Abstract
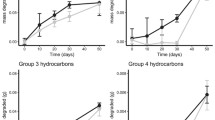
The aim of the present study was to assess the bioremediation of estuarine sediments contaminated with diesel oil. The following two experiments were performed: natural attenuation (NA) and stimulated natural attenuation (SNA), using rhamnolipid as biosurfactant. Sediment samples were accommodated into glass columns and then contaminated with diesel oil on the top. The column profiles were separated into surface, middle, and bottom for the analyses. The 16 polycyclic aromatic hydrocarbons (PAHs) prioritized by US Environmental Protection Agency (EPA) were monitored for 349 days. Those with three and four rings showed increasing concentrations through the operation period in the middle and bottom samples, particularly between days 111 and 338, and in the SNA experiment. Those with five and six rings were also detected in the deeper portions of the columns, suggesting the percolation of PAHs with a high molecular weight. Total organic carbon was reduced by 91 and 89 % in the NA and SNA samples, respectively, although no statistically significant differences (p > 0.05) were found between the two treatments. The analyses by denaturing gradient gel electrophoresis indicated a slight shift in the microbial community structure over the experiments. Microorganisms belonging to the γ-Proteobacteria phylum were the main bacteria involved. The archaeal community exhibited dominance of hydrogenotrophic methanogens, indicating the obligate anaerobic biodegradation of intermediate compounds from hydrocarbon degradation.







Similar content being viewed by others
References
Abdel-Mawgoud AM, Lépine F, Déziel E (2010) Rhamnolipids: diversity of structures, microbial origins and roles. Appl Microbiol Biotechnol 86(5):1323–1336. doi:10.1007/s00253-010-2498-2
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic Local Alignment Search Tool. J Mol Biol 215(3):403–410. doi:10.1016/S0022-2836(05)80360-2
Alves BJR, Baêta AM, Alves JV (1999) Embrapa agrobiology protocol for nitrogen analysis in organic fertilizers, soil and tissue. Seropédica: Embrapa Agrobiologia 100 (in Portuguese)
APHA/AWWA American Public Health Association, American Water Works Association (2005) Standard methods for the examination of water and wastewater 21th ed
Bacosa HP, Inoue C (2015) Polycyclic aromatic hydrocarbons (PAHs) biodegradation potential and diversity of microbial consortia enriched from tsunami sediments in Miyagi, Japan. J Hazard Mater 283:689–697. doi:10.1016/j.jhazmat.2014.09.068
Ball A, Truskewycz A (2013) Polyaromatic hydrocarbon exposure: an ecological impact ambiguity. Environ Sci Pollut Res 20:4311–4326. doi:10.1007/s11356-013-1620-2
Bayer DM, Chagas-Spinelli ACO, Gavazza S, Florencio L, Kato MT (2013) Natural attenuation and biosurfactant-stimulated bioremediation of estuarine sediments contaminated with diesel oil. Appl Biochem Biotechnol 171:173–188. doi:10.1007/s12010-013-0358-0
Bento FM, Camargo FAO, Okeke BC, Frankenberger WT Jr (2005) Diversity of biosurfactant producing microorganisms isolated from soils contaminated with diesel oil. Microbiol Res 160(3):249–255. doi:10.1016/j.micres.2004.08.005
Berry DF, Francis AJ, Bollag J-M (1987) Microbial metabolism of homocyclic and heterocyclic aromatic compounds under anaerobic conditions. Microbiol Rev 51(1):43–59
Boopathy R (2003) Use of anaerobic soil slurry reactors for the removal of petroleum hydrocarbons in soil. Int Biodeterior Biodegrad 52:161–166. doi:10.1016/S0964-8305(03)00054-4
