Abstract
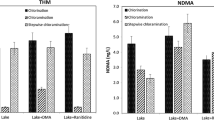
Formation of toxic N-nitrosodimethylamine (NDMA) by chloramination of ranitidine, a drug to block histamine, was still an ongoing issue and posed a risk to human health. In this study, the effect of ozonation prior to chloramination on NDMA formation and the transformation pathway were determined. Influencing factors, including ozone dosages, pH, hydroxyl radical scavenger, bromide, and NOM, were studied. The results demonstrated that small ozone dosage (0.5 mg/L) could effectively control NDMA formation from subsequent chloramination (from 40 to 0.8%). The NDMA molar conversion was not only influenced by pH but also by ozone dosages at various pre-ozonation pH (reached the highest value of 5% at pH 8 with 0.5 mg/L O3 but decreased with the increasing pH with 1 mg/L O3). The NDMA molar yield by chloramination of ranitidine without pre-ozonation was reduced by the presence of bromide ion due to the decomposition of disinfectant. However, due to the formation of brominated intermediate substances (i.e., dimethylamine (DMA), dimethyl-aminomethyl furfuryl alcohol (DFUR)) with higher NDMA molar yield than their parent substances, more NDMA was formed than that without bromide ion upon ozonation. Natural organic matter (NOM) and hydroxyl radical scavenger (tert-butyl alcohol, tBA) enhanced NDMA generation because of the competition of ozone and more ranitidine left. The NDMA reduction mechanism by pre-ozonation during chloramination of ranitidine may be due to the production of oxidation products with less NDMA yield (such as DMA) than parent compound. Based on the result of Q-TOF and GC-MS/MS analysis, three possible transformation pathways were proposed. Different influences of oxidation conditions and water quality parameters suggest that strategies to reduce NDMA formation should vary with drinking water sources and choose optimal ozone dosage.






Similar content being viewed by others
References
Ahmad S, Ali S, Alam N, Alam I, Alam S, Ali D (2016) Drug interactions of H2 receptor antagonists-ranitidine: a review. RJPT 9(3):275–280
Andrzejewski P, Kasprzyk-Hordern B, Nawrocki J (2008) N-nitrosodimethylamine (NDMA) formation during ozonation of dimethylamine-containing waters. Water Res 42(4-5):863–870. https://doi.org/10.1016/j.watres.2007.08.032
Chen Z, Valentine RL (2008) The influence of the pre-oxidation of natural organic matter on the formation of N-nitrosodimethylamine (NDMA). Environ Sci Technol 42(14):5062–5067. https://doi.org/10.1021/es8006673
China Environmental Protection Agency (2010) Determination of free chlorine and total chlorine -Spectrophotonetric method using N, N-diethyl-1, 4-phenylenediamine (HJ-586-2010)
Choi J, Valentine RL (2002) Formation of N-nitrosodimethylamine (NDMA) from reaction of monochloramine: a new disinfection by-product. Water Res 36(4):817–824. https://doi.org/10.1016/S0043-1354(01)00303-7
Christophoridis C, Nika MC, Aalizazadeh R, Thomaidis NS (2016) Ozonation of ranitidine: effect of experimental parameters and identification of transformation products. Sci Total Environ 557-558:170–182. https://doi.org/10.1016/j.scitotenv.2016.03.026
Elovitz MS, von Gunten U (1999) Hydroxyl Radical/Ozone Ratios During Ozonation Processes. I The Rct Concept Ozone Sci Eng 21(3):239–260
Flowers RC, Singer PC (2013) Anion exchange resins as a source of nitrosamines and nitrosamine precursors. Environ Sci Technol 47:107208649
Gan W, Bond T, Yang X, Westerhoff P (2015) Role of chlorine dioxide in N-Nitrosodimethylamine formation from oxidation of model amines. Environ Sci Technol 49(19):11429–11437. https://doi.org/10.1021/acs.est.5b01729
