Abstract
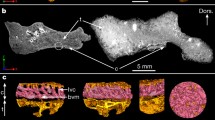
Echinoderms are characterized by a calcite endoskeleton with a unique microstructure, which is optimized for multiple functions. For instance, some light-sensitive ophiuroids (Ophiuroidea) and asteroids (Asteroidea) possess skeletal plates with multi-lens arrays that are thought to act as photosensory organs. The origins of these lens-like microstructures have long been unclear. It was recently proposed that the complex photosensory systems in certain modern ophiuroids and asteroids could be traced back to at least the Late Cretaceous (ca. 79 Ma). Here, we document similar structures in ophiuroids and asteroids from the Early Cretaceous of Poland (ca. 136 Ma) that are approximately 57 million years older than the oldest asterozoans with lens-like microstructures described thus far. We use scanning electron microscopy, synchrotron tomography, and electron backscatter diffraction combined with focused ion beam microscopy to describe the morphology and crystallography of these structures in exceptional detail. The results indicate that, similar to Recent light-sensitive ophiuroids, putative microlenses in Cretaceous ophiuroids and asteroids exhibit a shape and crystal orientation that would have minimized spherical aberration and birefringence. We suggest that these lens-like microstructures evolved by secondary deposition of calcite on pre-existing porous tubercles that were already present in ancestral Jurassic forms.



Similar content being viewed by others
References
Aizenberg, J., & Hendler, G. (2004). Designing efficient microlens arrays: lessons from nature. Journal of Materials Chemistry, 14, 2066–2072.
Aizenberg, J., Tkachenko, A., Weiner, S., Addadi, L., & Hendler, G. (2001). Calcitic microlenses as part of the photoreceptor system in brittlestars. Nature, 412, 819–822.
Aronson, R. B. (1987). Predation on fossil and Recent ophiuroids. Paleobiology, 13, 187–192.
Aronson, R. B. (1989). A community-level test of the Mesozoic marine revolution theory. Paleobiology, 15, 20–25.
Aronson, R. B. (1991). Predation, physical disturbance and sub-lethal arm damage in ophiuroids: A Jurassic-Recent comparison. Marine Ecology Progress Series, 74, 91–97.
Blake, D.B., Tintori, A., & Hagdorn, H. (2000). A new asteroid (Echinodermata) from the Norian (Triassic) Calcare di Zorzino of northern Italy: Its stratigraphic occurrence and phylogenetic significance. Rivista Italiana di Paleontologia e Stratigrafia, 106, 141–156.
Cunningham, J. A., Rahman, I. A., Lautenschlager, S., Rayfield, E. J., & Donoghue, P.C.J. (2014). A virtual world of palaeontology. Trends in Ecology & Evolution, 29, 347–357.
Delroisse, J., Mallefet, J., & Flammang, P. (2016). De novo adult transcriptomes of two European brittle stars: Spotlight on opsin-based photoreception. PLoS ONE, 11, e0152988. doi:10.1371/journal.pone.0152988.
Döderlein, L. (1898). Ueber “Krystallkörper” bei Seesternen. Denkschriften der Medizinisch Naturwissenschaftlichen Gesellschaft zu Jena, 8, 491–494.
Dubois, P., & Hayt, S. (1990). Ultrastructure des ossicules d’échinodermes à stéréome non perforé. In C. De Ridder, P. Dubois; M.C. Lahaye & M. Jangoux (Eds.), Echinoderm research (pp. 217–223). Rotterdam: Balkema.
Gale, A. (2011). Asteroidea (Echinodermata) from the Oxfordian (Late Jurassic) of Savigna, Départment [sic!] du Jura, France. Swiss Journal of Palaeontology, 130, 69–89.
Garm, A., & Nilsson, D. E. (2014). Visual navigation in starfish: first evidence for the use of vision and eyes in starfish. Proceedings of the Royal Society B: Biological Sciences, 281, 2013–3011.
Głuchowski, E. (1987). Jurassic and Early Cretaceous articulate Crinoidea from the Pieniny Klippen Belt and Tatra Mts. Poland. Studia Geologica Polonica, 94, 1–102.
Gorzelak, P., & Salamon, M. A. (2009). Signs of benthic predation on Late Jurassic stalked crinoids, preliminary data. Palaios, 24, 70–73.
Gorzelak, P., Salamon. M. A., & Baumiller, T. K. (2012). Predator-induced macroevolutionary trends in Mesozoic crinoids. Proceedings of the National Academy of Sciences of the United States of America, 109, 7004–7007.
