Abstract
Purpose of Review
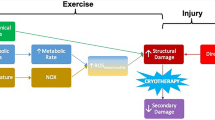
Here, we review potential causes of muscle dysfunction seen in many patients with myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) such as the effects of oxidative and nitrosative stress (O&NS) and mitochondrial impairments together with reduced heat shock protein production and a range of metabolic abnormalities.
Recent Findings
Several studies published in the last few years have highlighted the existence of chronic O&NS, inflammation, impaired mitochondrial function and reduced heat shock protein production in many patients with ME/CFS. These studies have also highlighted the detrimental effects of chronically elevated O&NS on muscle functions such as reducing the time to muscle fatigue during exercise and impairing muscle contractility. Mechanisms have also been revealed by which chronic O&NS and or impaired heat shock production may impair muscle repair following exercise and indeed the adaptive responses in the striated muscle to acute and chronic increases in physical activity.
Summary
The presence of chronic O&NS, low-grade inflammation and impaired heat shock protein production may well explain the objective findings of increased muscle fatigue, impaired contractility and multiple dimensions of exercise intolerance in many patients with ME/CFS.
Similar content being viewed by others
References
Recently published papers of particular interest have been highlighted as: • Of importance
Brurberg KG, Fonhus MS, Larun L, Flottorp S, Malterud K. Case definitions for chronic fatigue syndrome/myalgic encephalomyelitis (CFS/ME): a systematic review. BMJ Open. 2014;4(2):e003973.
Sharpe M, Archard L, Banatvala J, Borysiewicz L, Clare A, David A. A report-chronic fatigue syndrome: guidelines for research. J R Soc Med. 1991;84:118–21.
Carruthers B, van de Sande M, De Meirleir K, Klimas N, Broderick G, Mitchell T, et al. Myalgic encephalomyelitis: international consensus criteria. J Intern Med. 2011;270:327–38.
Jason LA, Brown A, Clyne E, Bartgis L, Evans M, Brown M. Contrasting case definitions for chronic fatigue syndrome, myalgic encephalomyelitis/chronic fatigue syndrome and myalgic encephalomyelitis. Eval Health Prof. 2012;35(3):280–304.
Jason LA, Kot B, Sunnquist M, Brown A, Evans M, Jantke R, et al. Chronic fatigue syndrome and myalgic encephalomyelitis: toward an empirical case definition. Health Psychol Behav Med. 2015;3(1):82–93.
Jason LA, McManimen S, Sunnquist M, Brown A, Furst J, Newton JL, et al. Case definitions integrating empiric and consensus perspectives. Fatigue : Biomed, Health Behav. 2016;4(1):1–23.
Morris G, Maes M. Case definitions and diagnostic criteria for myalgic encephalomyelitis and chronic fatigue syndrome: from clinical-consensus to evidence-based case definitions. Neuro Endocrinol Lett. 2013;34:185–99.
• Morris G, Berk M, Galecki P, Walder K, Maes M. The neuro-immune pathophysiology of central and peripheral fatigue in systemic immune-inflammatory and neuro-immune diseases. Mol Neurobiol. 2015;53(2):1195–219. This paper reviews pathways and abnormalities underpinning the incapacitating fatigue suffered by many people suffering from a range of systemic and neuro-inflammatory disorders including ME/CFS.
Shan ZY, Kwiatek R, Burnet R, Del Fante P, Staines DR, Marshall-Gradisnik SM, et al. Progressive brain changes in patients with chronic fatigue syndrome: a longitudinal MRI study. J Magn Reson Imaging. 2016.
Barnden LR, Crouch B, Kwiatek R, Burnet R, Del Fante P. Evidence in chronic fatigue syndrome for severity-dependent upregulation of prefrontal myelination that is independent of anxiety and depression. NMR Biomed. 2015;28(3):404–13.
Hornig M, Montoya JG, Klimas NG, Levine S, Felsenstein D, Bateman L et al. Distinct plasma immune signatures in ME/CFS are present early in the course of illness. Sci Adv. 2015;1(1).
• Russell L, Broderick G, Taylor R, Fernandes H, Harvey J, Barnes Z, et al. Illness progression in chronic fatigue syndrome: a shifting immune baseline. BMC Immunol. 2016;17(1):1–11. This study involved patients diagnosed with ME/CFS according to very narrow criteria and the authors reported longitudinal changes in their immune profile with elevated levels of IL-8 in the recently afflicted but the opposite held in patients ill for more than 2 years. Similarly, early ME/CFS patients displayed low levels of IL-6 but the reverse was true in patients suffering from incapacitating fatigue for over 2 years.