Jeon D, Kim J, Shin J, Hidayat ZR, Na S, Lee Y (2016) Transformation of ranitidine during water chlorination andozonation: moiety-specific reaction kinetics and elimination efficiency of NDMA formation potentia. J Hazard Mater 318:802–809. https://doi.org/10.1016/j.jhazmat.2016.06.039
Kasprzyk-Hordern B (2003) Catalytic ozonation and methods of enhancing molecular ozone reactions in water treatment. Appl Catal B Environ 46(4):639–669. https://doi.org/10.1016/S0926-3373(03)00326-6
Kasprzyk-Hordern B, Dinsdale RM, Guwy AJ (2008) The occurrence of pharmaceuticals, personal care products, endocrine disruptors and illicit drugs in surface water in South Wales, UK. Water Res 42(13):3498–3518. https://doi.org/10.1016/j.watres.2008.04.026
Kasprzyk-Hordern B, Dinsdale RM, Guwy AJ (2009) The removal of pharmaceuticals, personal care products, endocrine disruptors and illicit drugs during wastewater treatment and its impact on the quality of receiving waters. Water Res 43(2):363–380. https://doi.org/10.1016/j.watres.2008.10.047
Kemper JM, Walse SS, Mitch WA (2010) Quaternary amines as nitrosamine precursors: a role for consumer products? Environ Sci Technol 44(4):1224–1231. https://doi.org/10.1021/es902840h
Krasner SW, Westerhoff P, Chen B, Rittmann BE, Nam S, Amy G (2009) Impact of wastewater treatment processes on organic carbon, organic nitrogen, and DBP precursors in effluent organic matter. Environ Sci Technol 43(8):2911–2918. https://doi.org/10.1021/es802443t
Lamsal R, Walsh ME, Gagnon GA (2011) Comparison of advanced oxidation processes for the removal of natural organic matter. Water Res 45(10):3263–3269. https://doi.org/10.1016/j.watres.2011.03.038
Lee C, Schmidt C, Yoon J, von Gunten U (2007) Oxidation of N-Nitrosodimethylamine (NDMA) precursors with ozone and chlorine dioxide: kinetics and effect on NDMA formation potential. Environ Sci Technol 41(6):2056–2063. https://doi.org/10.1021/es062484q
Liao X, Chen C, Zhang J, Dai Y, Zhang X, Xie S (2015) Dimethylamine biodegradation by mixed culture enriched from drinking water biofilter. Chemosphere 119:935–940. https://doi.org/10.1016/j.chemosphere.2014.09.020
Liu YD, Selbes M, Zeng C, Zhong R, Karanfil T (2014) Formation mechanism of NDMA from ranitidine, trimethylamine, and other tertiary amines during Chloramination: a computational study. Environ Sci Technol 48(15):8653–8663. https://doi.org/10.1021/es500997e
Liu JJ, Liao XB, Zhou ZM, Li F, Yuan BL (2015) Determination of Seven N-Nitrosamines in Eutrophic Drinking Water after Chlorination by High Performance Liquid Chromatography-Tandem Mass Spectrometry. Chinese J Anal Chem 4:502–506
Lv J, Li Y, Song Y (2013) Reinvestigation on the ozonation of N-nitrosodimethylamine: influencing factors and degradation mechanism. Water Res 47(14):4993–5002. https://doi.org/10.1016/j.watres.2013.05.035
Lv J, Wang L, Song Y, Li Y (2015) N-nitrosodimethylamine formation from ozonation of chlorpheniramine: influencing factors and transformation mechanism. J Hazard Mater 299:584–594. https://doi.org/10.1016/j.jhazmat.2015.07.062
Marti EJ, Pisarenko AN, Peller JR, Dickenson ERV (2015) N-nitrosodimethylamine (NDMA) formation from the ozonation of model compounds. Water Res 72:262–270. https://doi.org/10.1016/j.watres.2014.08.047
McCurry DL, Krasner SW, von Gunten U, Mitch WA (2015) Determinants of disinfectant pretreatment efficacy for nitrosamine control in chloraminated drinking water. Water Res 84:161–170. https://doi.org/10.1016/j.watres.2015.07.024
McCurry DL, Ishida KP, Oelker GL, Mitch WA (2017) Reverse osmosis shifts chloramine speciation causing re-formation of NDMA during potable reuse of wastewater. Environ Sci Technol 51(15):8589–8596. https://doi.org/10.1021/acs.est.7b01641
