Abstract
Background
The aim of this study was to investigate the efficacy and safety of an alternating regimen of S-1 plus low-dose cisplatin and S-1 alone as adjuvant therapy in patients with advanced gastric cancer.
Patients and Methods
The study group comprised 100 patients with stage IIIA, stage IIIB, or stage IV. Patients postoperatively received three 5-week cycles of chemotherapy. In the first cycle, S-1 (80 mg/m2) was given daily for 3 weeks, followed by 2 weeks of rest, and low-dose cisplatin (10 mg) was given on days 1 to 5 and 8 to 12. In the second and third 5-week cycles, S-1 alone was given. The primary endpoints were median survival time, and survival at 1 and 3 years. Secondary endpoints were safety and overall response rates.
Results
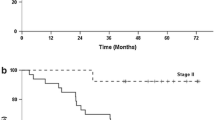
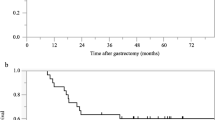
Median survival time was 18 months in stage IV and 32 months in stage IIIB. The rates of survival at 1 and 3 years were 68.7% and 30.6% in stage IV, 100% and 68.4% in stage IIIA, and 100% and 46.6% in stage IIIB, respectively. Adverse events of grade 3 or 4 occurred in 14% of the patients. The overall response rate of target lesions was 54%.
Conclusion
Our regimen is effective and safe for adjuvant therapy in patients with curatively resected stage III gastric cancer.


Similar content being viewed by others
References
Macdonald JS, Schein PS, Woolley PV, et al. 5-Fluorouracil, doxorubicin and mitomycin (FAM) combination chemotherapy for advanced gastric cancer. Ann Intern Med 1980; 93: 533–36.
Vanhoefer U, Rougier P, Wilke H, et al. Final results of a randomized phase III trial of sequential high-dose methotrexate, fluorouracil, and doxorubicin versus etoposide, leucovorin, and fluorouracil versus infusional fluorouracil and cisplatin in advanced gastric cancer: A trial of the European Organization for Research and Treatment of Cancer Gastrointestinal Tract Cancer Cooperative Group. J Clin Oncol 2000; 18: 2648–57.
Ohtsu A, Shimada Y, Shirako K, et al. Randomized phase III trial of fluorouracil alone versus fluorouracil plus cisplatin versus uracil and tegafur plus mitomycin in patients with unresectable, advanced gastric cancer: The Japan Clinical Oncology Group Study (JCOG9205). J Clin Oncol 2003; 21: 54–59.
Preusser P, Wilke H, Achterrath W, et al. Phase II study with the combination etoposide, doxorubicin, and cisplatin in advanced measurable gastric cancer. J Clin Oncol 1989; 7: 1310–17.
Wilke H, Preusser P, Fink U, et al. High dose folinic acid/etoposide/5-fluorouracil in advanced gastric cancer-a phase II study in elderly patients or patients with cardiac risk. Invest New Drugs 1990; 8: 65–70.
Findlay M, Cunningham D, Norman A, et al. A phase II study in advanced gastro-esophageal cancer using epirubicin and cisplatin in combination with continuous infusion 5-fluorouracil (ECF). Ann Oncol 1994; 5: 609–16.
Cutsem EV, Moiseyenko VM, Tjulandin S, et al. Phase III study of docetaxel and cisplatin plus fluorouracil compared with cisplatin and fluorouracil as first-line therapy for advanced gastric cancer: A report of the V325 Study Group. J Clin Oncol 2006; 24: 4991–97.
Kang YK, Kang WK, Shin DB, et al. Capecitabine/cisplatin versus 5-fluorouracil/cisplatin as first-line therapy in patients with advanced gastric cancer: A randomised phase III noninferiority trial. Ann Oncol. 2009 Apr;20(4):666–73.
Cunningham D, Starling N, Rao S, et al. Capecitabine and oxaliplatin for advanced esophagogastric cancer. N Engl J Med 2008; 358: 36–46.
Koizumi W, Narahara H, Hara T, et al. S-1 plus cisplatin versus S-1 alone for first-line treatment of advanced gastric cancer (SPIRITS trial): A phase III trial. Lancet Oncol 2008; 9: 215–21.
