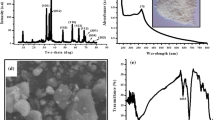
Abstract
In this work, zinc oxide nanoparticles-loaded calcium alginate films were investigated for their moisture uptake behavior at different temperatures. The equilibrium uptake data was interpreted quantitatively by GAB isotherm models. The monolayer moisture contents were 0.301 ± 0.003, 0.0214 ± 0.092, and 0.171 ± 0.102 at 20, 30, and 37°C, respectively. The water vapor transmission rate was found to be 0.816 ± 0.143, 1.42 ± 0.045, and 1.632 ± 0.064 g s−1 m−2 respectively. For the moisture content range of 0.2 to 0.6, the net ∆H and ∆S values were found to be 22.73 to 11.14 KJ/mol and 0.064 to 0.034 KJ/mol/K, respectively. The moisture uptake of films increased with water activity but showed negative temperature dependence. The enthalpy of sorption (∆H) and differential entropy (∆S) were determined at different moisture content values, ranging from 0.2 to 0.6 g/g db. The two parameters showed a higher degree of correlation. The equilibrium moisture content data was used to evaluate harmonic mean temperature T hm. Finally, the biocidal action of films was tested against model bacteria Escherichia coli.







Similar content being viewed by others
References
Alhamdan, A. M., & Hassan, B. H. (1999). Water sorption isotherms of Date Pasres as influenced by Date cultivar and storage temperature. Journal of Food Engineering, 39, 301–306.
ASTM. (1993). Standard test method for water vapor transmission of materials. Designation E, 96–93, 701–708.
Bajpai, S. K., & Tankhiwale, R. (2007). Preparation, characterization preliminary calcium release study of floating sodium alginate/dextran based hydrogel beads: Part-1. Polymer International, 56(1), 57–65.
Bellagha, S., Sahli, A., & Farhat, A. (2008). Desorption isotherms and isosteric heat of three Tunisian date cutivars. Food and Bioprocesses Technology, 1(4), 270–275.
Bourtoom, T. (2008). Edible films and coatings. International Food Research Journal, 15(3), 237–248.
Bourtoom, T., & Chinnan, M. S. (2008). Preparation and properties of rice–starch–chitosan blend biodegradable film. LWT Food Science and Technology, 41, 1633–1641.
Bozdemir, O. A., & Tutas, M. (2003). Plasticizer effect on water vapour permeability properties of Locust bean gum-based edible films. Turkish Journal of Chemistry, 27, 773–782.
Braccine, I., & Perez, S. (2001). Molecular basis of Ca2+ induced gelation in alginates and pectins: The egg box model revisited. Biomacromolecules, 2, 1089–1096.
Bruno, M., Giancone, T., Torrieri, E., Masi, P., & Moresi, M. (2008). Engineering properties of edible transglutaminase cross-linked caseinate based films. Food and Bioprocesses Technology, 1(4), 393–404.
Campo, E. J. A., Peiteado, M., Caballero, A. C., & Villegas, M. (2009). Room temperature synthesis of high purity 2D ZnO nanoneedles. Journal of Ceramic Processing Research, 10(4), 477–481.
Caykara, T., Demirci, S., Erogke, M. S., & Guven, O. (2005). Poly(ethylene oxide) and its blends with sodium alginate. Polymer, 46(24), 10750–10757.
Chaibi, A., Ababouch, L. H., Belasri, K., Boucetta, S., & Busta, F. F. (1997). Inhibition of germination and vegetative growth of Bacillus cereus T and Clostridium botulinum 62A spores by essential oils. Food Microbiology, 14, 161–174.
Chindaduang, A., Darangkaew, P., Pratontep, S., & Tumcharern, G. (2009). Structural, optical and photovoltaic properties of ZnO-MWCNTs electrodes. Journal of Microscopy Society of Thailand, 23(1), 115–118.
Conte, A., Serpanza, B., Sinigaglia, M., & Del Nobbile, M. A. (2007). Use of lemon extract to inhibit the growth of malolactic bacteria. Journal of Food Protection, 70(1), 114–118.
