Abstract
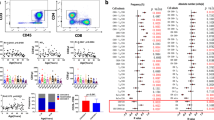
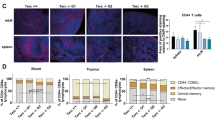
Immunosenescence results from a continuous deterioration of immune responses resulting in a decreased response to vaccines. A well-described age-related alteration of the immune system is the decrease of de novo generation of T and B cells. In addition, the accumulation of memory cells and loss of diversity in antigen specificities resulting from a lifetime of exposure to pathogens has also been described. However, the effect of aging on subsets of γδTCR+ T cells and Tregs has been poorly described, and the efficacy of the recall response to common persistent infections in the elderly remains obscure. Here, we investigated alterations in the subpopulations of the B and T cells among 24 healthy young (aged 19–30) and 26 healthy elderly (aged 53–67) individuals. The analysis was performed by flow cytometry using freshly collected peripheral blood. γδTCR+ T cells were overall decreased, while CD4+CD8− cells among γδTCR+ T cells were increased in the elderly. Helios+Foxp3+ and Helios−Foxp3+ Treg cells were unaffected with age. Recent thymic emigrants, based on CD31 expression, were decreased among the Helios+Foxp3+, but not the Helios−Foxp3+ cell populations. We observed a decrease in Adenovirus-specific CD4+ and CD8+ T cells and an increase in CMV-specific CD4+ T cells in the elderly. Similarly, INFγ+TNFα+ double-positive cells were decreased among activated T cells after Adenovirus stimulation but increased after CMV stimulation. The data presented here indicate that γδTCR+ T cells might stabilize B cells, and functional senescence might dominate at higher ages than those studied here.



Similar content being viewed by others
Abbreviations
- TCR:
-
T cell receptor
- Tregs:
-
regulatory T cells
- CM:
-
central memory
- EM:
-
effector memory
- TEMRA:
-
terminally differentiated effector memory
- RTE:
-
recent thymic emigrants
References
Agius E, Lacy KE, Vukmanovic-Stejic M et al (2009) Decreased TNF-alpha synthesis by macrophages restricts cutaneous immunosurveillance by memory CD4+ T cells during aging. J Exp Med 206:1929–1940. doi:10.1084/jem.20090896
Alexander T, Sattler A, Templin L et al (2013) Foxp3+ Helios+ regulatory T cells are expanded in active systemic lupus erythematosus. Ann Rheum Dis 72:1549–1558. doi:10.1136/annrheumdis-2012-202216
Andreu-Ballester JC, García-Ballesteros C, Benet-Campos C et al (2012) Values for αβ and γδ T-lymphocytes and CD4+, CD8+, and CD56+ subsets in healthy adult subjects: assessment by age and gender. Cytometry B Clin Cytom 82:238–244. doi:10.1002/cyto.b.21020
Appay V, Sauce D (2014) Naive T cells: The crux of cellular immune aging? Exp Gerontol 54C:90–93. doi:10.1016/j.exger.2014.01.003
Argentati K, Re F, Donnini A et al (2002) Numerical and functional alterations of circulating gammadelta T lymphocytes in aged people and centenarians. J Leukoc Biol 72:65–71
Bacher P, Kniemeyer O, Schönbrunn A et al (2013) Antigen-specific expansion of human regulatory T cells as a major tolerance mechanism against mucosal fungi. Mucosal Immunol. doi:10.1038/mi.2013.107
Betts MR, Nason MC, West SM et al (2006) HIV nonprogressors preferentially maintain highly functional HIV-specific CD8+ T cells. Blood 107:4781–4789. doi:10.1182/blood-2005-12-4818
Beveridge NER, Price DA, Casazza JP et al (2007) Immunisation with BCG and recombinant MVA85A induces long-lasting, polyfunctional Mycobacterium tuberculosis-specific CD4+ memory T lymphocyte populations. Eur J Immunol 37:3089–3100. doi:10.1002/eji.200737504
Booth NJ, McQuaid AJ, Sobande T et al (2010) Different proliferative potential and migratory characteristics of human CD4+ regulatory T cells that express either CD45RA or CD45RO. J Immunol 184:4317–4326. doi:10.4049/jimmunol.0903781
Britanova OV, Putintseva EV, Shugay M et al (2014) Age-related decrease in TCR repertoire diversity measured with deep and normalized sequence profiling. J Immunol 192:2689–2698. doi:10.4049/jimmunol.1302064
