Abstract
Moxibustion is the main alternative medicine treatment that has been beneficial to diabetic peripheral neuropathy (DPN), a common complication secondary to diabetic microvascular injury. However, the underlying protective mechanism of moxibustion against neuroinflammation remains unclear. We hypothesized that moxibustion treats DPN by regulating the balance of nuclear factor-2 erythroid-related factor-2 (Nrf2)-nuclear factor-kappa light chain enhancer of B cells (NF-кB). In vivo, diabetes was induced in rats by injecting streptozotocin (STZ; 60 mg/kg; i.p.). Moxibustion was then applied to “Zusanli” (ST 36), “Guanyuan” (BL 26), and “Yishu” (EX-B 3) acupuncture points. Nerve conduction was detected. Serum interleukin (IL)-1β, IL-6, and IL-8 levels were determined through enzyme-linked immunosorbent assay. NF-κB and Nrf2 proteins were examined through immunoblot analysis. The mRNA of NF-κB and Nrf2 was evaluated through RT-PCR. We found that the conduction velocity and amplitude of the action potentials of sciatic nerve conduction were reduced in the DPN model group but were rescued by moxibustion treatment. Moxibustion also improved the effect of DPN on other parameters, including ultrastructural changes, NF-κB and Nrf2 expression in the sciatic nerve, and serum IL-1β, IL-6, and IL-8 levels. Our data suggested that moxibustion may alleviate neuroinflammation by inhibiting NF-κB and by activating Nrf2. Moxibustion may also provide therapeutic effects for patients with DPN by simultaneously targeting Nrf2 and NF-κB.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
According to the World Health Organization (WHO), the prevalence of diabetes mellitus is predicted to increase by 170% between 1995 and 2025 [1]; furthermore, 353 million individuals will suffer from diabetes mellitus by 2030 [2]. Diabetic neuropathy is the most common complication associated with peripheral neuropathy; this condition may be caused by diseases, nerve trauma, or side effects induced by systemic illness [3]. Diabetic neuropathy also results in large disease burdens, such as incapacity to work, poor quality of life, and consumption of health care resources [4]. However, the etiology of diabetic neuropathy is poorly understood. Although diabetic neuropathy is treated through glycemic control and diligent foot care, other treatments have yet to be developed. Therapeutic methods, including those currently evaluated through clinical trials, have been used to treat diabetic peripheral neuropathy (DPN) with limited efficacy. DPN is mainly prevented by glycemic control or proper pathological management; nevertheless, these strategies are poorly implemented [5, 6]. To relieve the pain associated with DPN, researchers should investigate common pharmacological treatments used to respond to the poor effect of common analgesics, such as non-steroidal anti-inflammatory drugs (NSAIDS) and opioids or gabapentin versus pregabalin clinical use; researchers should also describe Oriental Medicine guidelines and a long list of natural compounds identified as possible therapeutic or alternative agents to replace or to combine with existing therapies [7].
Acupuncture and moxibustion have experienced widespread adoption worldwide and their popularity has seen a gradual rise, but in China, they have a long history of over 4,000 years. Moxibustion is a therapy that treats and prevents diseases using moxa floss. As an external treatment to treat or prevent diseases and promote health of the body, the combustion of the moxa floss permits transmission of thermal effect on certain points or areas of the body surface to treat disease. Acupuncture methods are widely applied to treat DPN on the basis of research on clinical curative effects [8,9,10] and experimental mechanisms [11]. The effectiveness of acupuncture points has also been confirmed. Thai foot massage using deep pressure and stretching can improve foot sensation in DPN patients after a 2-week treatment [12]. One RCT suggested that electroacupuncture is more effective in improving physical activity and QoL and reduces the need for oral analgesic medication than manual acupuncture [13]. Likewise, moxibustion treatment is used to clinically treat arthritis [14], knee osteoarthritis [15, 16], and diabetes [17, 18]. Previous studies have assessed acupuncture’s preventive effect on DPN by improving the morphology and function of the myelinated fiber of the sciatic nerve. Garrow et al [19] have published the only randomized, placebo-controlled study of acupuncture in the management of lower limb DPN, reporting analgesic effect in the acupuncture group compared with little change in the sham group. Abuaisha et al [20] also reported nerve conduction velocity and a variety of subjective symptoms associated with this progressive disabling disorder improvement before and after acupuncture treatment. Tong et al [21] found significant improvements in both numbness in the upper extremities (severity) and sensation of rigidity (extent) in the acupuncture treatment group, but not in the sham group. However, the biological mechanisms underlying the treatment of DPN by moxibustion have yet to be fully elucidated.
Persistent hyperglycemia is possibly the main cause of neuroinflammation and nerve damage leading to neuropathic pain. Nrf2 is a redox-regulated transcription factor involved in the modulation of antioxidant defense systems. Nrf2 stimulates the production of endogenous antioxidant defenses and detoxifying enzymes. NF-κB is a transcription factor involved in proinflammatory cytokine production, in addition to its immunological function. The regulation of Nrf2 is coordinated with that of NF-κB to maintain redox homeostasis in healthy cells. However, this regulation is perturbed under pathological conditions; as such, an opportunity for therapeutic intervention becomes evident. Diabetic neuropathy is a condition, in which change in the expression pattern of Nrf2 and NF-κB has been reported [22].