Dutta, P. K., Tripathi, S., Mehrotra, G. K., & Dutta, J. (2009). Prespectives for chitosan based antimicrobial films in food applications. Food Chemistry, 114, 1173–1182.
Ertesvag, H., & Valla, S. (1998). Biosynthesis and applications of alginates. Polymer Degradation and Stability, 59, 85–91.
Eswaranandam, S., Hettiarachychy, N. S., & Johnson, M. G. (2004). Antimicrobial activity of citric, lactic, malic or tartaric acids and nisin incorporated soy protein film against Listeria monocytogenes, Escherichia coli O157:H7, and Salmonella gaminara. Journal of Food Science, 69(3), 79–84.
Fang, M., Chen, J. H., Xu, X. L., Yang, P. H., & Hildebrand, H. F. (2006). Antibacterial activity of on inorganic agents on six bacteria associated with oral infection s by two susceptibility test. International Journal of Antimicrobial Agents, 26, 513–517.
Farahnaky, A., Ansari, S., & Maizoobi, M. (2009). Effect of glycerol on the moisture sorption isotherms of figs. Journal of Food Engineering, 93, 468–473.
Gabas, A. L., Telis, V. R. N., Sobral, P. J. A., & Telis-Romero, J. (2007). Effect of maltodextrin and Arabic gum in water vapour sorption thermodynamic properties of vacuum dried pineapple pulp powder. Journal of Food Engineering, 82, 246–252.
Gucbilmez, C. M., Yemenicioglu, A., & Arslanoglu, A. (2007). Antimicrobial and antioxidant activity of edible zein films incorporated with lysozyme, albumin proteins and disodium EDTA. Food Research International, 40, 80–91.
Hailwood, A. J., & Horrobin, S. (1946). Absorption of water by polymers: Analysis in terms of a simple model. Transaction of Faraday Society, 42B, 84–92.
Hong, S. I., & Krochta, J. M. (2004). Whey protein isolates coating LDPE film as a novel oxygen barrier in the composite structure. Packaging Technology and Science, 17(1), 13–21.
Jones, N., Ray, B., Ranjit, K. T., & Manna, A. C. (2007). Antibacterial activity of ZnO nanoparticle suspensions on a broad spectrum of micro-organism. FEMS Microbiology Letters, 279, 71–76.
Khalloufi, S., Giasson, J., & Ratti, C. (2000). Water activity of freeze dried mushrooms and berries. Canadian Agricultural Engineering, 42(1), 7.1–7.13.
Kim, K. M., Marx, D. B., Weller, C. L., & Hanna, M. A. J. (2003). Influence of sorghum wax, glycerin and sorbitol on physical properties of soy protein isolate films. Journal of the American Oil Chemists’ Society, 80(1), 71–76.
Kohen, R., & Nyska, A. (2002). Oxidation of biological system: oxidative stress phenomena, antioxidants, redox reactions, and methods for their quantification. Toxicologic Pathology, 31, 620–650.
Krug, R. R., Hunter, W. G., & Grieger, R. A. (1976). Enthalpy–entropy compensation, some fundamental statistical problems associated with the analysis of Vant Hoff and Arrhenius data. The Journal of Physical Chemistry, 80, 2335–2341.
Leffler, J. E. (1955). The enthalpy entropy relationship and its implication for organic chemistry. The Journal of Organic Chemistry, 20, 1202–1231.
Li, C., Li, Y., Luo, L., & Fu, M. (2008). Synthesis and characterization of ZnO microcrystal tubes. Journal of the Chilean Chemical Society, 53(3), 1615–1616.
Mutasem, O., Taha, W. N., Adel, A., & Hatim Alkhatib, S. (2008). Sodium lauryl sulphate impedes drug release from zinc crosslinked alginate beads: Switching from enteric coating release into biphasic profiled. International Journal of Pharmaceutics, 350, 291–300.
Olivas, G. I., & Barbosa-Canovas, G. V. (2008). Water vapour permeability and mechanical properties as affected by plasticizer and relative humidity. LWT Food Science and Technology, 41, 359–566.
Oluwamukomi, M. O. (2009). Adsorption isotherm modeling of soy-melon enriched and un-enriched ‘gari’ using GAB equation. African Journal of Food Science, 3(5), 117–124.