Bordoloi NK, Konwar BK (2009) Bacterial biosurfactant in enhancing solubility and metabolism of petroleum hydrocarbons. J Hazard Mater 170:495–505. doi:10.1016/j.jhazmat.2009.04.136
Bustamante M, Durán N, Diez MC (2012) Biosurfactants are useful tools for the bioremediation of contaminated soil: a review. J Soil Sci Plant Nutr [online] 12(4):667–687. doi:10.4067/S0718-95162012005000024
Cerniglia CE (1992) Biodegradation of polycyclic aromatic hydrocarbons. Biodegradation 3:351–368. doi:10.1007/BF00129093
Chagas-Spinelli ACO, Kato MT, Lima ES, Gavazza S (2012) Bioremediation of a tropical clay soil contaminated with diesel oil. J Environ Manag 113:510–516. doi:10.1016/j.jenvman.2012.05.027
Chang Y-T, Lee JF, Chao H-P (2007) Variability of communities and physiological characteristics between free-living bacteria and attached bacteria during the PAH biodegradation in a soil/water system. Eur J Soil Biol 43:283–296. doi:10.1016/j.ejsobi.2007.02.012
Chen Q, Bao M, Fan X, Liang S, Sun P (2013) Rhamnolipids enhance marine oil spill bioremediation in laboratory system. Mar Pollut Bull 71:269–275. doi:10.1016/j.marpolbul.2013.01.037
Chiou CT (1989) Partition and adsorption on soil and mobility of organic pollutants and pesticides. Toxic Org Chem Porous Media 73:163–175. doi:10.1007/978-3-642-74468-6_7
Chrzanowski Ł, Dziadas M, Ławniczak Ł, Cyplik P, Białas W, Szulc A, Lisiecki P, Jeleń H (2012a) Biodegradation of rhamnolipids in liquid cultures: effect of biosurfactant dissipation on diesel fuel/B20 blend biodegradation efficiency and bacterial community composition. Bioresour Technol 111:328–335. doi:10.1016/j.biortech.2012.01.181
Chrzanowski Ł, Ławniczak Ł, Czaczyk K (2012b) Why do microorganisms produce rhamnolipids? World J Microbiol Biotechnol 28(2):401–419. doi:10.1007/s11274-011-0854-8
Colleran E (1997) Uses of bacteria in bioremediation. In: Sheehan D (ed) Bioremediation protocols—methods in biotechnology 2. Humana Press, New Jersey, pp. 3–22. doi:10.1385/0-89603-437-2:3
Delle Site A (2001) Factors affecting sorption of organic compounds in natural sorbent/water systems and sorption coefficients for selected pollutants: a review. J Phys Chem Ref Data 30(1):187–439. doi:10.1063/1.1347984
Díaz E (2004) Bacterial degradation of aromatic pollutants: a paradigm of metabolic versatility. Int Microbiol 7:173–180
EPA United States Environmental Protection Agency (1996a) Method 3540C. http://www.epa.gov/solidwaste/hazard/testmethods/sw846/pdfs/3540c.pdf Accessed 17 April 2015
EPA United States Environmental Protection Agency (1996b) Method 3610B. http://www.epa.gov/solidwaste/hazard/testmethods/sw846/pdfs/3610b.pdf Accessed 17 April 2015
EPA United States Environmental Protection Agency (1996c) Method 3630C. http://www.epa.gov/solidwaste/hazard/testmethods/sw846/pdfs/3630c.pdf Accessed 17 April 2015
EPA United States Environmental Protection Agency (1997) Method 8270D. http://www.epa.gov/osw/hazard/testmethods/sw846/pdfs/8270d.pdf Accessed 17 April 2015
EPA United States Environmental Protection Agency (2016) Priority pollutant list. https://www.epa.gov/sites/production/files/2015-09/documents/priority-pollutant-list-epa.pdf Accessed 15 June 2016
Giri C, Ochieng E, Tieszen LL, Zhu Z, Singh A, Loveland T, Masek J, Duke N (2010) Status and distribution of mangrove forests of the world using earth observation satellite data. Glob Ecol Biogeogr 20:154–159. doi:10.1111/j.1466-8238.2010.00584.x