In our study, a rat model of DPN was established and histological changes in periodontal tissues were observed under an ultrascope. Nerve conduction indicators were detected with the electrophysiological method. The expression levels of NF-κB and Nrf2 were observed through immunoblot. Our study aimed to investigate the role of Nrf2 and NF-κB in diabetic neuropathy and to summarize the therapeutic outcomes of moxibustion targeted at Nrf2–NF-κB in diabetic neuropathy.
Materials and Methods
Reagents and Animals
Three-month-old male Wistar rats with a body weight of 200–220 g were purchased from Shanghai Slaccas Experimental Animal Co., Ltd. (Shanghai, China; Certificate no. SCXK 2015-0012). Moxibustion was purchased from the National Institute for the Control of Pharmaceutical and Biological Products (Beijing, China). All of the rats were provided free access to water and food and maintained in a 12 h:12 h light/dark cycle at 22 ± 2 °C and 65–69% relative humidity for 8 weeks. This study was approved by the ethics committee of Hubei University of Chinese Medicine (Wuhan, China). The animal research protocol was conducted in accordance with the European Community guidelines for the use of experimental animals. STZ was purchased from Hangzhou Baitong Biological Technology Co., Ltd. (Hangzhou, China). IL-1β, IL-6, and IL-8 ELISA commercially available kits (R&D Systems, Minneapolis, MN, USA) were used. Rabbit antibody against β-actin (ab189073), rabbit anti-NF-κB polyclonal antibody (ab7971), and rabbit anti-Nrf2 polyclonal antibody (ab31163) were purchased from Abcam (Cambridge, MA, USA). Total RNA was extracted from freshly frozen neural tissues by using an Ultrapure RNA kit (CWbio Co., Ltd., China) and then reverse-transcribed with a HiFi-MMLV cDNA kit (CWbio Co., Ltd., China). Real-time PCR was performed in a Bioer line gene PCR instrument (BIOER, China) by using Invitrogen primers.
Animal Groups and Model
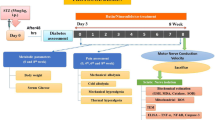
In this experiment, 100 rats were used. After the rats were subjected to fasting overnight, diabetes was induced to 80 rats by intraperitoneally injecting STZ dissolved in 0.1 M sodium citrate buffer (pH 4.5) at a dose of 60 mg/kg [23]. The successful induction of diabetes was confirmed when fasting blood glucose exceeded 16.7 mmol/L 3 days after STZ was injected and remained at > 16.7 mmol/L throughout the study. In the normal control group (N), the 20 remaining rats were treated with the same volume of cold citrate buffer and considered as nondiabetic rats.
Ischemia-reperfusion was induced to the diabetic rats in the DPN model group, as previously described [24]. In brief, the STZ-diabetic rats were anesthetized by intraperitoneally administering 50 mg/kg soluble pentobarbital sodium [25] after 4 weeks of induction. Ischemia was induced by occluding the abdominal aorta, right common iliac artery, and femoral artery with artery clips, which were removed after 3 h. Sixty-three rats were included in the final study conducted for 4 weeks. Seventeen rats were excluded from the total 80 rats because of death during surgery due to infection (n = 5, the percentage is 6.25%) or because of an insufficient increase in fasting blood glucose (< 16.7 mmol/L; n = 12, the percentage is 15% ), which is almost similar to the result from the previous experiment [26]. During the last week after infection, every cage received hydrated gel (Clear H2O, Portland, ME), a solid form of fluid replacer that was maintained off the bedding in a disposable dish. Topical antibiotic ointment (Antibiotic Ointment, CVS Pharmacy brand) was applied to any rat that rat tail and toe joints developed erosion or even necrosis. Surviving rats were not expected to exhibit additional health concerns and therefore were checked daily by animal care staff until the final experiment. Any rat that experienced prolonged inactivity or moribundity (pale, tachypnea, cold and transparent ears, corneal opacity and dull eyes) was euthanized by CO2 narcosis, and any rat that spontaneously died due to infection were removed immediately from the cages. We retained 20% CO2 in the enclosed flow cage (30.5 cm in width × 30.5 cm in. height × 61 cm in length) for euthanasia of rats. The animal experiment protocol was approved by the Animal Ethics Committee of Hubei University of Chinese Medicine in China.
