lateral sclerosis mice. Journal of Neurochemistry, 93(5), 1087–1098.
Sakaguchi, K., Herrera, J. E., Saito, S., Miki, T., Bustin, M., Vassilev, A., et al. (1998). DNA damage activates p53 through a phosphorylation–acetylation cascade. Genes and Development, 12(18), 2831–2841.
Servillo, G., Della Fazia, M. A., & Sassone-Corsi, P. (2002). Coupling cAMP signaling to transcription in the liver: Pivotal role of CREB and CREM. Experimental Cell Research, 275(2), 143–154.
Stefanko, D. P., Barrett, R. M., Ly, A. R., Reolon, G. K., & Wood, M. A. (2009). Modulation of long-term memory for object recognition via HDAC inhibition. Proceedings of the National Academy of Sciences of the United States of America, 106(23), 9447–9452.
Sugai, F., Yamamoto, Y., Miyaguchi, K., Zhou, Z., Sumi, H., Hamasaki, T., et al. (2004). Benefit of valproic acid in suppressing disease progression of ALS model mice. European Journal of Neuroscience, 20(11), 3179–3183.
Suzuki, K. T., Torres, L. C., Sugayama, S. M., Aguiar Alves Bda, C., Moreira-Filho, C. A., & Carneiro-Sampaio, M. (2013). New CBP mutations in Brazilian patients with Rubinstein–Taybi syndrome. Clinical Genetics, 83(3), 291–292.
Tanaka, Y., Naruse, I., Maekawa, T., Masuya, H., Shiroishi, T., & Ishii, S. (1997). Abnormal skeletal patterning in embryos lacking a single Cbp allele: A partial similarity with Rubinstein–Taybi syndrome. Proceedings of the National Academy of Sciences of the United States of America, 94(19), 10215–10220.
Udaka, T., Kurosawa, K., Izumi, K., Yoshida, S., Tsukahara, M., Okamoto, N., et al. (2006). Screening for partial deletions in the CREBBP gene in Rubinstein–Taybi syndrome patients using multiplex PCR/liquid chromatography. GeneticTesting, 10(4), 265–271.
Urdinguio, R. G., Sanchez-Mut, J. V., & Esteller, M. (2009). Epigenetic mechanisms in neurological diseases: Genes, syndromes, and therapies. The Lancet Neurology, 8(11), 1056–1072.
Vecsey, C. G., Hawk, J. D., Lattal, K. M., Stein, J. M., Fabian, S. A., Attner, M. A., et al. (2007). Histone deacetylase inhibitors enhance memory and synaptic plasticity via CREB: CBP-dependent transcriptional activation. Journal of Neuroscience, 27(23), 6128–6140.
Verhoeven, W. M., Tuinier, S., Kuijpers, H. J., Egger, J. I., & Brunner, H. G. (2010). Psychiatric profile in Rubinstein–Taybi syndrome. A review and case report.Psychopathology, 43(1), 63–68.
Vo, N., Klein, M. E., Varlamova, O., Keller, D. M., Yamamoto, T., Goodman, R. H., et al. (2005). A cAMP-response element binding protein-induced microRNA regulates neuronal morphogenesis. Proceedings of the National Academy of Sciences of the United States of America, 102(45), 16426–16431.
Wallerstein, R., Anderson, C. E., Hay, B., Gupta, P., Gibas, L., Ansari, K., et al. (1997). Submicroscopic deletions at 16p13.3 in Rubinstein–Taybi syndrome: Frequency and clinical manifestations in a North American population. Journal of Medical Genetics, 34(3), 203–206.
Wang, J., Liu, X., Wu, H., Ni, P., Gu, Z., Qiao, Y., et al. (2010). CREB up-regulates long non-coding RNA, HULC expression through interaction with microRNA-372 in liver cancer. Nucleic Acids Research, 38(16), 5366–5383.
Wiley, S., Swayne, S., Rubinstein, J. H., Lanphear, N. E., & Stevens, C. A. (2003). Rubinstein–Taybi syndrome medical guidelines. American Journal of Medical Genetics. Part A, 119A(2), 101–110.
Wood, M. A., Kaplan, M. P., Park, A., Blanchard, E. J., Oliveira, A. M., Lombardi, T. L., et al. (2005). Transgenic mice expressing a truncated form of CREB-binding protein (CBP) exhibit deficits in hippocampal synaptic plasticity and memory storage. Learn Memory, 12(2), 111–119.
Yagihashi, T., Kosaki, K., Okamoto, N., Mizuno, S., Kurosawa, K., Takahashi, T., et al. (2012). Age-dependent change in behavioral feature in Rubinstein–Taybi syndrome. Congenit Anom (Kyoto), 52(2), 82–86.
Yuan, Z. M., Huang, Y., Ishiko, T., Nakada, S., Utsugisawa, T., Shioya, H., et al. (1999a). Function for p300 and not CBP in the apoptotic response to DNA damage. Oncogene, 18(41), 5714–5717.
Yuan, Z. M., Huang, Y., Ishiko, T., Nakada, S., Utsugisawa, T., Shioya, H., et al. (1999b). Role for p300 in stabilization of p53 in the response to DNA damage. Journal of Biological Chemistry, 274(4), 1883–1886.
Acknowledgments
This study was supported by NIH NS 067283 (H.R.) and Brain Science Flagship Grant (2E24380) (H.R.) from Korea Institute of Science and Technology (KIST).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Elizabeth Park and Yunha Kim have equally contributed to this work.
Rights and permissions
About this article
Cite this article
Park, E., Kim, Y., Ryu, H. et al. Epigenetic Mechanisms of Rubinstein–Taybi Syndrome. Neuromol Med 16, 16–24 (2014). https://doi.org/10.1007/s12017-013-8285-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-013-8285-3