Abstract
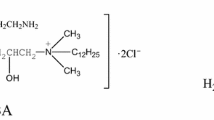
The surface properties of a series of imidazoline-based dissymmetric bis-quaternary ammonium (DBA) salts with different hydrophobic chain lengths DBA-12, DBA-14 and DBA-16 were determined. The adsorption behaviors of these compounds in 1 M HCl solution and their inhibitive effect on Q235 steel (U12350) were investigated using the weight-loss method, polarization, and electrochemical impedance spectroscopy. Results indicate that the three inhibitors all showed good inhibition performance for Q235 Steel in 1 M HCl solution and were found to be mixed-type inhibitors. The adsorption behavior of the three inhibitors can fit the Langmuir isotherm equation and the values of \( \Updelta G_{\text{ads}}^{0} \) are around or lower than 40 kJ mol−1, indicating that there is a stronger chemical adsorption.








Similar content being viewed by others
References
Bentiss F, Bouanis M, Mernari B, Traisnel M, Vezin H, Lagrenée M (2007) Understanding the adsorption of 4H–1,2,4-triazole derivatives on mild steel surface in molar hydrochloric acid. Appl Surf Sci 253:3696–3704
Abboud Y, Abourriche A, Saffaj T, Berrada M, Charrouf M, Bennamara A, Cherqaoui A, Takky D (2006) The inhibition of mild steel corrosion in acidic medium by 2,2′-bis(benzimidazole). Appl Surf Sci 252:8178–8184
Khaled KF, Hackerman N (2003) Investigation of the inhibitive effect of ortho-substituted anilines on corrosion of iron in 1 M HCl solutions. Electrochim Acta 48:2715–2723
Negm NA, Morsy SMI, Said MM (2005) Corrosion inhibition of some novel hydrazone derivatives. J Surfactant Deterg 8:95–98
Negm NA, El-Farragry AF, Abdelrahman NR (2011) New Schiff base cationic surfactants: surface and thermodynamic properties and applicability in bacterial growth and metal corrosion prevention. J Surfactant Deterg 14:505–514
Popova A, Christov M, Raicheva S, Sokolova E (2004) Adsorption and inhibitive properties of benzimidazole derivatives in acid mild steel corrosion. Corros Sci 46:1333–1350
Solmaz R, Kardas G, Yazici B (2005) Inhibition effect of rhodanine for corrosion of mild steel in hydrochloric acid solution. Prot Met 41:581–585
Liu FG, Du M, Zhang J (2009) Electrochemical behavior of Q235 steel in saltwater saturated with carbon dioxide based on new imidazoline derivative inhibitor. Corros Sci 51:102–109
Knag M, Bilkova K, Gulbrandsen E, Carlsen P, Sjöblom J (2006) Langmuir-Blodgett films of dodecyltrimethylammonium bromide and octadecanol on iron. Deposition and corrosion inhibitor performance in CO2 containing brine. Corros Sci 48:2592–2613
Emregul KC, Kurtaran R, Atakol O (2003) An investigation of chloride-substituted Schiff bases as corrosion inhibitors for steel. Corros Sci 45:2803–2817
Quraishi MA, Rafiquee MZ (2007) Corrosion inhibition of aluminium in acid solutions by some imidazoline derivatives. J Appl Electrochem 37:1153–1162
Manuel EP, Crescencio OO-X, Natalya VL, Jonathan-Boanerge P-N (2011) Imidazolium, pyridinium and dimethyl-ethylbenzyl ammonium derived compounds as mixed corrosion inhibitors in acidic medium. J Surfactant Deterg 14:211–220
Chen Y, Hong T, Gopal M, Jepson WP (2001) EIS studies of a corrosion inhibitor behavior under multiphase flow conditions. Corros Sci 42:979–990
Zhu S, Cheng F, Wang J, Yu JG (2006) Anionic gemini surfactants: synthesis and aggregation properties in aqueous solutions. Colloids Surf A 281:35–39
Mohammad M, Ali RTB, Krister H (2011) Comparison of a cationic gemini surfactant and the corresponding monomeric surfactant for corrosion protection of mild steel in hydrochloric acid. J Surfactant Deterg 14:605–613
Zana R (2002) Adv. Dimeric and oligomeric surfactants. Behavior at interfaces and in aqueous solution: a review. Colloid Interface Sci 97:205–253
Mathias JH, Rosen MJ, Davenport L (2001) Fluorescence study of premicellar aggregation in cationic gemini surfactants. Langmuir 17:6148–6154
Achouri ME, Infante MR, Izquierdo F, Kertit S, Gouttaya HM, Nciri B (2001) Synthesis of some cationic gemini surfactants and their inhibitive effect on iron corrosion in hydrochloric acid medium. Corros Sci 43:19–35
Qiu LG, Xie AJ, Shen YH (2005) The adsorption and corrosion inhibition of some cationic gemini surfactants on carbon steel surface in hydrochloric acid. Corros Sci 47:273–278
El Achouri M, Kertit S, Gouttaya HM, Nciri B, Bensouda Y, Perez L, Infante MR, Elkacemi K (2001) Corrosion inhibition of iron in 1 M HCl by some gemini surfactants in the series of alkanediyl-α,ω-bis-(dimethyl tetradecyl ammonium bromide). Prog Org Coat 43:267–273
Qiu LG, Wang YM, Jiang X (2008) Synergistic effect between cationic gemini surfactant and chloride ion for the corrosion inhibition of steel in sulphuric acid. Corros Sci 50:576–582