Animal Treatment
Acupuncture points were referred to in the “Map of the Experimental Animal Acupuncture Points” developed by the Experimental Acupuncture Institute of China Association of Acupuncture and Moxibustion. Moxibustion was applied to the rats with DPN in the morning by Jia Li, who is a Doctor of Philosophy major in Acupuncture and Moxibustion. The rats were administered with mild anesthesia of 300 mg/kg chloral hydrate (including normal control and model groups, 4 ml/kg, less than the normal dose of 7 ml/kg); as a result, the rats could crawl after appropriate stimulation was administered. The rats completely recovered from the effects of anesthesia within 30 min. These rats were divided into three groups: DPN group (DPN, n = 23), DPN + moxibustion with 15 min/day group (M1, n = 19), and DPN + moxibustion with 30 min/day group (M2, n = 21). The moxibustion group were treated with moxibustion at “Zusanli” (ST 36), “Guanyuan” (BL 26), and “Yishu” (EX-B3) from the 4th week to the 8th week with a grain-sized moxa cone of approximately the size of a wheat grain (1 mg of pure moxa punk with a base of 2.5–3.0 mm and a height of 4–5 mm), once a day for 4 weeks at a rate of 15 and 30 min/day separately in M1 and M2 groups, respectively (the burning time of each cone was 10–12 s, with a temperature of 48–52 °C at acupoints, and the cone was removed before burning out in order to avoid the pain caused by heat stimulation in rats). Briefly, a grain-sized moxa cone was put on the acupoints using tweezers and ignited with a line-incense stick. A new moxa cone was applied when the prior one was burned up.
Electrophysiology
Sciatic nerve conduction velocity (SNCV), nerve action potential (NAP) amplitude, and tail nerve conduction velocity (TNCV) were measured by using the BL-420F biological function experimental system (Chengdu Technology & Market Co., Ltd., China) with stimulating and recording electrodes. After the rats were anesthetized with a dose of 300 mg/kg chloral hydrate, the right sciatic nerves were exposed by blunt dissection. The nerve was stimulated proximally and the electric signal was recorded from the distal digital nerve. The TNCV was measured similarly by using single needle stimulating electrodes inserted into the proximal tail and the recording electrodes inserted distally. The time (T) and distance (S) between the stimulating electrodes and the recording electrodes were used to calculate CV expressed as follows: CV = S/T.
Ultrastructural Observation
For histological analysis, the rats were anesthetized by intraperitoneally injecting with a dose of 300 mg/kg chloral hydrate and quickly decapitated. The sciatic nerve was removed at about 1 cm away from the sections used for the electrophysiology tests. The tissues were rapidly fixed with 2.5% pre-cooled glutaraldehyde in 0.1 M phosphate-buffered saline for 1–2 h, trimmed, and located. The suspension was mixed with 1% osmium tetrachloride for 1.5 h, dehydrated, embedded, sectioned, and examined using a Hitachi H-600 transmission electron microscope (Hitachi Company, Tokyo, Japan).
Determination of the Serum Levels of Inflammatory Cytokines Through ELISA
Serum was isolated from the blood samples through centrifugation at 10000 rpm for 10 min and stored at − 20 °C prior to the assays. The serum levels of IL-1β, IL-6, and IL-8 cytokines were evaluated using specific ELISA kits in accordance with the manufacturer’s protocol.
Immunoblot Analysis
Quick-frozen sciatic nerve samples were homogenized in dry ice and boiled in 50 μl of SDS sample buffer (1.5% dithiothreitol, 2% SDS, 80 mM Tris-HCl (pH 6.8), 10% glycerol, and 0.01% bromophenol blue) for 5 min and then separated through SDS-polyacrylamide gel electrophoresis (SDS-PAGE). After protein was transferred, the membrane was blocked with bovine serum albumin or milk for 1 h and probed by using a primary antibody, followed by horseradish peroxidase-conjugated secondary antibody. The levels of NF-κB and Nrf2 protein were quantified through the scanning densitometry of immunoblots (Fuji Multigauge software).
Real-time PCR
The sciatic nerve tissues were subjected to quantitative RT-PCR experiments. Total RNA was isolated from maxilla tissues by using Trizol reagent in accordance with the manufacturer’s protocol. The extracted RNA (1 μg) was reverse-transcribed into cDNA by using a PrimeScript RT reagent kit. RT-PCR was performed and from each PCR reaction. The primer sequences of NF-κB, Nrf2, and GAPDH were designed as follows: NF-κB p65, FW: 5′-GACGAGGCTCGGAGAGCCCA-3′ and RV: 5′-CTGGGGCGGCTGACCGAATG-3′; Nrf2, FW: 5′-TCCATTTCCGAGTCACTGAACCCA-3′ and RV: 5′-TGACTCTGACTCCGGCATTTCACT-3′; GAPDH, FW: 5′-CCTGGAGAAACCTGCCAAG-3′ and RV: 5′-CACAGGAGACAACCTGGTCC-3′. After PCR was completed, the products were analyzed through electrophoresis on 1.2% agarose gel and photographed under UV light in an EC3 Imaging System (UVP, Upland, CA).
Data Processing
All statistical analysis was performed using Prism 6 software (GraphPad Software, San Diego, CA). Differences between pairs of groups were analyzed by the Student–Newman–Keuls test or Dunn’s method. Values of n refer to the number of experiments used to obtain each value. p < 0.05 was considered to be significant. PS: Power and Sample Size Calculation software (Vanderbilt University) was used to determine the sample size.
