Abstract
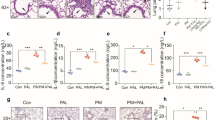
Emu oil is derived from the emu (Dromaius novaehollandiae), which originated in Australia, and has been reported to have anti-inflammatory properties. Inflammation was induced in anesthetized CD-1 mice by applying 50 μL of 2% croton oil to the inner surface of the left ear. After 2 h, the area was treated with 5μL of emu, fish, flaxseed, olive, or liquified chicken fat, or left untreated. Animals were euthanized at 6 h postapplication of different oils, and earplugs (FP) and plasma samples were collected. Inflammation was evaluated by change in earlobe thickness, increase in weight of EP tissue (compared to the untreated ear), and induction in cytokines interleukin (IL)-1α and tumor necrosis factor-α (TNF-α) in EP homogenates. Al-though reductions relative to control (croton oil) were noted for all treatments, auricular thickness and EP weights were, significantly reduced (−72 and −71%, respectively) only in the emu oil-treated group. IL-1α levels in homogenates of auricular tissue were significantly reduced in the fish oil (−57%) and emu oil (−70%) groups relative to the control group. The cytokine TNF-α from auricular homogenates was significantly reduced in the olive oil (−52%) and emu oil (−60%) treatment groups relative to the control group. Plasma cytokine levels were not changed by croton oil treatment. Although auricular thickness and weight were significantly correlated with each other (r=0.750, P<0.003), auricular thickness but not weight was significantly correlated with cytokine IL-1α (r=0.750, P<0.006) and TNF-α (r=0.690, P<0.02). These studies indicate that topical emu oil has anti-inflammatory properties in the CD-1 mouse that are associated with decreased auricular thickness and weight, and with the cytokines IL-1α and TNF-α.
Similar content being viewed by others
Abbreviations
- EP:
-
ear plug
- IL:
-
interleukin
- TNF-α:
-
tumor necrosis factor-α
References
Lopez, A., Sims, E.D., Ablett, F.R., Skinner, E.R., Leger, W.L., Lariviere, M.C., Jamieson, A.L., Burnes, M.J., and Zawadska, G.G. (1999) Effect of Emu Oil on Auricular Inflammation Induced with Croton Oil in Mice, Am. J. Vet. Res. 60, 1558–1561.
Snowden, J.M., and Whitehouse, M.W. (1997) Anti-inflammatory Activity of Emu Oil in Rats, Inflammopharmacology 5, 127–132.
Whitehouse, M.W., and Turner, A.G. (1997) Emu Oil(s): A Source of Non-toxic Transdermal Anti-inflammatory Agents in Aborginal Medicine, Am. Emu Assoc. Newslett. 6, 15–16.
Politis, M.J., and Dmytrowich, A. (1998) Promotion of Second Intention Wound Healing by Emu Oil Lotion: Comparative Results with Furasin, Polysporin and Cortisone, Plast. Reconstr. Surg. 102, 2404–2407.
Balto, K., Sasaki, H., and Stashenko, P. (2001) Interleukin-6 Deficiency Increases Inflammatory Bone Destruction, Infect. Immun. 69, 744–750.
Hou, L., Sasaki, H., and Stashenko, P. (2000) B-Cell Deficiency Predisposes Mice to Disseminating Anaerobic Infections: Protection by Passive Antibody Transfer, Infect. Immun. 68, 5645–5651.
Wang, C.Y., and Stashenko, P. (1993) The Role of Interleukin-1α in the Pathogenesis of Periapical Bone Destruction in a Rat Model, Oral Microbiol. Immunol. 8, 50–56.
Blok, W.L., Katan, M.B., and Van Der Meer, J.W.M. (1996) Modulation of Inflammation and Cytokine Production by Dietary (n−3) Fatty Acids, J. Nutr. 126, 1515–1533.
James, M.J., Gibson, R.A., and Cleland, L.G. (2000) Dietary Polyunsaturated Fatty Acids and Inflammatory Mediator Production, Am. J. Clin. Nutr. 71, (Suppl.), 343S-348S.
Tubaru, A., Dri, P., and Delbello, G. (1985) The Croton Oil Ear Test Revisited, Agents Actions 17, 347–349.
Leazer, T.M., Barbee, B., Ebron-McCoy, M., Henry-Sam, G.A., and Rogers, J.M. (2002) Role of the Maternal Acute Phase Response and Tumor Necrosis Factor Alpha in the Developmental Toxicity of Lipopolysaccharide in the CD-1 Mouse, Reprod. Toxicol. 16, 173–179.
Soybir, G.R., Koyuncu, H., Koksoy, F., Yalcin, O., Ozsker, A., Alatli, C., and Topuzlu, C. (1996) Protective Effect of Desferrioxamin Against TPA Caused Inflammation in CD-1 Mouse Skin, Surg. Oncol. 5, 253–258.
Skisak, C. (1991) The Role of Chronic Acanthosis and Sub Acute Inflammation in Tumor Promotion in CD-1 Mice by Petroleum Middle Distillate, Toxicol. Appl. Pharmacol. 109, 399–411.