Tsubone K, Arakawa Y, Rosen M (2003) Structural effects on surface activities and micellar properties of alkanediyl-α,ω-bis(sodium N-acyl-β-alaninate) gemini surfactants. J Colloid Interface Sci 262:516–524
Jiang B, Zhang J, Du M, Sun ZD (2009) Synthesis and inhibition performance of dissymmetric bis-quaternary ammonium with imidazoline ring. Fine Chemicals 26:761–764
Jiang LN, Du M, Du L (2005) Application of weak polarization technique for corrosion monitoring of metallic materials in seawater. Corros Sci Prot Technol 17:192–194
Kenichi S, Shin U, Mamoru T, Yuichiro T, Koji T (2008) Adsorption and micellization behavior of novel gluconamide-type gemini surfactants. J Colloid Interface Sci 318:440–448
Liu XP, Feng J, Zhang L, Gong QT (2010) Synthesis and properties of a novel class of anionic gemini surfactants with polyoxyethylene spacers. Colloids Surf A 362:39–46
Tsubone K, Arakawa Y, Rosen MJ (2003) Structural effects on surface activities and micellar properties of alkanediyl-α,ω-bis (sodium N-acyl-β-alaninate) gemini surfactants. J Colloid Interface Sci 262:516–524
Rosen MJ, Mathias JH, Davenport L (1999) Aberrant aggregation behavior in cationic gemini surfactants investigated by surface tension, interfacial tension, and fluorescence methods. Langmuir 15:7340–7346
Rosen MJ (2004) Surfactants and interfacial phenomena, 3rd edn. Wiley, New York 2004
Shi Z, Yang WG (1994) Synthesis of quaternary ammonium salt surfactants and determination of their physical chemical properties. Chem Res Chin Univ 15:1013–1016
Olivares-Xometl O, Likhanova NV, Dominggues-Aguilar MA, Arce E, Dorantes H, Lozada PA (2008) Synthesis and corrosion inhibition of α-amino acids alkylamides for mild steel in acidic environment. Mater Chem Phys 110:344–351
Maaya AK, Al-Rawashdeh NAF (2004) Inhibition of acidic corrosion of pure aluminum by some organic compounds. Corros Sci 46:1129–1140
Zhang QB, Hua YX (2009) Corrosion inhibition of mild steel by alkylimidazolium ionic liquids in hydrochloric acid. Electrochim Acta 54:1881–1887
Wang XM, Yang HY, Wang FH (2010) A cationic gemini-surfactant as effective inhibitor for mild steel in HCl solutions. Corros Sci 52:1268–1276
Nesic S (2007) Key issues related to modelling of internal corrosion of oil and gas pipelines—a review. Corros Sci 49:4308–4338
Wang B, Du M, Zhang J (2009) Study of the inhibition mechanism of imidazoline derivative inhibitor on CO2 corrosion for Q235 steel. Adv Mater Res 79–82:981–984
Abdallah M (2002) Rhodanine azosulpha drugs as corrosion inhibitors for corrosion of 304 stainless steel in HCl solution. Corros Sci 44:717–728
Abdallah M (2003) Ethoxylated fatty alcohols as corrosion inhibitors for dissolution of zinc in hydrochloric acid. Corros Sci 45:2705–2716
Olivares-Xometl O, Likhanova NV, Domínguez-Aguilar MA (2006) Surface analysis of inhibitor films formed by imidazolines and amides on mild steel in an acidic environment. Appl Surf Sci 252:2139–2152
Solmaz R, Kardsa G, Yazici B, Erbil M (2008) Adsorption and corrosion inhibitive properties of 2-amino-5-mercapto-1,3,4-thiadiazole on mild steel in hydrochloric acid media. Colloids Surf A 312:7–17
Bentiss F, Traisnel M, Lagrenée M (2000) The inhibition action of 3,6-bis(2-methoxyphenyl)-1,2-dihydro-1,2,4,5-tetrazine on the corrosion of mild steel in acidic media. Corros Sci 42:127–146
Bouklah M, Hammouti B, Lagrenée M, Bentiss F (2006) Thermodynamic properties of 2,5-bis(4-methoxyphenyl)-1,3,4-oxadiazole as a corrosion inhibitor for mild steel in normal sulfuric acid medium. Corros Sci 48:2831–2842
Flis J, Zakroczymski T (1996) Impedance study of reinforcing steel in simulated pore solution with tannin. J Electrochem Soc 143:2458–2464
Ali SA, Saeed MT, Rahman SV (2003) The isoxazolidines: a new class of corrosion inhibitors of mild steel in acidic medium. Corros Sci 45:253–266
Yurt A, Ulutas S, Dal H (2006) Electrochemical and theoretical investigation on the corrosion of aluminium in acidic solution containing some Schiff bases. Appl Surf Sci 253:919–925
Moretti G, Guidi F, Grion G (2004) Tryptamine as a green iron corrosion inhibitor in 0.5 M deaerated sulphuric acid. Corros Sci 46:387–403
Acknowledgments
The authors gratefully acknowledge the financial support of the National Natural Science Foundation of China (Project No.40806030).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Zhang, J., Zhu, F., Song, W. et al. Corrosion Inhibition Mechanism of Imidazoline-Based Dissymmetric Bis-Quaternary Ammonium Salts with Different Hydrophobic Chain Length on Q235 Steel in 1 M HCl Solution. J Surfact Deterg 16, 559–569 (2013). https://doi.org/10.1007/s11743-012-1390-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11743-012-1390-8