Gorzelak, P., Salamon, M. A., Lach, R., Loba, M., & Ferré, B. (2014). Microlens arrays in the complex visual system of Cretaceous echinoderms. Nature Communications, 5, 3576. doi:10.1038/ncomms4576.
Heatfield, B. M. (1971). Growth of the calcareous skeleton during regeneration of spines of the sea urchin Strongylocentrotus purpuratus (Stimpson); a light and scanning electron microscope study. Journal of Morphology, 134, 57–90.
Hendler, G. (1984). Brittlestar color-change and phototaxis (Echinodermata: Ophiuroidea: Ophiocomidae). Marine Ecology, 5, 379–401.
Hendler, G. (2004). An echinoderm’s eye view of photoreception and vision. In T. Heinzeller, & J. Nebelsick (Eds.), Echinoderms; Munchen; Proceedings of the 11th International Echinoderm Conference. (pp. 339–350). Leiden: A.A. Balkema Publishers.
Hendler, G., & Byrne, M. (1987). Fine structure of the dorsal arm plate of Ophiocoma wendti (Echinodermata, Ophiuroidea). Zoomorphology, 107, 261–272.
Hess, H., Salamon, M. A., & Gorzelak, P. (2011). Late Jurassic-Early Cretaceous (Tithonian-Berriasian) cyrtocrinids from south-eastern Poland. Neues Jahrbuch für Geologie und Paläontologie, Abhandlungen, 260, 119–128.
Kaim, A. (2001). Faunal dynamics of juvenile gastropods and associated organisms across the Valanginian transgression-regression cycle in central Poland. Cretaceous Research, 22, 333–351.
Killian, C. E., & Wilt, F. H. (2008). Molecular aspects of biomineralization of the echinoderm endoskeleton. Chemical Reviews, 108, 4463–4474.
Mah, C. L. (2005). A phylogeny of Iconaster and Glyphodiscu s (Goniasteridae; Valvatida; Asteroidea) with descriptions of four new species. Zoosystema, 27, 131–167.
Politi, Y., Arad, T., Klein, E., Weiner, S., & Addadi, L. (2004). Sea urchin spine calcite forms via a transient amorphous calcium phase. Science, 306, 1161–1164.
Raup, D. M. (1966). The endoskeleton. In R. A. Boolootian (Ed.), Physiology of echinodermata (pp. 379–395). New York: Interscience.
Salamon, M. A. (2007). First record of bourgueticrinid crinoids from the Cenomanian of southern Poland. Cretaceous Research, 28, 459–499.
Salamon, M. A. (2008). The Callovian (Middle Jurassic) crinoids from northern Lithuania. Paläontologische Zeitschrift, 82, 269–278.
Salamon, M. A. (2009). Early Cretaceous (Valanginian) sea lilies (Echinodermata, Crinoidea) from Poland. Swiss Journal of Geosciences, 102, 77–88.
Salamon, M. A., & Gorzelak, P. (2010a). Cyrtocrinids (Echinodermata, Crinoidea) from Upper Jurassic Štramberk-type limestones in southern Poland. Palaeontology, 53, 869–885.
Salamon, M. A., & Gorzelak, P. (2010b). Late Cretaceous crinoids (Crinoidea) from Eastern Poland. Palaeontographica Abt. A, 291, 1–43.
Salamon, M. A., Niedźwiedzki, R., Lach, R., Brachaniec, T., & Gorzelak, P. (2012). Ophiuroids discovered in the Middle Triassic hypersaline environment. PLoS ONE, 7(11), e49798. doi:10.1371/journal.pone.0049798.
Salamon, M. A., & Zatoń, M. (2007). Late Bajocian through Callovian (Middle Jurassic) crinoid fauna from the epicontinental deposits of Poland. Swiss Journal of Geosciences, 100, 153–164.
Smith, A. B. (1990). Biomineralization in echinoderms. In J. G. Carter (Ed.), Skeletal biomineralization: Patterns, processes, and evolutionary trends (pp. 413–443). New York: Van Nostrand Reinhold.
Stampanoni, M., et al. (2007). TOMCAT: A beamline for tomographic microscopy and coherent radiology experiments. AIP Conference Proceedings, 879, 848.
Sutton, M. D., Rahman, I. A., & Garwood, R. J. (2014). Techniques for virtual palaeontology. New York: Wiley.
Sweatman, H.P.A. (1995). A field study of fish predation on juvenile crown-of-thorns starfish. Coral Reefs, 14(1), 47–53.
