Summary
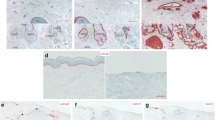
Skin wound healing is a complex event, and interrupted wound healing process could lead to scar formation. The aim of this study was to examine the morphological changes of scar tissue. Pathological staining (HE staining, Masson’s trichrome staining, methenamine silver staining) was used to evaluate the morphological changes of regenerating epidermis in normal skin and scar tissue, and immunofluorescence staining to detect the expression of collagen IV, a component of basement membrane (BM), and the expression of integrinβ4, a receptor for BM laminins. Additionally, the expression of CK14, CK5, and CK10 was measured to evaluate the proliferation and differentiation of keratinocytes in normal skin and scar tissue. The results showed that the structure of the skin was histologically changed in scar tissue. Collagen IV, expressed under the epidermis of normal skin, was reduced distinctly in scar tissue. Integrinβ4, expressed in the basal layer of normal skin, was found absent in the basal layer of scar tissue. Additionally, it was found that keratinocytes in scarring epidermis were more proliferative than in normal skin. These results indicate that during the skin wound healing, altered formation of BM may affect the proliferation of keratinocytes, reepithelial and tissue remodeling, and then result in scar formation. Thus, remodeling BM structure during wound repair may be beneficial for improving healing in cutaneous wounds during clinical practice.
Similar content being viewed by others
References
Hu MS, Rennert RC, McArdle A, et al. The role of stem cells during scarless skin wound healing. Adv Wound Care, 2014,3(4):304–314
Gornowicz-Porowska J, Bowszyc-Dmochowska M, Dmochowski M. Autoimmunity-driven enzymatic remodeling of the dermal-epidermal junction in bullous pemphigoid and dermatitis herpetiformis. Autoimmunity, 2012,45(1):71–80
Nakamura T, Yoshitomi Y, Sakai K, et al. Epiprofin orchestrates epidermal keratinocyte proliferation and differentiation. J Cell Sci, 2014,127(24):5261–5272
Ko MS, Marinkovich MP. Role of dermal-epidermal basement membrane zone in skin, cancer, and developmental disorders. Dermatol Clin, 2010,28(1):1–16
Breitkreutz D, Mirancea N, Nischt R. Basement membranes in skin: unique matrix structures with diverse functions? Histochem Cell Biol, 2009,132(1): 1–10
Ghahary A, Ghaffari A. Role of keratinocyte-fibroblast cross-talk in development of hypertrophic scar. Wound Repair Regen, 2007,15(Suppl 1):S46–S53
Werner S, Krieg T, Smola H. Keratinocyte-fibroblast interactions in wound healing. J Invest Dermatol, 2007,127(5):998–1008
Reinke JM, Sorg H. Wound repair and regeneration. Eur Surg Res, 2012,49(1):35–43
Yu Y, Tan M, Chen H, et al. Non-thermal plasma suppresses bacterial colonization on skin wound and promotes wound healing in mice. J Huazhong Univ Sci Technolog Med Sci, 2011,31(3):390–394
Yang S, Ma K, Geng Z, et al. Oriented cell division: new roles in guiding skin wound repair and regeneration. Biosci Rep, 2015,35(6)
Corr DT, Hart DA. Biomechanics of scar tissue and uninjured skin. Adv Wound Care, 2013,2(2):37–43
Jones N. Scar tissue. Curr Opin Otolaryngol Head Neck Surg, 2010,18(4):261–265
Zhou W, Liu GH, Yang SH, et al. Low-intensity treadmill exercise promotes rat dorsal wound healing. J Huazhong Univ Sci Technolog Med Sci, 2016,36(1): 121–126
Balana ME, Charreau HE, Leiros GJ. Epidermal stem cells and skin tissue engineering in hair follicle regeneration. World J Stem Cells, 2015,7(4):711–727
Broughton G, 2nd, Janis JE, Attinger CE. The basic science of wound healing. Plast Reconstr Surg, 2006,117(7 Suppl):12S–34S
Bellemare J, Roberge CJ, Bergeron D, et al. Epidermis promotes dermal fibrosis: role in the pathogenesis of hypertrophic scars. J Pathol, 2005,206(1):1–8
Iozzo RV. Basement membrane proteoglycans: from cellar to ceiling. Nat Rev Mol Cell Biol, 2005,6(8):646–656
Wang Y, Zhang L, Lei R, et al. Effects of blocking two sites of transforming growth factor-beta/Smads signaling on the formation of scar-related proteins in human skin fibroblasts. Zhonghua Shao Shang Za Zhi (Chinese), 2015,31(5):372–377
Sa Y, Li C, Li H, et al. TIMP-1 Induces alpha-smooth muscle actin in fibroblasts to promote urethral scar formation. Cell Physiol Biochem, 2015,35(6):2233–2243
Khanom R, Sakamoto K, Pal SK, et al. Expression of basal cell keratin 15 and keratin 19 in oral squamous neoplasms represents diverse pathophysiologies. Histol Histopathol, 2012,27(7):949–959
Su L, Morgan PR, Lane EB. Keratin 14 and 19 expression in normal, dysplastic and malignant oral epithelia. A study using in situ hybridization and immunohistochemistry. J Oral Pathol Med, 1996, 25(6):293–301
Author information
Authors and Affiliations
Corresponding authors
Additional information
This work was supported in part by the National Nature Science Foundation of China (No. 81372067) and the National Basic Science and Development Program of China (973 Program, No. 2012CB518105).
Rights and permissions
About this article
Cite this article
Yang, Sw., Geng, Zj., Ma, K. et al. Comparison of the histological morphology between normal skin and scar tissue. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 36, 265–269 (2016). https://doi.org/10.1007/s11596-016-1578-7
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-016-1578-7