Kerr JR. Gene profiling of patients with chronic fatigue syndrome/myalgic encephalomyelitis. Curr Rheumatol Rep. 2008;10(6):482–91.
Jason LA, Corradi K, Torres-Harding S, Taylor RR, King C. Chronic fatigue syndrome: the need for subtypes. Neuropsychol Rev. 2005;15(1):29–58.
Zhang L, Gough J, Christmas D, Mattey DL, Richards SCM, Main J, et al. Microbial infections in eight genomic subtypes of chronic fatigue syndrome/myalgic encephalomyelitis. J Clin Pathol. 2010;63(2):156–64.
Maletic V, Raison C. Integrated neurobiology of bipolar disorder. Front Psychiatr. 2014;5:98.
• Maes M. A new case definition of neuro-inflammatory and oxidative fatigue (NIOF), a neuroprogressive disorder, formerly known as chronic fatigue syndrome or myalgic encephalomyelitis: results of multivariate pattern recognition methods and external validation by neuro-immune biomarkers. Neuro Endocrinol Lett. 2015;36(4):320–9. This paper provides evidence demonstrating that ME/CFS is not a single illness with a unitary pathogenesis and pathophysiology. This paper outlines a new diagnostic schema offering a pathway to selecting a “subgroup ” of CFS patients which could form the basis of testing many of the explanations proposed to underpin the well-documented muscle pathology seen in many patients who have been afforded a CFS diagnosis.
Maes M, Twisk FN, Johnson C. Myalgic encephalomyelitis (ME), chronic fatigue syndrome (CFS), and chronic fatigue (CF) are distinguished accurately: results of supervised learning techniques applied on clinical and inflammatory data. Psychiatry Res. 2012;200(2-3):754–60.
• Giloteaux L, Goodrich JK, Walters WA, Levine SM, Ley RE, Hanson MR. Reduced diversity and altered composition of the gut microbiome in individuals with myalgic encephalomyelitis/chronic fatigue syndrome. Microbiomed. 2016;4(1):1–12. This is an important study as it builds on earlier work demonstrating the existence of intestinal dysbiosis in many CFS patients. This is significant as it potentially explains the presence of commensal antigens in the periheral blood of ME/CFS patients repeatedly reported by researchers. The chronic activation of pattern recognition receptors such as TLR-4 by bacterial antigens translocated from the gut could go some way to explaining the presence of activated inflammatory pathways and chronic O&NS in ME/CFS patients diagnosed according to international consensus guidelines.
• Shukla SK, Cook D, Meyer J, Vernon SD, Le T, Clevidence D, et al. Changes in gut and plasma microbiome following exercise challenge in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). PLoS One. 2015;10(12):e0145453. This study provided an avenue whereby patients with disuse muscle deconditioning could be differentiated from patients with ME/CFS as these changes in the commensal population and plasma LPS levels with exercise have not been reported in the former group of people. This increase in systemic markers of inflammation can also go some way to explaining the phenomenon of a worsening of global symptoms during and in the immediate aftermath of very slight increases in physical activity.
Maes M, Twisk FN, Kubera M, Ringel K, Leunis JC, Geffard M. Increased IgA responses to the LPS of commensal bacteria is associated with inflammation and activation of cell-mediated immunity in chronic fatigue syndrome. J Affect Disord. 2012;136(3):909–17.
Maes M, Mihaylova I, Leunis J-C. Increased serum IgA and IgM against LPS of enterobacteria in chronic fatigue syndrome (CFS): indication for the involvement of gram-negative enterobacteria in the etiology of CFS and for the presence of an increased gut–intestinal permeability. J Affect Disord. 2007;99(1-3):237–40.
• Morris G, Berk M, Klein H, Walder K, Galecki P, Maes M. Nitrosative stress, hypernitrosylation, and autoimmune responses to nitrosylated proteins: new pathways in neuroprogressive disorders including depression and chronic fatigue syndrome. Molec Neurobiol. 2016. This review examines recent studies investigating the adverse effect of dysfunctional nitric oxide signalling effect by reversible S-nitrosylation as a source of pathology. The mechanisms regulating this form of signalling breakdown in an environment of chronic O&NS and such a breakdown could explain in part many of the abnormalities observed in ME/CFS patients in the immunological, autoimmune and bioenergetic domains.
White PD, Goldsmith KA, Johnson AL, Potts L, Walwyn R, DeCesare JC, et al. Comparison of adaptive pacing therapy, cognitive behaviour therapy, graded exercise therapy, and specialist medical care for chronic fatigue syndrome (PACE): a randomised trial. Lancet. 2011;377(9768):823–36.