Mitch WA, Gerecke AC, Sedlak DL (2003a) A N-Nitrosodimethylamine (NDMA) precursor analysis for chlorination of water and wastewater. Water Res 37(15):3733–3741. https://doi.org/10.1016/S0043-1354(03)00289-6
Mitch WA, Sharp JO, Trussell RR, Valentine RL, Alvarez-Cohen L, Sedlak DL (2003b) N-Nitrosodimethylamine (NDMA) as a drinking water contaminant: a review. Environ Eng Sci 20(5):389–404. https://doi.org/10.1089/109287503768335896
MOE (2003) Ontario Regulation 268/03 made Under the Safe Drinking Water Act
Najm I, Trussell RR (2001) NDMA formation in water and wastewater. J Am Water Works Ass 99:92–99
OEHHA (2006) Public health goal for N-nitrosodimethylamine and cadmium in drinking water
Oya M, Kosaka K, Asami M, Kunikane S (2008) Formation of N-nitrosodimethylamine (NDMA) by ozonation of dyes and related compounds. Chemosphere 73(11):1724–1730. https://doi.org/10.1016/j.chemosphere.2008.09.026
Padhye L, Luzinova Y, Cho M, Mizaikoff B, Kim J, Huang C (2011) PolyDADMAC and dimethylamine as precursors of N-Nitrosodimethylamine during ozonation: reaction kinetics and mechanisms. Environ Sci Technol 45(10):4353–4359. https://doi.org/10.1021/es104255e
Radjenović J, Sirtori C, Petrović M, Barceló D, Malato S (2010) Characterization of intermediate products of solar photocatalytic degradation of ranitidine at pilot-scale. Chemosphere 79(4):368–376. https://doi.org/10.1016/j.chemosphere.2010.02.014
Roux LJ, Gallard H, Croué J (2011) Chloramination of nitrogenous contaminants (pharmaceuticals and pesticides): NDMA and halogenated DBPs formation. Water Res 45(10):3164–3174. https://doi.org/10.1016/j.watres.2011.03.035
Roux JL, Gallard H, Croué J, Papot S, Deborde M (2012a) NDMA formation by Chloramination of ranitidine: kinetics and mechanism. Environ Sci Technol 46(20):11095–11103. https://doi.org/10.1021/es3023094
Roux LJ, Gallard H, Croué J (2012b) Formation of NDMA and halogenated DBPs by Chloramination of tertiary amines: the influence of bromide ion. Environ Sci Technol 46(3):1581–1589. https://doi.org/10.1021/es203785s
Sacher F, Lenz S, Brauch HJ (1997) Analysis of primary and secondary aliphatic amines in waste water and surface water by gas chromatography-mass spectrometry after derivatization with 2,4-dinitrofluorobenzene or benzenesulfonyl chloride. J Chromatogr A 764:85–93
Schmidt CK, Brauch H (2008) N,N-Dimethylsulfamide as precursor for N-Nitrosodimethylamine (NDMA) formation upon ozonation and its fate during drinking water treatment. Environ Sci Technol 42(17):6340–6346. https://doi.org/10.1021/es7030467
Selbes M, Kim D, Ates N, Karanfil T (2013) The roles of tertiary amine structure, background organic matter and chloramine species on NDMA formation. Water Res 47(2):945–953. https://doi.org/10.1016/j.watres.2012.11.014
Selbes M, Kim D, Karanfil T (2014) The effect of pre-oxidation on NDMA formation and the influence of pH. Water Res 66:169–179. https://doi.org/10.1016/j.watres.2014.08.015
Sgroi M, Roccaro P, Oelker G, Snyder SA (2016) N-nitrosodimethylamine (NDMA) formation during ozonation of wastewater and water treatment polymers. Chemosphere 144:1618–1623. https://doi.org/10.1016/j.chemosphere.2015.10.023
Shah AD, Mitch WA (2012) Halonitroalkanes, Halonitriles, Haloamides, and N-nitrosamines: a critical review of nitrogenous disinfection byproduct formation pathways. Environ Sci Technol 46(1):119–131. https://doi.org/10.1021/es203312s
Shen R, Andrews SA (2011a) NDMA formation kinetics from three pharmaceuticals in four water matrices. Water Res 45(17):5687–5694. https://doi.org/10.1016/j.watres.2011.08.034
Shen R, Andrews SA (2011b) Demonstration of 20 pharmaceuticals and personal care products (PPCPs) as nitrosamine precursors during chloramine disinfection. Water Res 45(2):944–952. https://doi.org/10.1016/j.watres.2010.09.036