Results
Effects of Moxibustion on the Nerve Conduction Velocity of the Rats with DPN
To determine the potential therapeutic effect of moxibustion on DPN, we initially identified the effect of moxibustion on the nerve conduction of the rats with DPN. Compared with those of the normal control (N) group, the SNCV, NAP amplitude, and TNCV were significantly reduced in the DPN model (DPN) group [N versus DPN; SNCV (m/s): 44.23 ± 5.3 versus 25.18 ± 3.35; NAP amplitude (mV): 12.2 ± 2.42 versus 6.25 ± 1.08; TNCV: 31.46 ± 6.14 versus 16.27 ± 2.83; n = 20 in group N while n = 23 in Group DPN, p < 0.01]. The SNCV, NAP amplitude, and TNCV increased with moxibustion treatment of either 15 min/day (M1, n = 19) or 30 min/day (M2, n = 21), compared with those detected in the DPN group (p < 0.05 for both comparisons; Fig. 1). There was no significant difference between groups M1 and M2.
Action potential amplitude (a), sciatic nerve conduction velocity (b), and tail nerve conduction velocity (c) in N (normal control, n = 20), DPN (model, n = 23), M1 (moxibustion for 15 min/day, n = 19), and M2 (moxibustion for 30 min/day, n = 21) groups (∆p < 0.05 versus the normal control group, *p < 0.05 versus the model group)
Sciatic Nerve Ultrastructure
Considering that moxibustion treatment improves the NCV of the rats with DPN, we hypothesized that moxibustion may protect the ultrastructure of the nerve tissue against diabetic and ischemic insults. The ultrastructure of the sciatic nerves was visualized by using a transmission electron microscope. The figures show the representative ultrastructure features in the N group: × 6000, figure a; the DPN group: × 6000, figure b; the M1 group: × 6000, figure c; and the M2 group: × 6000, figure d. Figure 2 shows the typical electron microscopy results that confirmed the edematous and ischemic nerve myelin. Figure 2 also reveals myelin sheath swelling and disruption, axonal atrophy, and cellular organelle reduction in the DPN model group. After moxibustion treatment was administered for 15 or 30 min, the pathological changes in the treated rats were reduced compared with those in the normal control group. The pathological changes included the mitigation of edema and injury to myelin sheath and axons.
Sciatic nerve ultrastructure (uranium-lead staining). Normal control group (N) (a): sciatic nerve with intact myelin sheaths and healthy axons with prolific cell organelles. Model group (DPN) (b): the sciatic nerve with severe loss of myelinated axons and increased endoneurial collagen, disrupted and convoluted myelin sheaths, and decreased cell organelles. M1 (c): moxibustion for 15 min/day. M2 (d): moxibustion for 30 min/day
Expression Levels of Serum IL-1β, IL-6, and IL-8
ELISA analysis demonstrated that the expression levels of serum IL-1β, IL-6, and IL-8 in the rat model significantly increased compared with the corresponding values in the DPN group (n = 23, p < 0.01). The moxibustion treatment significantly decreased the expression levels of IL-1β, IL-6, and IL-8 in the M1 and M2 groups compared with those in the DPN groups (n = 19 in group M1 while n = 21 in group M2, p < 0.05). No changes were observed in the moxibustion group (Fig. 3).
Immunoblot of the Sciatic Nerve
We determined the protein expression of NF-κB and Nrf2 in the sciatic nerve to confirm the data obtained from previous experiments. Compared with group N (n = 20), the expression level of NF-κB in the sciatic samples from the DPN group was significantly increased, whereas the expression level of Nrf2 significantly decreased (n = 23, p < 0.01). After the rats were treated with moxibustion at different durations, the expression levels of NF-κB in the sciatic nerve was significantly reduced, whereas the expression level of Nrf2 significantly increased in the moxibustion groups compared with the DPN group (n = 19 in group M1 while n = 21 in group M2, p < 0.05). No significant difference was observed between M1 and M2 groups (Fig. 4).
RT-PCR of the Sciatic Nerve
To confirm the observations obtained from the immunoblot experiment, we further investigated the mRNA expression of NF-κB and Nrf2. Compared with the N group (n = 20), the mRNA expression level of NF-κB in the sciatic nerve in the DPN group was significantly increased (n = 23, p < 0.05). After the moxibustion treatment was administered, the mRNA expression levels of NF-κB in the sciatic nerve were significantly decreased in the moxibustion groups compared with the model group (n = 19 in Group M1 while n = 21 in group M2, p < 0.05). By contrast, the mRNA expression of Nrf2 was decreased in the DPN group compared with the normal control group but relatively increased in the moxibustion-treated groups. However, no significant difference was observed between M1 and M2 groups (Fig. 5).
Discussion
DPN accounts for most common long-term complications of diabetes mellitus. As such, neural cells play an important role in the pathogenesis of DPN. Neural deficits are associated with abnormal reactions of glial cells and neurons in response to hyperglycemia [27]. Moxibustion is administered as an effective and safe treatment for DPN [28, 29]; however, the therapeutic mechanism has yet to be fully elucidated. In this study, neural ultrastructural changes and conduction in the sciatic nerve of the rats were quantified after DPN was induced. Whether these changes are associated with NF-κB and Nrf2 during moxibustion treatment was also investigated.