Macdonald JS, Smalley SR, Benedetti J, et al. Chemoradiotherapy after surgery compared with surgery alone for adenocarcinoma of the stomach or gastroesophageal junction. N Engl J Med 2001; 345: 725–30.
Cummingham D, Allum WH, Stenning SP, et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N Engl J Med 2006; 355: 11–20.
Sakuramoto S, Sasako M, Yamaguchi T, et al. Adjuvant chemotherapy for gastric cancer with S-1, an oral fluoropyrimidine. N Engl J Med 2007; 357: 1810–20.
Shirasaka T, Shimamoto Y, Ohshimo H, et al. Development of a novel form of an oral 5-fluorouracil derivative (S-1) directed to the potentiation of the tumor selective cytotoxicity of 5-fluorouracil by two biochemical modulators. Anticancer Drugs 1996; 7: 548–57.
Sugimachi K, Maehara Y, Horikoshi N, et al. An early phase II study of oral S-1, a newly developed 5-fluorouracil derivative for advanced and recurrent gastrointestinal cancers. Oncology 1999; 57: 202–10.
Sakata Y, Ohtsu A, Horikoshi N, et al. Late phase II study of novel oral fluoropyrimidine anticancer drug S-1 (1 M tegafur-0.4 M gimestat-1 M otastat potassium) in advanced gastric cancer patients. Eur J Cancer 1998; 34: 1715–20.
Koizumi W, Kurihara M, Nakao S, et al. Phase II study of S-1, a novel oral derivative of 5-fluorouracil, in advanced gastric cancer. Oncology 2000; 58: 191–97.
Koizumi W, Tanabe S, Saigenji K, et al. Phase I/II study of S-1 combined with cisplatin in patients with advanced gastric cancer. Br J Cancer 2003; 89: 2207–12.
Ajani JA, Lee FC, Singh DA, et al. MultiCentre phase II trial of S-1 plus cisplatin in patients with untreated advanced gastric or gastroesophageal junction adenocarcinoma. J Clin Oncol 2006; 24: 663–67.
Japanese Gastric Cancer Association. Japanese classification of gastric carcinoma—2nd English edition. Gastric cancer 1998; 1: 10–24.
Boku N, Yamamoto S, Fukuda H, et al. Fluorouracil versus combination of irinotecan plus cisplatin versus S-1 in metastatic gastric cancer: A randomised phase 3 study. Lancet Oncol. 2009 Nov;10(11):1063–9.
Nakajima T, Nashimoto A, Kitamura M, et al. Adjuvant mitomycin and fluorouracil followed by oral uracil plus tegafur in serosa-negative gastric cancer: A randomized trial. Lancet 1999; 354: 273–7.
Oba K. Efficacy of adjuvant chemotherapy using tegafur-based regimen for curatively resected gastric cancer: Update of a meta-analysis. Int J Clin Oncol. 2009 Apr;14(2):85–9.
Hyodo I, Nishina T, Moriwaki T, et al. A phase I study of S-1 combined with weekly cisplatin for metastatic gastric cancer in an outpatient setting. Eur J Cancer 2003; 39: 2328–33.
Iwase H, Shimada M, Tsuzuki T, et al. A phase II multicentric trial of S-1 combined with 24 h-infusion of cisplatin in patients with advanced gastric cancer. Anticancer Res 2005; 25: 1297–1302.
Tsuji A, Shima Y, Morita S, et al. Combination chemotherapy of S-1 and low-dose twice-weekly cisplatin for advanced and recurrent gastric cancer in an outpatient setting: A retrospective study. Anticancer Res 2008; 28: 1433–38.
Chang HY, Sung HN, Yong IK, et al. Comparison of prognostic significance of nodal staging between old (4th edition) and new (5th edition) UICC TNM classification for gastric carcinoma. World J Surg 1999; 23: 492–98.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mita, K., Ito, H., Fukumoto, M. et al. Alternating Treatment with S-1 Plus Low-Dose Cisplatin and S-1 Alone for Advanced Gastric Cancer. J Gastrointest Surg 15, 791–796 (2011). https://doi.org/10.1007/s11605-011-1476-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-011-1476-z