Sharpe M, Goldsmith KA, Johnson AL, Chalder T, Walker J, White PD. Rehabilitative treatments for chronic fatigue syndrome: long-term follow-up from the PACE trial. Lancet Psychiatr. 2015;2(12):1067–74.
Lavergne MR, Cole DC, Kerr K, Marshall LM. Functional impairment in chronic fatigue syndrome, fibromyalgia, and multiple chemical sensitivity. Can Fam Physician. 2010;56(2):e57–65.
Nacul LC, Lacerda EM, Campion P, Pheby D, Drachler Mde L, Leite JC, et al. The functional status and well being of people with myalgic encephalomyelitis/chronic fatigue syndrome and their carers. BMC Public Health. 2011;11:402.
Snell CR, Stevens SR, Davenport TE, Van Ness JM. Discriminative validity of metabolic and workload measurements for identifying people with chronic fatigue syndrome. Phys Ther. 2013;93(11):1484–92.
• Keller BA, Pryor JL, Giloteaux L. Inability of myalgic encephalomyelitis/chronic fatigue syndrome patients to reproduce VO(2)peak indicates functional impairment. J Transl Med. 2014;12:104. This study shows that repeated cardiopulmonary exercise tests may be employed as a clinical indicator for diagnosing ME/CFS. Moreover, ME/CFS patients are unable to reproduce physiological measures at maximal/ventilatory threshold.
Jammes Y, Steinberg JG, Delliaux S. Chronic fatigue syndrome: acute infection and history of physical activity affect resting levels and response to exercise of plasma oxidant/antioxidant status and heat shock proteins. J Intern Med. 2012;272(1):74–84.
• Rutherford G, Manning P, Newton JL. Understanding muscle dysfunction in chronic fatigue syndrome. J Aging Res. 2016;2016:2497348. Another mechanism that may explain muscle fatigue in patients with ME/CFS is the overutilisation of the lactate dehydrogenase pathway and slowed acid clearance after exercise.
Earl K, Sakellariou G, Owens D, Sinclair M, Fenech M, Close G et al. The role of cytokines in muscle fatigue in patients with chronic fatigue syndrome (CFS). The FASEB J. 2015;29(1 Supplement).
Fukuda K. The chronic fatigue syndrome: a comprehensive approach to its definition and study. Ann Intern Med. 1994;121(12):953.
He J, Hollingsworth KG, Newton JL, Blamire AM. Cerebral vascular control is associated with skeletal muscle pH in chronic fatigue syndrome patients both at rest and during dynamic stimulation. NeuroImage: Clin. 2013;2:168–73.
Nijs J, Aelbrecht S, Meeus M, Van Oosterwijck J, Zinzen E, Clarys P. Tired of being inactive: a systematic literature review of physical activity, physiological exercise capacity and muscle strength in patients with chronic fatigue syndrome. Disabil Rehabil. 2011;33(17-18):1493–500.
Davenport TE, Stevens SR, Baroni K, Van Ness JM, Snell CR. Reliability and validity of short form 36 version 2 to measure health perceptions in a sub-group of individuals with fatigue. Disabil Rehabil. 2011;33(25-26):2596–604.
Davenport TE, Stevens SR, Baroni K, Van Ness M, Snell CR. Diagnostic accuracy of symptoms characterising chronic fatigue syndrome. Disabil Rehabil. 2011;33(19-20):1768–75.
Davenport TE, Stevens SR, VanNess MJ, Snell CR, Little T. Conceptual model for physical therapist management of chronic fatigue syndrome/myalgic encephalomyelitis. Phys Ther. 2010;90(4):602–14.
Dimauro I, Mercatelli N, Caporossi D. Exercise-induced ROS in heat shock proteins response. Free Radical Biology Med. 2016.
Jammes Y, Steinberg J, Mambrini O, Bregeon F, Delliaux S. Chronic fatigue syndrome: assessment of increased oxidative stress and altered muscle excitability in response to incremental exercise. J Intern Med. 2005;257:299–310.
Jammes Y, Steinberg JG, Delliaux S, Bregeon F. Chronic fatigue syndrome combines increased exercise-induced oxidative stress and reduced cytokine and Hsp responses. J Intern Med. 2009;266(2):196–206.
Molvarec A, Rigó J, Lázár L, Balogh K, Makó V, Cervenak L, et al. Increased serum heat-shock protein 70 levels reflect systemic inflammation, oxidative stress and hepatocellular injury in preeclampsia. Cell Stress Chaperones. 2009;14(2):151–9.