Shen R, Andrews SA (2013a) Formation of NDMA from ranitidine and sumatriptan: the role of pH. Water Res 47(2):802–810. https://doi.org/10.1016/j.watres.2012.11.004
Shen R, Andrews SA (2013b) NDMA formation from amine-based pharmaceuticals-impact from prechlorination and water matrix. Water Res 47(7):2446–2457. https://doi.org/10.1016/j.watres.2013.02.017
Silva BFD, Jelic A, López-Serna R, Mozeto AA, Petrovic M, Barceló D (2011) Occurrence and distribution of pharmaceuticals in surface water, suspended solids and sediments of the Ebro river basin, Spain. Chemosphere 85(8):1331–1339. https://doi.org/10.1016/j.chemosphere.2011.07.051
Spahr S, Cirpka OA, von Gunten U, Hofstetter TB (2017) Formation of N-Nitrosodimethylamine during Chloramination of secondary and tertiary amines: role of molecular oxygen and radical intermediates. Environ Sci Technol 51(1):280–290. https://doi.org/10.1021/acs.est.6b04780
USEPA (1993) N-Nitrosodimethylamine, Integrated Risk Information System
Vikesland PJ, Ozekin K, Valentine RL (2001) Monochloramine decay in model and distribution system waters. Water Res 35(7):1766–1776. https://doi.org/10.1016/S0043-1354(00)00406-1
von Gunten U (2003) Ozonation of drinking water: part I. Oxidation kinetics and product formation. Water Res 37(7):1443–1467. https://doi.org/10.1016/S0043-1354(02)00457-8
von Gunten U, Salhi E, Schmidt CK, Arnold WA (2010) Kinetics and mechanisms of N-Nitrosodimethylamine formation upon ozonation of N,N-Dimethylsulfamide-containing waters: bromide catalysis. Environ Sci Technol 44(15):5762–5768. https://doi.org/10.1021/es1011862
Wang X, Yang H, Zhou B, Wang X, Xie Y (2015) Effect of oxidation on amine-based pharmaceutical degradation and N-Nitrosodimethylamine formation. Water Res 87:403–411. https://doi.org/10.1016/j.watres.2015.07.045
Zeng T, Mitch WA (2015) Contribution of N-nitrosamines and their precursors to domestic sewage by greywaters and Blackwaters. Environ Sci Technol 49(22):13158–13167. https://doi.org/10.1021/acs.est.5b04254
Zeng T, Mitch WA (2016) Oral intake of ranitidine increases urinary excretion of N-Nitrosodimethylamine. Carcinogenesis 37(6):625–634. https://doi.org/10.1093/carcin/bgw034
Zhao Y, Boyd JM, Woodbeck M, Andrews RC, Qin F, Hrudey SE, Li X (2008) Formation of N-nitrosamines from eleven disinfection treatments of seven different surface waters. Environ Sci Technol 42(13):4857–4862. https://doi.org/10.1021/es7031423
Acknowledgements
This work was financially supported by National Natural Science Foundation of China (No. 51508209; 51678255), 1st class General Financial Grant (No. 2015M580561), and Special Financial Grant (No. 2016T90595) from China Post-Doctoral Funding, Natural Science Foundation of Fujian province (No. 2015J05102), Scientific Research project for Young teachers supported by Education Department of Fujian Province (No. JA15033), the Science Research Foundation of Huaqiao University (No. 15BS105), and it was also supported by Promotion Program for Young and Middle-aged Teacher in Science and Technology Research of Huaqiao University (ZQN-PY413).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible editor: Bingcai Pan
Electronic supplementary material
ESM 1
(DOCX 1616 kb)
Rights and permissions
About this article
Cite this article
Zou, R., Liao, X., Zhao, L. et al. Reduction of N-nitrosodimethylamine formation from ranitidine by ozonation preceding chloramination: influencing factors and mechanisms. Environ Sci Pollut Res 25, 13489–13498 (2018). https://doi.org/10.1007/s11356-018-1470-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-1470-z