STZ is a broad-spectrum antibiotic, which is widely administered to experimental animals to induce diabetes; this antibiotic elicits toxic effects on the pancreatic β cells of pancreatic islets and thus causes the loss of insulin secretion of the islets [30]. STZ also triggers an immune response that leads to diabetes [31, 32]. The animals used to establish the model are usually inbred Wistar rats or Sprague-Dawley rats. Daniel et al. [33] reported that the success rate of the model establishment in male rats is significantly higher than that in female rats. Monea et al. [34] demonstrated that the sensitivity of rats to STZ occurs in an age-dependent manner. Our study used male Wistar rats with an average body weight of 212.5 g. After the rats were subjected to fasting overnight, diabetes was induced by intraperitoneally injecting 60 mg/kg STZ diluted in citrate buffer (pH 4.4). STZ-induced diabetes is characterized by hyperglycemia, severe body weight loss, polydipsia, polyphagia, and polyuria. Three days after STZ was injected, the blood glucose levels were assessed and the rats with blood glucose levels of > 16.7 mmol/l were considered diabetic. Compared with the normal rats, the STZ-treated rats yielded significantly increased blood glucose levels, reduced body weights, increased water and food consumption, and increased urine output (data not shown). These results confirmed that diabetes was effectively induced.
As a common complication of diabetes, DPN may lead to diabetic foot ulcers, even to foot or limb amputations. The first step in the treatment of DPN is prevention. DPN cannot be cured, and nerve damage cannot be repaired; therefore, individuals with diabetes must prevent the occurrence of DPN. No approved treatments have been administered to prevent or cure diabetic neuropathy, although symptomatic pain therapies of variable efficacy are available. In early 1987, WHO proposed 43 kinds of acupunctural indications, in which peripheral neuropathy was included. The traditional duration of a moxibustion treatment is 15 min and is targeted to fixed acupoints. Although some randomized and controlled trials have confirmed the efficiency of traditional moxibustion [35], strong evidence supporting the therapeutic effectiveness of this treatment is insufficient. Specific acupoints on the body are treated with moxibustion; thus, this treatment can produce the effect to regulate qi and blood, dredge the meridians, and balance the functions [36]. This treatment can also promote the elimination of contaminants and alleviate the pathological effects, such as inflammation, adhesion, exudation, and hematoma [37]. The duration of moxibustion treatment is often longer than that of normal treatment because duration is an important factor affecting the efficacy of moxibustion [38]. Therefore, this experiment administered 15- and 30-min moxibustion treatments. The results revealed that the application of moxibustion for 15 and 30 min/day significantly improved the neural conduction and alleviated the inflammation of the rats with DPN after 4 weeks of treatment. However, the difference between the two treatment durations was not significant. These results suggested that the 15-min moxibustion treatment sufficiently improves DPN.
Acupuncture and moxibustion preventive treatment for disease, which is the important component of the prevention treatment for disease in TCM, has widespread application since ancient times. Acupuncture can improve the ability to resist disease and can restore the ability to maintain the body’s steady state. This is also the recruitment of Chinese medicine “keep healthy, do not be evil.” Points ST 36 and BL 26 have a close relationship with congenital kidney and aquired spleen and stomach qi respectively, while EX-B 3 eliminate unhealthy trends. The point combination tends to address both the symptoms and root causes by paying equal attention to prevention.
NF-κB and Nrf2 pathways mediate cellular homeostasis by controlling oxidative stress and inflammation. NF-κB suppresses the transcriptional activity of Nrf2. In summary, NF-κB and Nrf2 individually affect many signaling cascades to maintain a redox homeostasis; these factors interact with each other to further modulate the level of key redox modulators of health conditions and diseases [39]. An imbalance between NF-κB and Nrf2 is a key step in the disruption of diabetes and is a major contributor to the development and progression of DPN. Previous studies supported the defensive role of Nrf2 in neurons exposed to oxidative stress and suggested that the NF-κB pathway is an important modulator of inflammatory damage in diabetic neuropathy [40]. Li et al. [41] demonstrated that Nrf2-deficient mice exhibit a greater induction of NF-κB-regulated pro-inflammatory genes, such as interleukins and TNF-α; thus, Nrf2 deficiency enhances NF-κB-mediated pro-inflammatory reactions. High glucose–induced reactive oxygen intermediate production and inflammatory damage are considered as contributors of nerve dysfunction and subsequent damage in diabetic neuropathy. Melatonin modulates neuroinflammation by decreasing the NF-κB activation cascade and regulates oxidative stress by increasing the Nrf2 expression; melatonin may elicit a neuroprotective effect against diabetic neuropathy [42]. Further studies support the defensive role of Nrf2 in neurons exposed to oxidative stress and suggest that the NF-κB pathway is an important modulator of inflammatory damage in diabetic neuropathy [43]. Diabetes involves chronic hyperglycemia caused by a range of genetic and environmental factors, and this condition is widely considered as a risk factor of periodontal disease. Previous studies also demonstrated that advanced glycosylation ends triggered by long-term hyperglycemia can stimulate phagocytes to release inflammatory cytokines, including tumor necrosis factor, IL-1β, and IL-6 [44]. Cytokines, such as IL-1β, IL-6, and IL-17, can sensitize the peripheral receptors and thus cause neuropathic injury [45].