McConnell KW, Fox AC, Clark AT, Chang N-YN, Dominguez JA, Farris AB, et al. The role of HSP70 in mediating age-dependent mortality in sepsis. J Immunol (Baltimore, Md : 1950). 2011;186(6):3718–25.
Fittipaldi S, Dimauro I, Mercatelli N, Caporossi D. Role of exercise-induced reactive oxygen species in the modulation of heat shock protein response. Free Radic Res. 2014;48(1):52–70.
Senf SM. Skeletal muscle heat shock protein 70: diverse functions and therapeutic potential for wasting disorders. Front Physiol. 2013;4:330.
Senf SM, Howard TM, Ahn B, Ferreira LF, Judge AR. Loss of the inducible Hsp70 delays the inflammatory response to skeletal muscle injury and severely impairs muscle regeneration. PLoS One. 2013;8(4):e62687.
Novak ML, Weinheimer-Haus EM, Koh TJ. Macrophage activation and skeletal muscle healing following traumatic injury. J Pathol. 2014;232(3):344–55.
Hindi SM, Kumar A. Toll-like receptor signalling in regenerative myogenesis: friend and foe. J Pathol. 2016;239(2):125–8.
Whiteside A, Hansen S, Chaudhuri A. Exercise lowers pain threshold in chronic fatigue syndrome. Pain. 2004;109(3):497–9.
Van Oosterwijck J, Nijs J, Meeus M, Lefever I, Huybrechts L, Lambrecht L, et al. Pain inhibition and postexertional malaise in myalgic encephalomyelitis/chronic fatigue syndrome: an experimental study. J Intern Med. 2010;268(3):265–78.
Meeus M, Roussel NA, Truijen S, Nijs J. Reduced pressure pain thresholds in response to exercise in chronic fatigue syndrome but not in chronic low back pain: an experimental study. J Rehabil Med. 2010;42(9):884–90.
Nijs J, Meeus M, Van Oosterwijck J, Ickmans K, Moorkens G, Hans G, et al. In the mind or in the brain? Scientific evidence for central sensitisation in chronic fatigue syndrome. Eur J Clin Invest. 2012;42(2):203–12.
Vecchiet J, Cipollone F, Falasca K, Mezzetti A, Pizzigallo E, Bucciarelli T. Relationship between musculoskeletal symptoms and blood markers of oxidative stress in patients with chronic fatigue syndrome. Neurosci Lett. 2003;335:151–4.
Light A, White A, Hughen R, Light K. Moderate exercise increases expression for sensory, adrenergic, and immune genes in chronic fatigue syndrome patients but not in normal subjects. J Pain. 2009;10:1099–112.
Light AR, Bateman L, Jo D, Hughen RW, Vanhaitsma TA, White AT, et al. Gene expression alterations at baseline and following moderate exercise in patients with chronic fatigue syndrome and fibromyalgia syndrome. J Intern Med. 2012;271(1):64–81.
Nijs J, Nees A, Paul L, De Kooning M, Ickmans K, Meeus M, et al. Altered immune response to exercise in patients with chronic fatigue syndrome/myalgic encephalomyelitis: a systematic literature review. Exerc Immunol Rev. 2014;20:94–116.
Kent-Braun J, Sharma K, Weiner M, Massie B, Miller R. Central basis of muscle fatigue in chronic fatigue syndrome. Neurology. 1993;43:125–31.
Schillings M, Kalkman J, van der Werf S, van Engelen B, Bleijenberg G, Zwartz M. Diminished central activation during maximal voluntary contraction in chronic fatigue syndrome. Clin Neurophysiol. 2004;115:2518–24.
Vermeulen R, Kurk R, Visser F, Sluiter W, Scholte H. Patients with chronic fatigue syndrome performed worse than controls in a controlled repeated exercise study despite a normal oxidative phosphorylation capacity. J Transl Med. 2010;8:93.
Morris G, Berk M. The many roads to mitochondrial dysfunction in neuroimmune and neuropsychiatric disorders. BMC Med. 2015;13(1):68.
Morris G, Maes M. Myalgic encephalomyelitis/chronic fatigue syndrome and encephalomyelitis disseminata/multiple sclerosis show remarkable levels of similarity in phenomenology and neuroimmune characteristics. BMC Med. 2013;11(1):205.