Torney, C., Lee, M. R., & Owen, A. W. (2014). Microstructure and growth of the lenses of schizochroal trilobite eyes. Palaeontology, 57, 783–799.
Ullrich-Lüter, E.M., Dupont, S., Arboleda, E., Hausen, H., & Arnone, M.I. (2011). Unique system of photoreceptors in sea urchin tube feet. Proceedings of the National Academy of Sciences of the United States of America, 108, 8367–8372.
Vermeij, G. J. (1977). The Mesozoic marine revolution; evidence from snails, predators and grazers. Paleobiology, 3, 245–258.
Villier, L. (2008). Sea star ossicles from the Callovian black clays of the Łuków area, eastern Poland. Neues Jahrbuch für Geologie und Paläontologie, Abh, 247, 147–160.
Villier, L., Charbonnier, S., & Riou, B. (2009). Sea stars from Middle Jurassic Lagerstätte of La Volute sur Rhône (Ardèche, France). Journal of Paleontology, 83, 389–398.
Villier, L., Kutscher, M., & Mah, C.H.L. (2004). Systematics and palaecology of middle Toarcian Asteroidea (Echinodermata) from the ‘Seuil du Poitou’, Western France. Geobios, 37, 807–825.
Vinogradova, E., Ruíz-Zepeda, F., Plascencia-Villa, G., & José-Yacamán, M. (2016). Calcitic microlens arrays in Archaster typicus: Microstructural evidence for an advanced photoreception system in modern starfish. Zoomorphology, 135, 83–87.
Walker, M. H. (1978). Food and feeding habits of Lethrinus chrysostomus Richardson (Pisces: Perciformes) and other Lethrinids on the Great Barrier Reef. Australian Journal of Marine & Freshwater Research, 29(5), 623–630.
Wei, T., Sun, Y., Zhang, B., Wang, R., & Xu, T. (2014). A mitogenomic perspective on the phylogenetic position of the Hapalogenys genus (Acanthopterygii: Perciformes) and the evolutionary origin of perciformes. PLoS ONE, 9(7), e103011. doi:10.1371/journal.pone.0103011.
Yamamoto, M., & Yoshida, M. (1978). Fine structure of the ocelli of a synaptid holothurian,Opheodesoma spectabilis, and the effects of light and darkness. Zoomorphologie, 90, 1–17.
Yoshida, M. (1966). Photosensitivity. In R. A. Boolootian (Ed.), Physiology of echinodermata (pp. 435–464). New York: Wiley.
Zatoń, M., Salamon, M. A., Boczarowski, A., & Sitek, S. (2008a). Taphonomy of dense ophiuroid accumulations from the Middle Triassic of Poland. Lethaia, 41(1), 47–58.
Zatoń, M., Salamon, M. A., & Kaźmierczak, J. (2008b). Cyrtocrinids (Crinoidea) and associated stalked crinoids from the Lower/Middle Oxfordian (Upper Jurassic) shelfal deposits of southern Poland. Geobios, 41, 559–569.
Zhang, S. (2003). Fabrication of novel biomaterials through molecular self-assembly. Nature Biotechnology, 21, 1171–1178.
Zhao, F., Bottjer, D. J., Hu, S., Yin, Z., & Zhu, M. (2013). Complexity and diversity of eyes in Early Cambrian ecosystems. Scientific Reports, 3, 2751. doi:10.1038/srep02751.
Acknowledgements
This work was completed while the first author was a recipient of a grant from the Polish National Science Centre (NCN) Grant number DEC-2011/03/N/ST10/04798 and was performed in part in the NanoFun laboratory co-financed by the European Regional Development Fund within the Innovation Economy Operational Programme POIG.02.02.00-00-025/09. IAR was funded by an 1851 Royal Commission Research Fellowship. SZ was funded by grants RYC-2012-10576 and CGL2013-48877 from the Spanish MINECO. We acknowledge the Paul Scherrer Institut, Villigen, Switzerland for the provision of synchrotron radiation beamtime on the TOMCAT beamline at the Swiss Light Source and thank Professor Charles G Messing (Nova Southeastern University) for providing the extant ophiuroid specimen. We also thank two anonymous reviewers for their supportive comments.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standards
Investigations comply with the current laws of the country in which they were performed.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gorzelak, P., Rahman, I.A., Zamora, S. et al. Towards a Better Understanding of the Origins of Microlens Arrays in Mesozoic Ophiuroids and Asteroids. Evol Biol 44, 339–346 (2017). https://doi.org/10.1007/s11692-017-9411-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11692-017-9411-1