The main strengths of this study have been indicated. To our knowledge, this study is the first to investigate the effects of moxibustion treatment on DPN; thus, this study provided preliminary results as a basis of future studies. The novel treatment strategy may have an important role in an experimental setting because current pharmacological approaches are insufficient for DPN-related neuroinflammation. We also obtained that the 15-min moxibustion treatment could sufficiently improve DPN,which significantly contributes to the existing literature.
Our study showed that moxibustion can regulate and restore the balance between the expression levels of NF-κB and Nrf2 and the pathological changes in the periodontal tissues of diabetic rats. Moxibustion alleviated the degradation of periodontal tissues and suppressed the synthesis and secretion of interleukins, such as IL-1β, IL-6, and IL-8, in DPN rats. Moxibustion inhibited NF-κB and rescued the Nrf2 expression in the sciatic nerve. In conclusion, moxibustion may elicit therapeutic effects that simultaneously target the Nrf2 and NF-κB pathways in DPN.
One of the limitations of this study is that although the rats with DPN were treated for 4 weeks to assess their nerve conduction, no continuous measurement was conducted to evaluate the long-term effect of moxibustion on the balance between NF-κB and Nrf2 expressions. Future studies should combine experimental evaluation with microscopic and molecular studies on nerve conduction to elucidate the long-term efficacy of moxibustion. Further studies should also elucidate whether moxibustion directly inhibits or regulates the body’s metabolic function by eliminating inflammatory factors and by indirectly regulating NF-κB and Nrf2 pathway. Besides, a transition to clinical research on humans needs further investigation.
Conclusions
Our results show that moxibustion inhibited the protein and mRNA expression of NF-κB but induced the protein and mRNA expression of Nrf2 in the sciatic nerve. Moxibustion restored the balance between NF-κB and Nrf2 in rats with DPN. Thus, neural inflammation was relieved by regulating interleukin inflammatory factors [Fig. 6].
References
Hsieh, Y. T., Tu, S. T., Cho, T. J., Chang, S. J., Chen, J. F., & Hsieh, M. C. (2012). Visit-to-visit variability in blood pressure strongly predicts all-cause mortality in patients with type 2 diabetes: a 5·5-year prospective analysis. European Journal of Clinical Investigation, 42(3), 245–253.
Harsha, S. N., Aldhubiab, B. E., Nair, A. B., Alhaider, I. A., Attimarad, M., Venugopala, K. N., Srinivasan, S., Gangadhar, N., & Asif, A. H. (2015). Nanoparticle formulation by Büchi B-90 Nano Spray Dryer for oral mucoadhesion. Drug Design, Development and Therapy, 9, 273–282.
Shen, H., Zhao, J., Liu, Y., & Sun, G. (2018). Interactions between and shared molecular mechanisms of diabetic peripheral neuropathy and obstructive sleep apnea in type 2 diabetes patients. Journal Diabetes Research, 2018, 3458615.
Callahan, D., Keeley, J., Alipour, H., DeVirgilio, C., Kaji, A., Plurad, D., & Kim, D. Y. (2016). Predictors of severity in diabetic foot infections. Annals of Vascular Surgery, 33, 103–108.
Urabe, H., Terashima, T., Lin, F., Kojima, H., & Chan, L. (2015). Bone marrow-derived TNF-α causes diabetic neuropathy in mice. Diabetologia., 58(2), 402–410.
Boulton, A. J., Vinik, A. I., Arezzo, J. C., Bril, V., Feldman, E. L., Freeman, R., Malik, R. A., Maser, R. E., Sosenko, J. M., & Ziegler, D. (2005). Diabetic neuropathies: a statement by the American Diabetes Association. Diabetes Care, 28(4), 956–962.
Galuppo, M., Giacoppo, S., Bramanti, P., & Mazzon, E. (2014). Use of natural compounds in the management of diabetic peripheral neuropathy. Molecules, 19(3), 2877–2895.
Bo, C., Xue, Z., Yi, G., Zelin, C., Yang, B., Zixu, W., & Yajun, W. (2012). Assessing the quality of reports about randomized controlled trials of acupuncture treatment on diabetic peripheral neuropathy. PLoS One, 7(7), e38461.
Jeon, E., Kwon, H., Shin, I., Kang, S., & Shon, H. (2014). Effect of acupuncture on diabetic peripheral neuropathy: an uncontrolled preliminary study from Korea. Acupuncture in Medicine, 32(4), 350–352.
Garrow, A. P., Xing, M., Vere, J., Verrall, B., Wang, L., & Jude, E. B. (2014). Role of acupuncture in the management of diabetic painful neuropathy (DPN): a pilot RCT. Acupuncture in Medicine, 32(3), 242–249.
Chatchawan, U., Eungpinichpong, W., Plandee, P., & Yamauchi, J. (2015). Effects of Thai foot massage on balance performance in diabetic patients with peripheral neuropathy: a randomized parallel-controlled trial. Medical Science Monitor Basic Research, 21, 68–75.
Hamza, M. A., White, P. F., Craig, W. F., Ghoname, E. S., Ahmed, H. E., Proctor, T. J., Noe, C. E., Vakharia, A. S., & Gajraj, N. (2000). Percutaneous electrical nerve stimulation: a novel analgesic therapy for diabetic neuropathic pain. Diabetes Care, 23(3), 365–370.