• Naviaux RK, Naviaux JC, Li K, Bright AT, Alaynick WA, Wang L, et al. Metabolic features of chronic fatigue syndrome. Proc Natl Acad Sci. 2016;113(37):E5472–80. The authors of this study reported a characteristic pattern of cellular hypometabolism characteristic of impaired mitochondrial function compared to normal subjects which has not been demonstrated in patients suffering from anxiety depression or disuse muscle deconditioning. The authors have proposed that their proteomic signature could provide the first objective test enabling a diagnosis of ME/CFS.
VanNess JM, Stevens SR, Bateman L, Stiles TL, Snell CR. Postexertional malaise in women with chronic fatigue syndrome. J Women’s Health (2002). 2010;19(2):239–44.
Fulle S, Pietrangelo T, Mancinelli R, Saggini R, Fano G. Specific correlations between muscle oxidative stress and chronic fatigue syndrome: a working hypothesis. J Muscle Res Cell Motil. 2007;28:355–62.
Jones D, Hollingsworth K, Taylor R, Blamire A, Newton J. Abnormalities in pH handling by peripheral muscle and potential regulation by the autonomic nervous system in chronic fatigue syndrome. J Intern Med. 2010;267:394–401.
Jones DE, Hollingsworth KG, Jakovljevic DG, Fattakhova G, Pairman J, Blamire AM, et al. Loss of capacity to recover from acidosis on repeat exercise in chronic fatigue syndrome: a case-control study. Eur J Clin Invest. 2012;42(2):186–94.
Morris G, Maes M. Oxidative and nitrosative stress and immune-inflammatory pathways in patients with myalgic encephalomyelitis (ME)/chronic fatigue syndrome (CFS). Curr Neuropharmacol. 2014;12:168–85.
McCully K, Natelson B. Impaired oxygen delivery to muscle in chronic fatigue syndrome. Clin Sci (Lond). 1999;97:603 - 8.
Wong R, Lopaschuk G, Zhu G, Walker D, Catellier D, Burton D. Skeletal muscle metabolism in the chronic fatigue syndrome. in vivo assessment by 31P nuclear magnetic resonance spectroscopy. Chest. 1992;102:1716–22.
Lane R, Barrett M, Taylor D, Kemp G, Lodi R. Heterogeneity in chronic fatigue syndrome: evidence from magnetic resonance spectroscopy of muscle. Neuromuscul Disord. 1998;8:204–9.
Barnes P, Taylor D, Kemp G, Radda G. Skeletal muscle bioenergetics in the chronic fatigue syndrome. J Neurol Neurosurg Psychiatry. 1993;56:679–83.
Fulle S, Mecocci P, Fano G, Vecchiet I, Vecchini A, Racciotti D, et al. Specific oxidative alterations in vastus lateralis muscle of patients with the diagnosis of chronic fatigue syndrome. Free Radic Biol Med. 2000;29(12):1252–9.
Reid MB, Moylan JS. Beyond atrophy: redox mechanisms of muscle dysfunction in chronic inflammatory disease. J Physiol. 2011;589(Pt 9):2171–9.
Reid MB, Lannergren J, Westerblad H. Respiratory and limb muscle weakness induced by tumor necrosis factor-alpha: involvement of muscle myofilaments. Am J Respir Crit Care Med. 2002;166(4):479–84.
Chambers MA, Moylan JS, Reid MB. Physical inactivity and muscle weakness in the critically ill. Crit Care Med. 2009;37(10 Suppl):S337–46.
Chambers MA, Moylan JS, Smith JD, Goodyear LJ, Reid MB. Stretch-stimulated glucose uptake in skeletal muscle is mediated by reactive oxygen species and p38 MAP-kinase. J Physiol. 2009;587(Pt 13):3363–73.
Michaelson LP, Iler C, Ward CW. ROS and RNS signaling in skeletal muscle: critical signals and therapeutic targets. Annu Rev Nurs Res. 2013;31:367–87.
Steinbacher P, Eckl P. Impact of oxidative stress on exercising skeletal muscle. Biomolecules. 2015;5(2):356–77.
Powers SK, Smuder AJ, Judge AR. Oxidative stress and disuse muscle atrophy: cause or consequence? Curr Opin Clin Nutrit Metab Care. 2012;15(3):240–5.
Powers SK, Jackson MJ. Exercise-induced oxidative stress: cellular mechanisms and impact on muscle force production. Physiol Rev. 2008;88(4):1243–76.
Reid MB. Nitric oxide, reactive oxygen species, and skeletal muscle contraction. Med Sci Sports Exerc. 2001;33(3):371–6.