Grasshoff K, Kremling K, Ehrhardt M (1999) Methods of seawater analysis. Wiley, New York
Guerin TF (2015) A safe, efficient and cost effective process for removing petroleum hydrocarbons from a highly heterogeneous and relatively inaccessible shoreline. J Environ Manag 162:190–198. doi:10.1016/j.jenvman.2015.07.016
IARC International Agency for Research on Cancer (2010) Agents classified by the IARC monographs, v. 1–100. http://monographs.iarc.fr/ENG/Classification/index.php Accessed 06 July 2015
Isaac P, Sánchez LA, Bourguignon N, Cabral ME, Ferrero MA (2013) Indigenous PAH-degrading bacteria from oil-polluted sediments in Caleta Cordova, Patagonia Argentina. Int Biodeterior Biodegrad 82:207–214. doi:10.1016/j.ibiod.2013.03.009
Johnsen AR, Wick LY, Harms H (2005) Principles of microbial PAH-degradation in soil. Environ Pollut 133:71–84. doi:10.1016/j.envpol.2004.04.015
Kudo Y, Nakajima T, Miyaki T, Oyaizu H (1997) Methanogen flora of paddy soils in Japan. FEMS Microbiol Ecol 22:39–48. doi:10.1016/S0168-6496(96)00073-6
Kumari B, Singh SN, Singh DP (2012) Characterization of two biosurfactant producing strains in crude oil degradation. Process Biochem 47:2463–2471. doi:10.1016/j.procbio.2012.10.010
Ławniczak Ł, Marecik R, Chrzanowski Ł (2013) Contributions of biosurfactants to natural or induced bioremediation. Appl Microbiol Biotechnol 97:2327–2339. doi:10.1007/s00253-013-4740-1
Lemos RT, de Carvalho PS, Zanardi-Lamardo E (2014) Petroleum hydrocarbons in water from a Brazilian tropical estuary facing industrial and port development. Mar Pollut Bull 82(1–2):183–188. doi:10.1016/j.marpolbul.2014.03.010
Leys NM, Bastiaens L, Verstraete W, Springael D (2005) Influence of the carbon/nitrogen/phosphorus ratio on polycyclic aromatic hydrocarbon degradation by Mycobacterium and Sphingomonas in soil. Appl Microbiol Biotechnol 66:726–736. doi:10.1007/s00253-004-1766-4
Li CH, Zhou HW, Wong YS, Tam NFY (2009) Vertical distribution and anaerobic biodegradation of polycyclic aromatic hydrocarbons in mangrove sediments in Hong Kong, South China. Sci Total Environ 407:5772–5779. doi:10.1016/j.scitotenv.2009.07.034
Liu Z, Liu J (2013) Evaluating bacterial community structures in oil collected from the sea surface and sediment in the northern Gulf of Mexico after the deepwater horizon oil spill. Microbiol Open 2(3):492–504. doi:10.1002/mbo3.89
Loring DH, Rantala RTT (1992) Manual for the geochemical analyses of marine sediments and suspended particulate matter. Earth-Sci Rev 32(4):235–283. doi:10.1016/0012-8252(92)90001-A
Lu X-Y, Zhang T, Fang HH-P (2011) Bacteria-mediated PAH degradation in soil and sediment. Appl Microbiol Biotechnol 89:1357–1371. doi:10.1007/s00253-010-3072-7
Maciel-Souza MC, Macrae A, Volpon AGT, Ferreira PS, Mendonça-Hagler LC (2006) Chemical and microbiological characterization of mangrove sediments after a large oil-spill in Guanabara Bay-RJ-Brazil. Braz J Microbiol 37(3):262–266. doi:10.1590/S1517-83822006000300013
Manoli E, Samara C (1999) Polycyclic aromatic hydrocarbons in natural waters: sources, occurrence and analysis. Trends Anal Chem 18(6):417–428. doi:10.1016/S0165-9936(99)00111-9
McElroy AE, Farrington JW, Teal JM (1989) Bioavailability of polycyclic aromatic hydrocarbons in the aquatic environment. In: Varanassi U (ed) Metabolism of polycyclic aromatic hydrocarbons in the aquatic environment. CRC Press, Florida, pp. 1–39 NOAA Contract 83-ABD-00012