Glickman-Simon, R., & Lepper, L. T. (2014). Panax ginseng for psychomotor performance and cognition, spinal manipulation for lumbar disk herniation, ginger for migraines, music therapy for mental illness, and acupuncture for diabetic peripheral neuropathy. Explore (New York, N.Y.), 10(6), 404–407.
Xu, X., Wang, M. M., Sun, Z. L., Zhou, D. P., Wang, L., Wang, F. Q., Xu, Z. Y., & Ma, Q. (2016). Discovery of serum proteomic biomarkers for prediction of response to moxibustion treatment in rats with collagen-induced arthritis: an exploratory analysis. Acupuncture in Medicine, 34(3), 184–193.
Chen, R., Chen, M., Su, T., Zhou, M., Sun, J., Xiong, J., Chi, Z., Xie, D., & Zhang, B. (2015). Heat-sensitive moxibustion in patients with osteoarthritis of the knee: a three-armed multicentre randomised active control trial. Acupuncture in Medicine, 33(4), 262–269.
Ren, X., Yao, C., Wu, F., Li, Z., Xing, J., & Zhang, H. (2015). Effectiveness of moxibustion treatment in quality of life in patients with knee osteoarthritis: a randomized, double-blinded, placebo-controlled trial. Evidence-based Complementary and Alternative Medicine, 2015, 569523.
Garcia-Vivas, J. M., Galaviz-Hernandez, C., Becerril-Chavez, F., Lozano-Rodriguez, F., Zamorano-Carrillo, A., Lopez-Camarillo, C., & Marchat, L. A. (2014). Acupoint catgut embedding therapy with moxibustion reduces the risk of diabetes in obese women. Journal of Research in Medical Sciences : The Official Journal of Isfahan University of Medical Sciences, 19(7), 610–616.
Zheg, H. T., Huang, X. M., & Sun, J. H. (1986). Treatment of diabetic cystopathy by acupuncture and moxibustion. Journal of Traditional Chinese Medicine, 6(4), 243–248.
Garrow, A. P., Xing, M., Vere, J., Verrall, B., Wang, L., & Jude, E. B. (2014). Role of acupuncture in the management of diabetic painful neuropathy (DPN): a pilot RCT. Acupuncture in Medicine, 32(3), 242–249.
Abuaisha, B. B., Costanzi, A. J. M., & Boulton, A. J. M. (1998). Acupuncture of the treatment of chronic painful peripheral diabetic neuropathy: a long-term study. Diabetes Research and Clinical Practice, 39(2), 115–121.
Tong, Y., Guo, H., & Han, B. (2010). Fifteen-day acupuncture treatment relieves diabetic peripheral neuropathy. Journal of Acupuncture and Meridian Studies, 3(2), 95–103.
Sandireddy, R., Yerra, V. G., Komirishetti, P., Areti, A., & Kumar, A. (2016). Fisetin imparts neuroprotection in experimental diabetic neuropathy by modulating Nrf2 and NF-κB pathways. Cellular and Molecular Neurobiology, 36(6), 883–892.
Farshid, A. A., & Tamaddonfard, E. (2015). Histopathological and behavioral evaluations of the effects of crocin, safranal and insulin on diabetic peripheral neuropathy in rats. Avicenna Journal of Phytomedicine, 5(5), 469–478.
Li, M., Huang, D., Liu, X., & Lin, L. (2015). Tang-Tong-Fang confers protection against experimental diabetic peripheral neuropathy by reducing inflammation. Evidence-based Complementary and Alternative Medicine, 2015, 574169.
Kiyatkin, E. A., & Brown, P. L. (2005). Brain and body temperature homeostasis during sodium pentobarbital anesthesia with and without body warming in rats. Physiology & Behavior, 84(4), 563–570.
Lei, T., Jing, D., Xie, K., Jiang, M., Li, F., Cai, J., Wu, X., Tang, C., Xu, Q., Liu, J., Guo, W., Shen, G., & Luo, E. (2013). Therapeutic effects of 15 Hz pulsed electromagnetic field on diabetic peripheral neuropathy in streptozotocin-treated rats. PLoS One, 8(4), e61414.
Li, G., Sun, C., Wang, Y., Liu, Y., Gang, X., Gao, Y., Li, F., Xiao, X., & Wang, G. (2014). A clinical and neuropathological study of Chinese patients with diabetic peripheral neuropathy. PLoS One, 9(3), e91772.
Zhao, J. L., & Li, Z. R. (2008). Clinical observation on mild-warm moxibustion for treatment of diabetic peripheral neuropathy. Zhongguo Zhen Jiu, 28(1), 13–16.
Sun, Y. Z., & Liu, T. T. (2005). Comparison of therapeutic effects of acupuncture and moxibustion on diabetic peripheral neuropathies. Zhongguo Zhen Jiu, 25(8), 539–541.
Zafar, M., & Naqvi, S. N. (2009). Altered liver morphology and enzyme in streptozotocin induced diabetic rats. International Journal of Morphology, 27, 719–725.
Xiong, X., Elkind-Hirsch, K. E., Xie, Y., Delarosa, R., Maney, P., Pridjian, G., & Buekens, P. (2013). Periodontal disease as a potential risk factor for the development of diabetes in women with a prior history of gestational diabetes mellitus. Journal of Public Health Dentistry, 73(1), 41–49.
