ABSTRACT
BACKGROUND
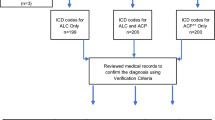
Alcohol withdrawal syndrome (AWS) occurs when alcohol-dependent individuals abruptly reduce or stop drinking. Hospitalized alcohol-dependent patients are at risk. Hospitals need a validated screening tool to assess withdrawal risk, but no validated tools are currently available.
OBJECTIVE
To examine the admission Alcohol Use Disorders Identification Test-(Piccinelli) Consumption (AUDIT-PC) ability to predict the subsequent development of AWS among hospitalized medical-surgical patients admitted to a non-intensive care setting.
DESIGN
Retrospective case–control study of patients discharged from the hospital with a diagnosis of AWS. All patients with AWS were classified as presenting with AWS or developing AWS later during admission. Patients admitted to an intensive care setting and those missing AUDIT-PC scores were excluded from analysis. A hierarchical (by hospital unit) logistic regression was performed and receiver-operating characteristics were examined on those developing AWS after admission and randomly selected controls. Because those diagnosing AWS were not blinded to the AUDIT-PC scores, a sensitivity analysis was performed.
PARTICIPANTS
The study cohort included all patients age ≥18 years admitted to any medical or surgical units in a single health care system from 6 October 2009 to 7 October 2010.
KEY RESULTS
After exclusions, 414 patients were identified with AWS. The 223 (53.9 %) who developed AWS after admission were compared to 466 randomly selected controls without AWS. An AUDIT-PC score ≥4 at admission provides 91.0 % sensitivity and 89.7 % specificity (AUC = 0.95; 95 % CI, 0.94–0.97) for AWS, and maximizes the correct classification while resulting in 17 false positives for every true positive identified. Performance remained excellent on sensitivity analysis (AUC = 0.92; 95 % CI, 0.90–0.93). Increasing AUDIT-PC scores were associated with an increased risk of AWS (OR = 1.68, 95 % CI 1.55–1.82, p < 0.001).
CONCLUSIONS
The admission AUDIT-PC score is an excellent discriminator of AWS and could be an important component of future clinical prediction rules. Calibration and further validation on a large prospective cohort is indicated.


Similar content being viewed by others
REFERENCES
Saitz R, Freedner N, Palfai TP, Horton NJ, Samet JH. The severity of unhealthy alcohol use in hospitalized medical patients. The spectrum is narrow. J Gen Intern Med. 2006;21(4):381–385.
Sharkey J, Brennan D, Curran P. The pattern of alcohol consumption of a general hospital population in North Belfast. Alcohol Alcohol. 1996;31:279–285.
Taylor CL, Passmore N, Kilbane P. Prospective study of alcohol-related admissions to an inner-city hospital. Lancet. 1986;2:265–268.
Dolman JM, Hawkes ND. Combining The Audit Questionnaire And Biochemical Markers To Assess Alcohol Use And Risk Of Alcohol Withdrawal In Medical Inpatients. Alcohol Alcohol. 2005;40(6):515–519.
Gupta M. Alcohol Withdrawal and Prolonged Hospital Stay in a Patient with Neuroimaging Abnormalities: A Case Report. Alcohol Alcohol. 2009;44(2):183–184.
Cross GM, Hennessey PT. Principles and practice of detoxification. Prim Care. 1993;20(1):81–93.
DeBellis R, Smith BS, Choi S, Malloy M. Management of Delirium Tremens. J Intensive Care Med. 2005;20(3):164–173.
Kaner EF, Beyer F, Dickinson HO, et al. Effectiveness of brief alcohol interventions in primary care populations. Cochrane Database Syst Rev. 2007;2, CD004148.
Saitz R. Candidate performance measures for screening for, assessing, and treating unhealthy substance use in hospitals: advocacy or evidence-based practice? Ann Intern Med. 2010;153(1):40–43.
Babor TF, McRee BG, Kassebaum PA, Grimaldi PL, Ahmed K, Bray J. Screening, Brief Intervention, and Referral to Treatment (SBIRT): Toward a public health approach to the management of substance abuse. Subst Abus. 2007;28(3):7–30.
Sullivan JT, Sykora K, Schneiderman J, Naranjo CA, Sellers EM. Assessment of alcohol withdrawal: the revised clinical institute withdrawal assessment for alcohol scale (CIWA-Ar). Br J Addict. 1989;84(11):1353–1357.
Saunders JB, Aasland OG, Amundsen A, Grant M. Alcohol consumption and related problems among primary health care patients: WHO Collaborative Project on Early Detection of Persons with Harmful Alcohol Consumption–I. Addiction. 1993;88(3):349–362.