Bulati M, Buffa S, Candore G et al (2011) B cells and immunosenescence: a focus on IgG+IgD-CD27- (DN) B cells in aged humans. Ageing Res Rev 10:274–284. doi:10.1016/j.arr.2010.12.002
Caccamo N, Dieli F, Wesch D et al (2006) Sex-specific phenotypical and functional differences in peripheral human Vgamma9/Vdelta2 T cells. J Leukoc Biol 79:663–666. doi:10.1189/jlb.1105640
Caraux A, Klein B, Paiva B et al (2010) Circulating human B and plasma cells. Age-associated changes in counts and detailed characterization of circulating normal CD138- and CD138+ plasma cells. Haematologica 95:1016–1020. doi:10.3324/haematol.2009.018689
Chong Y, Ikematsu H, Yamaji K et al (2005) CD27(+) (memory) B cell decrease and apoptosis-resistant CD27(−) (naive) B cell increase in aged humans: implications for age-related peripheral B cell developmental disturbances. Int Immunol 17:383–390. doi:10.1093/intimm/dxh218
Colonna-Romano G, Potestio M, Aquino A et al (2002) Gamma/delta T lymphocytes are affected in the elderly. Exp Gerontol 37:205–211. doi:10.1016/S0531-5565(01)00185-1
Colonna-Romano G, Bulati M, Aquino A et al (2003) B cells in the aged: CD27, CD5, and CD40 expression. Mech Ageing Dev 124:389–393
Colonna-Romano G, Akbar AN, Aquino A et al (2007) Impact of CMV and EBV seropositivity on CD8 T lymphocytes in an old population from West-Sicily. Exp Gerontol 42:995–1002. doi:10.1016/j.exger.2007.05.006
Colonna-Romano G, Bulati M, Aquino A et al (2009) A double-negative (IgD-CD27-) B cell population is increased in the peripheral blood of elderly people. Mech Ageing Dev 130:681–690. doi:10.1016/j.mad.2009.08.003
Coomes SM, Pelly VS, Wilson MS (2013) Plasticity within the ab+CD4+ T-cell lineage: when, how and what for? Open Biol 3:120157. doi:10.1098/rsob.120157
Derhovanessian E, Solana R, Larbi A, Pawelec G (2008) Immunity, ageing and cancer. Immun Ageing 5:11. doi:10.1186/1742-4933-5-11
Ferrick DA, Schrenzel MD, Mulvania T et al (1995) Differential production of interferon-gamma and interleukin-4 in response to Th1- and Th2-stimulating pathogens by gamma delta T cells in vivo. Nature 373:255–257. doi:10.1038/373255a0
Foster AD, Sivarapatna A, Gress RE (2011) The aging immune system and its relationship with cancer. Aging Health 7:707–718. doi:10.2217/ahe.11.56
Frasca D, Landin AM, Lechner SC et al (2008) Aging down-regulates the transcription factor E2A, activation-induced cytidine deaminase, and Ig class switch in human B cells. J Immunol 180:5283–5290
Frasca D, Diaz A, Romero M et al (2011) Age effects on B cells and humoral immunity in humans. Ageing Res Rev 10:330–335. doi:10.1016/j.arr.2010.08.004
Frentsch M, Stark R, Matzmohr N et al (2013) CD40L expression permits CD8+ T cells to execute immunologic helper functions. Blood 122:405–412. doi:10.1182/blood-2013-02-483586
Giachino C, Granziero L, Modena V et al (1994) Clonal expansions of V delta 1+ and V delta 2+ cells increase with age and limit the repertoire of human gamma delta T cells. Eur J Immunol 24:1914–1918. doi:10.1002/eji.1830240830
Goronzy JJ, Weyand CM (2013) Understanding immunosenescence to improve responses to vaccines. Nat Immunol 14:428–436. doi:10.1038/ni.2588
Gregg R, Smith CM, Clark FJ et al (2005) The number of human peripheral blood CD4+ CD25high regulatory T cells increases with age. Clin Exp Immunol 140:540–546. doi:10.1111/j.1365-2249.2005.02798.x
Haas J, Fritzsching B, Trübswetter P et al (2007) Prevalence of newly generated naive regulatory T cells (Treg) is critical for Treg suppressive function and determines Treg dysfunction in multiple sclerosis. J Immunol 179:1322–1330
Haq K, McElhaney JE (2014) Immunosenescence: influenza vaccination and the elderly. Curr Opin Immunol 29:38–42. doi:10.1016/j.coi.2014.03.008
Hassan J, Reen DJ (2001) Human recent thymic emigrants--identification, expansion, and survival characteristics. J Immunol 167:1970–1976