Almeida Abdo, J., Cirano, F. R., Casati, M. Z., Ribeiro, F. V., Giampaoli, V., Viana Casarin, R. C., & Pimentel, S. P. (2013). Influence of dyslipidemia and diabetes mellitus on chronic periodontal disease. Journal of Periodontology, 84(10), 1401–1408.
Daniel, R., Gokulanathan, S., Shanmugasundaram, N., Lakshmigandhan, M., & Kavin, T. (2012). Diabetes and periodontal disease. Journal of Pharmacy & Bioallied Sciences, 4(Suppl 2), S280–S282.
Monea, A., Mezei, T., & Monea, M. (2012). The influence of diabetes mellitus on periodontal tissues: a histological study. Romanian Journal of Morphology and Embryology, 53(3), 491–495.
Xu, M., Li, D., & Zhang, S. (2018). Acupuncture for acute stroke. Cochrane Database of Systematic Reviews, 3, CD003317.
Hinman, R. S., McCrory, P., Pirotta, M., Relf, I., Crossley, K. M., Reddy, P., Forbes, A., Harris, A., Metcalf, B. R., Kyriakides, M., Novy, K., & Bennell, K. L. (2012). Efficacy of acupuncture for chronic knee pain: protocol for a randomised controlled trial using a Zelen design. BMC Complementary and Alternative Medicine, 12(1), 161.
Yuan, Y. D., Zhao, X. P., Deng, Z. L., Song, Q. H., Zhang, L. Y., & Zhang, H. Y. (2015). Interference of strength training can obviously increase the effect of moxibustion treatment on patients with knee joints injuries. International Journal of Clinical and Experimental Medicine, 8(8), 13748–13754.
Xiao, A. J., He, L., Ouyang, X., Liu, J. M., & Chen, M. R. (2018). Comparison of the anti-apoptotic effects of 15- and 35-min suspended moxibustion after focal cerebral ischemia/reperfusion injury. Neural Regeneration Research, 13(2), 257–264.
Ganesh Yerra, V., Negi, G., Sharma, S. S., & Kumar, A. (2013). Potential therapeutic effects of the simultaneous targeting of the Nrf2 and NF-κB pathways in diabetic neuropathy. Redox Biology, 1(1), 394–397.
Negi, G., Kumar, A., & Sharma, S. S. (2011). Nrf2 and NF-κB modulation by sulforaphane counteracts multiple manifestations of diabetic neuropathy in rats and high glucose-induced changes. Current Neurovascular Research, 8(4), 294–304.
Li, W., Khor, T. O., Xu, C., Shen, G., Jeong, W. S., Yu, S., & Kong, A. (2008). Activation of Nrf2-antioxidant signaling attenuates NFkappaB-inflammatory response and elicits apoptosis. Biochemical Pharmacology, 76(11), 1485–1489.
Negi, G., Kumar, A., & Sharma, S. S. (2011). Melatonin modulates neuroinflammation and oxidative stress in experimental diabetic neuropathy: effects on NF-κB and Nrf2 cascades. Journal of Pineal Research, 50(2), 124–131.
Kumar, A., Negi, G., & Sharma, S. S. (2012). Suppression of NF-κB and NF-κB regulated oxidative stress and neuroinflammation by BAY 11-7082 (IκB phosphorylation inhibitor) in experimental diabetic neuropathy. Biochimie., 94(5), 1158–1165.
Schara, R., Skaleric, E., Seme, K., & Skaleric, U. (2013). Prevalence of periodontal pathogens and metabolic control of type 1 diabetes patients. Journal of the International Academy of Periodontology, 15(1), 29–34.
Wang, X. M., Lehky, T. J., Brell, J. M., & Dorsey, S. G. (2012). Discovering cytokines as targets for chemotherapy-induced painful peripheral neuropathy. Cytokine., 59(1), 3–9.
Funding
This study was funded by the Foundation of Hubei Provincial Department of Education of China (No. D20142006) and the Acupuncture and Moxibustion Research Team of Hubei University of Traditional Chinese Medicine Project (No. 2017ZXZ004) and supported by a project grant from NSFC (Natural Science Foundation of China, Nos. 81804165 and 81574065).
Author information
Authors and Affiliations
Contributions
Hua Wang and H. T. Tang conceived and designed the experiments. Jia Li and Huanjiao Zhou performed the experiments. X. Hu analyzed the data. Jianmin Liu and F.X. Liang contributed reagents/materials/analysis tools. Jia Li wrote the paper. Jiaoping Liu helped revise the paper.
Corresponding authors
Ethics declarations
The animal experiment protocol was approved by the Animal Ethics Committee of Hubei University of Chinese Medicine in China.
Conflict of Interest
The authors declare that there is no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Li, J., Hu, X., Liang, F. et al. Therapeutic effects of moxibustion simultaneously targeting Nrf2 and NF-κB in diabetic peripheral neuropathy. Appl Biochem Biotechnol 189, 1167–1182 (2019). https://doi.org/10.1007/s12010-019-03052-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-019-03052-8