Reinert DF, Allen JP. The Alcohol Use Disorders Identification Test: An Update of Research Findings. Alcohol Clin Exp Res. 2007;31(2):185–199.
Roux JP, Malte CA, Kivlahan DR. The alcohol use disorders identification test (AUDIT) predicts alcohol withdrawal symptoms durin ginpatient detoxification. J Addict Dis. 2002;4:81–91.
Bush K, Kivlahan DR, McDonell MB, Fihn SD, Bradley KA. The AUDIT alcohol consumption questions (AUDIT-C): an effective brief screening test for problem drinking. Ambulatory Care Quality Improvement Project (ACQUIP). Arch Intern Med. 1998;158(16):1789–1795.
Aertgeerts B, Buntinx F, Ansoms S, Fevery J. Questionnaires are better than laboratory tests to screen for current alcohol abuse or dependence in a male inpatient population. Acta Clin Belg. 2002;57:241–249.
Gomez A, Conde A, Santana JM, Jorrin A. Diagnostic usefulness of brief versions of Alcohol Use Disorders Identification Test (AUDIT) for detecting hazardous drinkers in primary care settings. J Stud Alcohol. 2005;66:305–308.
Piccinelli M, Tessari E, Bortolomasi M, et al. Efficacy of the alcohol use disorders identification test as a screening tool for hazardous alcohol intake and related disorders in primary care: A validity study. Br Med J. 1997;314(7078):420–424.
Reoux JP, Malte CA, Kivlahan DR, Saxon AJ. The alcohol use disorders identification test (AUDIT) predicts alcohol withdrawal symptoms durin ginpatient detoxification. J Addict Dis. 2002;4:81–91.
Landis JR, Koch GG. The measurement of observer agreement for categorical data. Biometrics. 1977;33(1):159–174.
Metz CE. Basic principles of ROC analysis. Semin Nucl Med. 1978;8(4):283–298.
Obuchowski NA. Receiver operating characteristic curves and their use in radiology. Radiology. 2003;229(1):3–8.
Wan S, Zhang B. Smooth semiparametric receiver operating characteristic curves for continuous diagnostic tests. Stat Med. 2007;26(12):2565–2586.
Alemayehu D, Zou KH. Applications of ROC Analysis in Medical Research: Recent Developments and Future Directions. Acad Radiol. 2012;19(12):1457–1464.
Metz CE. Receiver operating characteristic analysis: a tool for the quantitative evaluation of observer performance and imaging systems. J Am Coll Radiol: JACR. 2006;3(6):413–422.
Schisterman EF, Perkins NJ, Liu A, Bondell H. Optimal cut-point and its corresponding Youden Index to discriminate individuals using pooled blood samples. Epidemiology. 2005;16(1):73–81.
Youden WJ. Index for rating diagnostic tests. Cancer. 1950;3(1):32–35.
Swets JA. The science of choosing the right decision threshold in high-stakes diagnostics. Am Psychol. 1992;47(4):522–532.
Swets JA, Dawes RM, Monahan J. Better decisions through science. Sci Am. 2000;283(4):82–87.
Acknowledgements
Funding Sources
This work was not funded by any external sources. Drs. Pecoraro and Woody receive salary support through the National Institute on Drug Abuse (NIDA): Clinical Trials Network (CTN) U10 DA-13043 (Pecoraro and Woody) and KO5 DA-17009 (Woody).
Prior Presentation
Results were presented as a poster at the 35th annual SGIM meeting in Orlando, FL on 9 May 2012.
Conflicts of Interest
Drs. Ewen and Kolm have received research grant funding unrelated to this manuscript from: Bristol-Myers Squibb (Ewen and Kolm), Sanofi-Aventis (Ewen and Kolm), and Astra Zeneca (Kolm). Dr. Woody received a consulting payment from Alkermes for work on clinical guidelines regarding how to start opioid-addicted individuals on extended release-injectable naltrexone (XR-NTX), and Alkermes has provided XR-NTX for a study of amphetamine addiction treatment in Iceland on which Dr. Woody is a Co-Investigator. Dr. Woody is on the scientific advisory board of RADARS, a non-profit organization administered by Denver Health to study abuse liability of prescription drugs that is funded by contracts with pharmaceutical companies. Drs. Pecoraro, Horton, and Mooney, and Ms. McGraw declare they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 256 kb)
Rights and permissions
About this article
Cite this article
Pecoraro, A., Ewen, E., Horton, T. et al. Using the AUDIT-PC to Predict Alcohol Withdrawal in Hospitalized Patients. J GEN INTERN MED 29, 34–40 (2014). https://doi.org/10.1007/s11606-013-2551-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11606-013-2551-9