• Debold EP. Potential molecular mechanisms underlying muscle fatigue mediated by reactive oxygen and nitrogen species. Front Physiol. 2015;6:239. This review highlights some of the recent efforts which may offer information on the underlying molecular basis of muscle fatigue, including increased O&NS. The author discusses that compelling evidence comes from studies showing that pre-treatment of intact muscle with ROS scavengers significantly attenuate O&NS and the development of fatigue. The author concludes that two important emerging sites are the regulatory protein troponin and the actin binding region of myosin.
Ferreira LF, Reid MB. Muscle-derived ROS and thiol regulation in muscle fatigue. J Appl Physiol. 2008;104(3):853–60.
Fabisiak JP, Ritov VB, Kagan VE. Reversible thiol-dependent activation of ryanodine-sensitive Ca2+ release channel by etoposide (VP-16) phenoxyl radical. Antioxid Redox Signal. 2000;2(1):73–82.
Gutierrez-Martin Y, Martin-Romero FJ, Inesta-Vaquera FA, Gutierrez-Merino C, Henao F. Modulation of sarcoplasmic reticulum Ca(2+)-ATPase by chronic and acute exposure to peroxynitrite. Europ J Biochem / FEBS. 2004;271(13):2647–57.
Arner ES, Holmgren A. Physiological functions of thioredoxin and thioredoxin reductase. Europ J Biochem / FEBS. 2000;267(20):6102–9.
Aronson D, Violan MA, Dufresne SD, Zangen D, Fielding RA, Goodyear LJ. Exercise stimulates the mitogen-activated protein kinase pathway in human skeletal muscle. J Clin Invest. 1997;99(6):1251–7.
Posterino GS, Cellini MA, Lamb GD. Effects of oxidation and cytosolic redox conditions on excitation-contraction coupling in rat skeletal muscle. J Physiol. 2003;547(Pt 3):807–23.
Smith MA, Reid MB. Redox modulation of contractile function in respiratory and limb skeletal muscle. Respir Physiol Neurobiol. 2006;151(2-3):229–41.
Moopanar TR, Allen DG. Reactive oxygen species reduce myofibrillar Ca2+ sensitivity in fatiguing mouse skeletal muscle at 37 degrees C. J Physiol. 2005;564(Pt 1):189–99.
Coirault C, Guellich A, Barbry T, Samuel JL, Riou B, Lecarpentier Y. Oxidative stress of myosin contributes to skeletal muscle dysfunction in rats with chronic heart failure. Am J Physiol Heart Circ Physiol. 2007;292(2):H1009–17.
Yamada T, Mishima T, Sakamoto M, Sugiyama M, Matsunaga S, Wada M. Oxidation of myosin heavy chain and reduction in force production in hyperthyroid rat soleus. J Appl Physiol (1985). 2006;100(5):1520–6.
Plant DR, Lynch GS, Williams DA. Hydrogen peroxide modulates Ca2+-activation of single permeabilized fibres from fast- and slow-twitch skeletal muscles of rats. J Muscle Res Cell Motil. 2000;21(8):747–52.
Kaneko M, Suzuki H, Masuda H, Yuan G, Hayashi H, Kobayashi A, et al. Effects of oxygen free radicals on Ca2+ binding to cardiac troponin. Jpn Circ J. 1992;56 Suppl 5:1288–90.
Powers SK, Ji LL, Kavazis AN, Jackson MJ. Reactive oxygen species: impact on skeletal muscle. Compre Physiol. 2011;1(2):941–69.
McKenna MJ, Bangsbo J, Renaud J-M. Muscle K+, Na+, and Cl− disturbances and Na+-K+ pump inactivation: implications for fatigue. J Appl Physiol. 2008;104(1):288–95.
Marechal G, Gailly P. Effects of nitric oxide on the contraction of skeletal muscle. Cell Molec Life Sci : CMLS. 1999;55(8-9):1088–102.
Pouvreau S, Allard B, Berthier C, Jacquemond V. Control of intracellular calcium in the presence of nitric oxide donors in isolated skeletal muscle fibres from mouse. J Physiol. 2004;560(Pt 3):779–94.
Viner RI, Williams TD, Schoneich C. Nitric oxide-dependent modification of the sarcoplasmic reticulum Ca-ATPase: localization of cysteine target sites. Free Radic Biol Med. 2000;29(6):489–96.
Viner RI, Krainev AG, Williams TD, Schoneich C, Bigelow DJ. Identification of oxidation-sensitive peptides within the cytoplasmic domain of the sarcoplasmic reticulum Ca2+-ATPase. Biochemistry. 1997;36(25):7706–16.