Oluwamukomi, M. O., Adeyemi, I. A., & Odeyemi, O. O. (2008). Adsorption isotherm and thermodynamic characteristics of soy melon enriched “Gari” semolina from cassava. Agricultural Engineering International, 10, 1–17.
Padmawathy, N., & Vijayaraghavan, R. (2008). Enhanced bioactivity of ZnO nanoparticles—an antimicrobial study. Science and Technology of Advanced Materials, 2008(9), 1–17.
Perez-Alonso, C., Beristain, C. I., Lobato-Calleras, C., Rodriguez-Huezo, M. E., & Vernon-Carter, E. J. (2006). Thermodynamic analysis of the sorption isotherms of pure and blended carbohydrate polymers. Journal of Food Engineering, 77(4), 753–760.
Qin, Y., Zhu, C., Chen, Y., & Zhang, C. (2006). The absorption and release of silver and zinc ions by chitosan fibers. Journal of Applied Polymer Science, 101, 766–771.
Reinose, E., Mittal, S. G., & Lim, L. T. (2008). Influence of whey protein composite coatings on plum (Prunus domestica L) Fruit quality. Food and Bioprocesses Technology, 1(4), 314–325.
Salleh, E., Muhamad, I., & Khairuddin, N. (2007). Preparation, characterization and antimicrobial analysis of antimicrobial starch based film incorporated with chitosan and lauric acid. Asian Chitin Journal, 3, 55–68.
Sharma, P., Singh, R. R. B., Singh, A. K., Patel, A. A., & Patil, G. R. (2009). Sorption isotherms and thermodynamics of water sorption of ready to use Basundi mix. LWT Food Science and Technology, 42, 441–445.
Shankar, T. J., Sokhannsanj, S., Bandhopahya, S., & Bawa, A. S. (2010). Storage properties of low fat and rice flour coextrudates. Food and Bioprocess Technology, 3(4), 481–490.
Shi, L., Zhou, J., & Gunasekaran, S. (2008). Low temperature fabrication of ZnO whey protein isolate nanocomposite. Material Letters, 62, 4383–4385.
Singh, M., Singh, S., Prasad, S., & Gambhir, I. S. (2008). Nanotechnology in medicine and antimicrobial effect of silver nanoparticles. Digest Journal of Nonmaterial and Biostructures, 3(3), 115–122.
Sondi, I., & Salopek-Sondi, B. (2004). Silver nanoparticles as antibacterial agent: A case study on E. coli as a model for gram negative bacteria. Journal of Colloid and Interface Science, 275, 177–182.
Tam, K. H., Djurisic, A. B., Chan, C. M. N., Xi, Y. Y., Tse, C. W., Leung, Y. H., et al. (2008). Antibacterial activity of ZnO nanorods prepared by a hydrothermal method. Thin Solid Films, 516(18), 6167–6174.
Wang, N., & Brennan, J. G. (1991). Moisture sorption characteristics of potatoes at four temperatures. Journal of Food Engineering, 14, 269–287.
Yadav, A., Prasad, V., Kathe, A. A., Raj, S., Yadav, D., Sundarmoorthy, C., et al. (2006). Functional finishing in cotton fabrics using zinc oxide nanoparticles. Bulletin of Material Science, 29(6), 641–645.
Yao Clement, Y. B., & Tano, K. (2008). Experimental determination of the sorption isotherm of “Beta” lactose, new water absorbent and sodium bicarbonate. Journal of Food Technology, 6(4), 152–157.
Acknowledgment
We thank Dr. O. P. Sharma, Prof. and Head of the Department of the Chemistry, at Govt. Model Science College, Jabalpur, India, to provide experimental facilities. We are also thankful to Dr. Rajshree Kapoor, Head of the Department of English, for proof reading and making appropriate corrections throughout the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bajpai, S.K., Chand, N. & Chaurasia, V. Nano Zinc Oxide-Loaded Calcium Alginate Films with Potential Antibacterial Properties. Food Bioprocess Technol 5, 1871–1881 (2012). https://doi.org/10.1007/s11947-011-0587-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11947-011-0587-6