Haynes L, Swain SL (2006) Why aging T cells fail: implications for vaccination. Immunity 24:663–666. doi:10.1016/j.immuni.2006.06.003
Hwang K-A, Kim H-R, Kang I (2009) Aging and human CD4(+) regulatory T cells. Mech Ageing Dev 130:509–517. doi:10.1016/j.mad.2009.06.003
Kang I, Hong MS, Nolasco H et al (2004) Age-associated change in the frequency of memory CD4+ T cells impairs long term CD4+ T cell responses to influenza vaccine. J Immunol 173:673–681
Kimmig S, Przybylski GK, Schmidt CA et al (2002) Two subsets of naive T helper cells with distinct T cell receptor excision circle content in human adult peripheral blood. J Exp Med 195:789–794
Koch S, Larbi A, Derhovanessian E et al (2008) Multiparameter flow cytometric analysis of CD4 and CD8 T cell subsets in young and old people. Immun Ageing 5:6. doi:10.1186/1742-4933-5-6
Kolar GR, Mehta D, Wilson PC, Capra JD (2006) Diversity of the Ig repertoire is maintained with age in spite of reduced germinal centre cells in human tonsil lymphoid tissue. Scand J Immunol 64:314–324. doi:10.1111/j.1365-3083.2006.01817.x
Lages CS, Suffia I, Velilla PA et al (2008) Functional regulatory T cells accumulate in aged hosts and promote chronic infectious disease reactivation. J Immunol 181:1835–1848
Lee N, Shin MS, Kang I (2012) T-cell biology in aging, with a focus on lung disease. J Gerontol A Biol Sci Med Sci 67:254–263. doi:10.1093/gerona/glr237
Li G, Yu M, Lee W-W et al (2012) Decline in miR-181a expression with age impairs T cell receptor sensitivity by increasing DUSP6 activity. Nat Med 18:1518–1524. doi:10.1038/nm.2963
National Institute on Aging (2011) Why population aging matters to health: A global perspective. National Institutes of Health
Nichol KL, Nordin JD, Nelson DB et al (2007) Effectiveness of influenza vaccine in the community-dwelling elderly. N Engl J Med 357:1373–1381. doi:10.1056/NEJMoa070844
Nikolich-Zugich J (2008) Ageing and life-long maintenance of T-cell subsets in the face of latent persistent infections. Nat Rev Immunol 8:512–522. doi:10.1038/nri2318
Olive M, Eisenlohr LC, Flomenberg P (2001) Quantitative analysis of adenovirus-specific CD4+ T-cell responses from healthy adults. Viral Immunol 14:403–413. doi:10.1089/08828240152716646
Osterholm MT, Kelley NS, Sommer A, Belongia EA (2012) Efficacy and effectiveness of influenza vaccines: a systematic review and meta-analysis. Lancet Infect Dis 12:36–44. doi:10.1016/S1473-3099(11)70295-X
Pang WW, Price EA, Sahoo D et al (2011) Human bone marrow hematopoietic stem cells are increased in frequency and myeloid-biased with age. Proc Natl Acad Sci U S A 108:20012–20017. doi:10.1073/pnas.1116110108
Pennesi G, Morellini M, Lulli P et al (2001) TCR Vβ repertoire in an Italian longeval population including centenarians. J Am Aging Assoc 24:63–70. doi:10.1007/s11357-001-0009-1
Pita-Lopez ML, Gayoso I, DelaRosa O et al (2009) Effect of ageing on CMV-specific CD8 T cells from CMV seropositive healthy donors. Immun Ageing A 6:11. doi:10.1186/1742-4933-6-11
Prelog M (2006) Aging of the immune system: a risk factor for autoimmunity? Autoimmun Rev 5:136–139. doi:10.1016/j.autrev.2005.09.008
R Core Team (2012) R: a language and environment for statistical computing. Vienna, Austria
Re F, Donnini A, Bartozzi B et al (2005) Circulating gammadelta T cells in young/adult and old patients with cutaneous primary melanoma. Immun Ageing A 2:2. doi:10.1186/1742-4933-2-2
Ross EM, Bourges D, Hogan TV et al (2014) Helios defines T cells being driven to tolerance in the periphery and thymus. Eur J Immunol. doi:10.1002/eji.201343999
Schwanninger A, Weinberger B, Weiskopf D et al (2008) Age-related appearance of a CMV-specific high-avidity CD8+ T cell clonotype which does not occur in young adults. Immun Ageing A 5:14. doi:10.1186/1742-4933-5-14
Sester M, Sester U, Alarcon Salvador S et al (2002) Age-related decrease in adenovirus-specific T cell responses. J Infect Dis 185:1379–1387. doi:10.1086/340502