Hidalgo C, Sanchez G, Barrientos G, Aracena-Parks P. A transverse tubule NADPH oxidase activity stimulates calcium release from isolated triads via ryanodine receptor type 1 S-glutathionylation. J Biol Chem. 2006;281(36):26473–82.
Hart JD, Dulhunty AF. Nitric oxide activates or inhibits skeletal muscle ryanodine receptors depending on its concentration, membrane potential and ligand binding. J Membr Biol. 2000;173(3):227–36.
Heunks LM, Machiels HA, Dekhuijzen PN, Prakash YS, Sieck GC. Nitric oxide affects sarcoplasmic calcium release in skeletal myotubes. J Appl Physiol (1985). 2001;91(5):2117–24.
Gehlert S, Bungartz G, Willkomm L, Korkmaz Y, Pfannkuche K, Schiffer T, et al. Intense resistance exercise induces early and transient increases in ryanodine receptor 1 phosphorylation in human skeletal muscle. PLoS One. 2012;7(11):e49326.
Bellinger AM, Reiken S, Carlson C, Mongillo M, Liu X, Rothman L, et al. Hypernitrosylated ryanodine receptor calcium release channels are leaky in dystrophic muscle. Nat Med. 2009;15(3):325–30.
Suhr F, Gehlert S, Grau M, Bloch W. Skeletal muscle function during exercise—fine-tuning of diverse subsystems by nitric oxide. Int J Mol Sci. 2013;14(4):7109–39.
Boczkowski J, Lisdero C, Lanone S, Carreras M, Aubier M, Poderoso J. Peroxynitrite-mediated mitochondrial dysfunction. Biol Signals Recept. 2001;10:66–80.
Hambrecht R, Adams V, Gielen S, Linke A, Mobius-Winkler S, Yu J, et al. Exercise intolerance in patients with chronic heart failure and increased expression of inducible nitric oxide synthase in the skeletal muscle. J Am Coll Cardiol. 1999;33(1):174–9.
Evangelista AM, Kohr MJ, Murphy E. S-nitrosylation: specificity, occupancy, and interaction with other post-translational modifications. Antioxid Redox Signal. 2013;19(11):1209–19.
Velasquez DA, Martinez G, Romero A, Vazquez MJ, Boit KD, Dopeso-Reyes IG, et al. The central Sirtuin 1/p53 pathway is essential for the orexigenic action of ghrelin. Diabetes. 2011;60(4):1177–85.
Moon Y, Cao Y, Zhu J, Xu Y, Balkan W, Buys ES et al. GSNOR deficiency enhances in situ skeletal muscle strength, fatigue resistance and RyR1 S-nitrosylation without impacting mitochondrial content and activity. Antioxid Redox Signal. 2016.
Piantadosi CA. Regulation of mitochondrial processes by protein S-nitrosylation. Biochim Biophys Acta. 2012;1820(6):712–21.
Albertini M, Lafortuna C, Aguggini G. Effects of nitric oxide on diaphragmatic muscle endurance and strength in pigs. Exp Physiol. 1997;82(1):99–106.
Belia S, Pietrangelo T, Fulle S, Menchetti G, Cecchini E, Felaco M, et al. Sodium nitroprusside, a NO donor, modifies Ca2+ transport and mechanical properties in frog skeletal muscle. J Muscle Res Cell Motil. 1998;19(8):865–76.
Zhou LZ, Johnson AP, Rando TA. NF kappa B and AP-1 mediate transcriptional responses to oxidative stress in skeletal muscle cells. Free Radic Biol Med. 2001;31(11):1405–16.
Haldar SM, Stamler JS. S-nitrosylation: integrator of cardiovascular performance and oxygen delivery. J Clin Invest. 2013;123(1):101–10.
Hess DT, Matsumoto A, Kim SO, Marshall HE, Stamler JS. Protein S-nitrosylation: purview and parameters. Nat Rev Mol Cell Biol. 2005;6(2):150–66.
Wu C, Liu T, Chen W, Oka S, Fu C, Jain MR, et al. Redox regulatory mechanism of transnitrosylation by thioredoxin. Molec Cell Proteomics : MCP. 2010;9(10):2262–75.
Dodd SL, Gagnon BJ, Senf SM, Hain BA, Judge AR. Ros-mediated activation of NF-kappaB and Foxo during muscle disuse. Muscle Nerve. 2010;41(1):110–3.
Derbre F, Ferrando B, Gomez-Cabrera MC, Sanchis-Gomar F, Martinez-Bello VE, Olaso-Gonzalez G, et al. Inhibition of xanthine oxidase by allopurinol prevents skeletal muscle atrophy: role of p38 MAPKinase and E3 ubiquitin ligases. PLoS One. 2012;7(10):e46668.