Huang, M.T., Ho, C.T., Wang, Z.Y., Ferraro, T., Finnegan-Olive, T., Lou, Y.R., Mitchell, J.M., Laskin, J.D., Newmark, H., Yang, C.S., and Conney, A.H. (1992) Inhibitory Effect of Topical Application of a Green Tea Polyphenol Fraction on Tumour Initiation and Promotion in Mouse Skin, Carcinogenesis 13, 947–954.
Kunkel, S.L., Ogawa, H., Ward, P.A., and Zurler, R.B. (1982) Suppresion of Chronic Inflammation by Evening Primrose Oil, Prog. Lipid. Res. 20, 885–888.
Hamsen, M., Lerche, A., Kassis, V., Lorenzen., J., and Sondergaard, J. (1983) Treatment of Rheumatoid Arthritis with Prostaglandin E1, Precursors, cis-Linolenic Acid and γ-Linolenic Acid, Scand. J. Rheumatol 12, 85–88.
Jantti, J., Nikkari, T., Solakivi, T., Vapaatalo, H., and Isomaki, H. (1989) Evening Primose, Oil in Rheumatoid Arthritis: Changes in Serum Lipid and Fatty Acids, Annals Rheum. Dis. 48, 124–127.
Smiley, T.S., Kaplan, M.H., and Grusby, M.J. (1997) Immunoglobulin E Production in the Absence of Interleukin-4-Secreting CD1-Dependent Cells, Science 275, 977–983.
Koyuncu, H., Berkarda, B., Baykut, F., Soybir, G., Alatli, C., Gul, H., and Altun, M. (1999) Preventive Effect of Hesperidin Against Inflammation in CD-1 Mouse Skin Caused by Tumor Promoter, Anticancer Res. 19, 3237–3241.
Berger, A., Monnard, I., Baur, M., Charbonnet, C., Safonova, I., and Jomard, A. (2002) Epidermal Anti-inflammatory Properties of 5,11,14, 20∶3: Effects on Mouse Ear Edema, PGE2 Levels in Cultured Keratinocytes and PPAR Activation, Lipids Health Dis. 1, 12–17.
Venkatraman, T.J., Chandrasekar, B., Weintraub, S.T., and Fernandes, G. (1995) Differential Effects of ω-6 and ω-3 Fatty Acids in Interleukin-2 Production and mRNA Expression by EL-4.IL-2 Cells, J. Nutr. Biochem. 6, 467–473.
Hughes, D.A., and Pinder, A.C. (2000) n−3 Polyunsaturated Fatty Acids Inhibit the Antigen-Presenting Function of Human Monocytes, Am. J. Clin. Nutr. 71, (Suppl.), 357S-360S.
Tomobe, Y.I., Morizawa, K., Tsuchida, M., Hibino, H., Nakano, Y., and Tanaka, Y. (2000) Dietary Docosahexaenoic Acid Suppresses Inflammation and Immunoresponses in Contact Hypersensitivity Reaction in Mice, Lipids 35, 61–69.
Puliti, M., Von Hunolstein, C., Verwaerde, C., Bistoni, F., Orefici, G., and Tissi, L. (2002) Regulatory Role of Interleukin-10 in Experimental Group B Streptococcal Arthritis, Infect. Immun. 70, 2862–2868.
Instute of Laboratory Animal Research, 1996, Guide for the Care and Use of Laboratory Animals, 140 pp. National Academies Press, Washington, D.C.
Matsumoto, K., Fujimoto, M., Ito, K., Tanaka, H., and Hirano, I. (1990) Comparison of the Effects of Biolobol and 12-O-Tetradecanoylphenol-13-acetate on Skin, and Test of Tumor Promoting Potential of Bilobol in CD-1 Mice, Toxicol. Sci. 15, 39–46.
Wixson, S.K. (1994) Rabbits and Rodents: Anesthesia and Analgesia, in Research Animal Anesthesia, Analgesia and Surgery (Smith, A.C., and Swindle, M.M., eds.), Scientist Center for Animal Welfare, Greebelt, Maryland, pp. 59–71.
Chong, K.S., Nicolosi, R.J., Rodger, R.F., Arrigo, D.A., Juan, R.W., Mackey, J.J., Georas, S., and Herbert, P. (1987) Effect of Dietary Fat Saturation on Plasma Lipoproteins and High Density Lipoprotein Metabolism of the Rhesus Monkey, J. Clin. Invest. 79, 675–683.
Nicolosi, R.J., Wilson, T.A., Rogers, E.J., and Kritchevsky, D. (1998) Effects of Specific Fatty Acids (8∶0, 14∶0, cis-18∶1, trans-18∶1) on Plasma Lipoproteins, Atherogenic Potential and LDL Oxidative Properties in the Hamster, J. Lipid Res. 39, 1972–1980.
Snedecor, G.W., and Cochran, W.G. (1980) Statistical Methods, The Iowa State University Press, Ames, Iowa.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Yoganathan, S., Nicolosi, R., Wilson, T. et al. Antagonism of croton oil inflammation by topical emu oil in CD-1 mice. Lipids 38, 603–607 (2003). https://doi.org/10.1007/s11745-003-1104-y
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11745-003-1104-y