Shi Y, Agematsu K, Ochs HD, Sugane K (2003) Functional analysis of human memory B-cell subpopulations: IgD+ CD27+ B cells are crucial in secondary immune response by producing high affinity IgM. Clin Immunol 108:128–137. doi:10.1016/S1521-6616(03)00092-5
Shi Y, Yamazaki T, Okubo Y et al (2005) Regulation of aged humoral immune defense against pneumococcal bacteria by IgM memory B cell. J Immunol 175:3262–3267
Simpson JG, Gray ES, Beck JS (1975) Age involution in the normal human adult thymus. Clin Exp Immunol 19:261–265
Smoot ME, Ono K, Ruscheinski J et al (2011) Cytoscape 2.8: new features for data integration and network visualization. Bioinforma 27:431–432. doi:10.1093/bioinformatics/btq675
Stervbo U, Meier S, Mälzer JN et al (2015) Effects of aging on human leukocytes (part I): immunophenotyping of innate immune cells. Age. doi:10.1007/s11357-015-9828-3
Stowe RP, Kozlova EV, Yetman DL et al (2007) Chronic herpesvirus reactivation occurs in aging. Exp Gerontol 42:563–570. doi:10.1016/j.exger.2007.01.005
Thornton AM, Korty PE, Tran DQ et al (2010) Expression of Helios, an Ikaros transcription factor family member, differentiates thymic-derived from peripherally induced Foxp3+ T regulatory cells. J Immunol 184:3433–3441. doi:10.4049/jimmunol.0904028
Vescovini R, Fagnoni FF, Telera AR et al (2014) Naïve and memory CD8 T cell pool homeostasis in advanced aging: impact of age and of antigen-specific responses to cytomegalovirus. Age 36:625–640. doi:10.1007/s11357-013-9594-z
Vukmanovic-Stejic M, Zhang Y, Cook JE et al (2006) Human CD4+ CD25hi Foxp3+ regulatory T cells are derived by rapid turnover of memory populations in vivo. J Clin Invest 116:2423–2433. doi:10.1172/JCI28941
Walford RL (1964) The immunologic theory of aging. The Gerontologist 4:195–197. doi:10.1093/geront/4.4.195
Wen L, Pao W, Wong FS et al (1996) Germinal center formation, immunoglobulin class switching, and autoantibody production driven by “non alpha/beta” T cells. J Exp Med 183:2271–2282
Wen L, Barber DF, Pao W et al (1998) Primary gamma delta cell clones can be defined phenotypically and functionally as Th1/Th2 cells and illustrate the association of CD4 with Th2 differentiation. J Immunol 160:1965–1974
Weng N (2006) Aging of the immune system: How much can the adaptive immune system adapt? Immunity 24:495–499. doi:10.1016/j.immuni.2006.05.001
Wickham H (2009) ggplot2: elegant graphics for data analysis, 1st ed. 2009. Corr. 3rd printing 2010. Springer
Wistuba-Hamprecht K, Frasca D, Blomberg B et al (2013) Age-associated alterations in γδ T-cells are present predominantly in individuals infected with Cytomegalovirus. Immun Ageing A 10:26. doi:10.1186/1742-4933-10-26
Acknowledgments
We would like to acknowledge the assistance of the BCRT Flow Cytometry Lab. The study was funded by the BMBF GerontoSys (Verbundprojekt PRIMAGE).
Competing interests
The authors declare that they have no competing interests.
Author contributions
CB, DR, SM, JM, MN, and AS performed the experiments. US, CB, DR, KJ, and UB performed the analysis. SO, NB, AG, AN, and AT conceptualized the overall strategy and developed clinical translation and implementation. All of the authors discussed the results and implications and commented on the manuscript at all stages. US drafted the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Ulrik Stervbo and Cecilia Bozzetti contributed equally to this work.
An erratum to this article can be found at http://dx.doi.org/10.1007/s11357-016-9928-8.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 2.21 mb)
About this article
Cite this article
Stervbo, U., Bozzetti, C., Baron, U. et al. Effects of aging on human leukocytes (part II): immunophenotyping of adaptive immune B and T cell subsets. AGE 37, 93 (2015). https://doi.org/10.1007/s11357-015-9829-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11357-015-9829-2