Kroller-Schon S, Jansen T, Hauptmann F, Schuler A, Heeren T, Hausding M, et al. alpha1AMP-activated protein kinase mediates vascular protective effects of exercise. Arterioscler Thromb Vasc Biol. 2012;32(7):1632–41.
McArdle F, Spiers S, Aldemir H, Vasilaki A, Beaver A, Iwanejko L, et al. Preconditioning of skeletal muscle against contraction-induced damage: the role of adaptations to oxidants in mice. J Physiol. 2004;561(Pt 1):233–44.
Morgan M, Liu Z. Crosstalk of reactive oxygen species and NF-kappaB signaling. Cell Res. 2011;21:103–15.
Cobley JN, Bartlett JD, Kayani A, Murray SW, Louhelainen J, Donovan T, et al. PGC-1alpha transcriptional response and mitochondrial adaptation to acute exercise is maintained in skeletal muscle of sedentary elderly males. Biogerontology. 2012;13(6):621–31.
Wright DC, Han DH, Garcia-Roves PM, Geiger PC, Jones TE, Holloszy JO. Exercise-induced mitochondrial biogenesis begins before the increase in muscle PGC-1alpha expression. J Biol Chem. 2007;282(1):194–9.
Hawley JA, Lessard SJ. Exercise training-induced improvements in insulin action. Acta physiologica (Oxford, England). 2008;192(1):127–35.
Olesen J, Kiilerich K, Pilegaard H. PGC-1alpha-mediated adaptations in skeletal muscle. Pflugers Arch - Eur J Physiol. 2010;460(1):153–62.
Koves TR, Li P, An J, Akimoto T, Slentz D, Ilkayeva O, et al. Peroxisome proliferator-activated receptor-gamma co-activator 1alpha-mediated metabolic remodeling of skeletal myocytes mimics exercise training and reverses lipid-induced mitochondrial inefficiency. J Biol Chem. 2005;280(39):33588–98.
Wu Z, Puigserver P, Andersson U, Zhang C, Adelmant G, Mootha V, et al. Mechanisms controlling mitochondrial biogenesis and respiration through the thermogenic coactivator PGC-1. Cell. 1999;98(1):115–24.
Marshall HE, Hess DT, Stamler JS. S-nitrosylation: physiological regulation of NF-kappaB. Proc Natl Acad Sci U S A. 2004;101(24):8841–2.
Numajiri N, Takasawa K, Nishiya T, Tanaka H, Ohno K, Hayakawa W, et al. On-off system for PI3-kinase-Akt signaling through S-nitrosylation of phosphatase with sequence homology to tensin (PTEN). Proc Natl Acad Sci U S A. 2011;108(25):10349–54.
Kelleher ZT, Matsumoto A, Stamler JS, Marshall HE. NOS2 regulation of NF-kappaB by S-nitrosylation of p65. J Biol Chem. 2007;282(42):30667–72.
Qi SH, Hao LY, Yue J, Zong YY, Zhang GY. Exogenous nitric oxide negatively regulates the S-nitrosylation p38 mitogen-activated protein kinase activation during cerebral ischaemia and reperfusion. Neuropathol Appl Neurobiol. 2013;39(3):284–97.
Nunez M, Fernandez-Sola J, Nunez E, Fernandez-Huerta JM, Godas-Sieso T, Gomez-Gil E. Health-related quality of life in patients with chronic fatigue syndrome: group cognitive behavioural therapy and graded exercise versus usual treatment. A randomised controlled trial with 1 year of follow-up. Clin Rheumatol. 2011;30(3):381–9.
Twisk FN, Maes M. A review on cognitive behavorial therapy (CBT) and graded exercise therapy (GET) in myalgic encephalomyelitis (ME) / chronic fatigue syndrome (CFS): CBT/GET is not only ineffective and not evidence-based, but also potentially harmful for many patients with ME/CFS. Neuro Endocrinol Lett. 2009;30(3):284–99.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors
Funding
There was no specific financial support for this specific research.
Additional information
This article is part of the Topical Collection on Chronic Pain
Rights and permissions
About this article
Cite this article
Gerwyn, M., Maes, M. Mechanisms Explaining Muscle Fatigue and Muscle Pain in Patients with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS): a Review of Recent Findings. Curr Rheumatol Rep 19, 1 (2017). https://doi.org/10.1007/s11926-017-0628-x
Published:
DOI: https://doi.org/10.1007/s11926-017-0628-x