Molina-Barahona L, Rodrı́guez-Vázquez R, Hernández-Velasco M, Vega-Jarquı́n C, Zapata-Pérez O, Mendoza-Cantú A, Albores A (2004) Diesel removal from contaminated soils by biostimulation and supplementation with crop residues. Appl Soil Ecol 27(2):165–175. doi:10.1016/j.apsoil.2004.04.002
Muyzer G, De Waal EC, Uitterlinden AG (1993) Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. Appl Environ Microbiol 59(3):695–700
Nielsen TA, Liu W-T, Filipe C, Grady L, Molin S, Stahl DA (1999) Identification of a novel group of bacteria in sludge from deteriorated biological phosphorus removal reactor. Appl Environ Microbiol 65:1251–1258
Niepceron M, Martin-Laurent F, Crampon M, Portet-Koltalo F, Akpa-Vinceslas M, Legras M, Bru D, Bureau F, Bodilis J (2013) Gamma Proteobacteria as a potential bioindicator of a multiple contamination by polycyclic aromatic hydrocarbons (PAHs) in agricultural soils. Environ Pollut 180:199–205. doi:10.1016/j.envpol.2013.05.040
Nikolopoulou M, Pasadakis N, Kalogerakis N (2013) Evaluation of autochthonous bioaugmentation and biostimulation during microcosm-simulated oil spills. Mar Pollut Bull 72:165–173. doi:10.1016/j.marpolbul.2013.04.007
Pacwa-Płociniczak M, Plaza GA, Poliwoda A, Piotrowska-Seget Z (2014) Characterization of hydrocarbon-degrading and biosurfactant-producing Pseudomonas sp. P-1 strain as a potential tool for bioremediation of petroleum-contaminated soil. Environ Sci Pollut Res 21:9385–9395. doi:10.1007/s11356-014-2872-1
Parshina SN, Sipma J, Nakashimada Y, Henstra AM, Smidt H, Lysenko AM, Lens PNL, Lettinga G, Stams AJM (2005) Desulfotomaculum carboxydivorans sp. nov., a novel sulfate-reducing bacterium capable of growth at 100 % CO. Int J Syst Evol Microbiol 55:2159–2165. doi:10.1099/ijs.0.63780-0
Protazio L, Neiva Tanaka SMC, Cavalcante PRS (2004) Evaluation of phosphorus sequential extraction procedures in sediment. Rev Anal 8 (in Portuguese)
Rengarajan T, Rajendran P, Nandakumar N, Lokeshkumar B, Rajendran P, Nishigaki I (2015) Exposure to polycyclic aromatic hydrocarbons with special focus on cancer. Asian Pac J Trop Biomed 5(3):182–189. doi:10.1016/S2221-1691(15)30003-4
Rocchetti L, Beolchini F, Hallberg KB, Johnson DB, Dell’Anno A (2012) Effects of prokaryotic diversity changes on hydrocarbon degradation rates and metal partitioning during bioremediation of contaminated anoxic marine sediments. Mar Pollut Bull 64:1688–1698. doi:10.1016/j.marpolbul.2012.05.038
Ruiz-Marín A, Zavala-Loria JC, Canedo-López Y, Cordova-Quiroz AV (2013) Tropical bacteria isolated from oil-contaminated mangrove soil: bioremediation by natural attenuation and bioaugmentation. Rev Mex Ing Quím 12(3):553–560 http://www.redalyc.org/pdf/620/62029966017.pdfAccessed 09 March 2016
Santos LCM, Cunha-Lignon M, Schaeffer-Novelli Y, Cintrón-Molero G (2012) Long-term effects of oil pollution in mangrove forests (Baixada Santista, Southeast Brazil) detected using a GIS-based multitemporal analysis of aerial photographs. Braz J Oceanogr 60(2):159–170. doi:10.1590/S1679-87592012000200006
Silva DMC, Araújo JA, Gavazza S, Souto-Maior AM, Chagas-Spinelli ACO (2009) Evaluation of rhamnolipid production efficiency from different nitrogen sources by Pseudomonas aeruginosa isolates from oil well. In: XVII National Symposium on Bioprocesses. Brazil, Natal (in Portuguese)
Szulc A, Ambrożewicz D, Sydow M, Ławniczak Ł, Piotrowska-Cyplik A, Marecik R, Chrzanowski Ł (2014) The influence of bioaugmentation and biosurfactant addition on bioremediation efficiency of diesel-oil contaminated soil: feasibility during field studies. J Environ Manag 132:121–128. doi:10.1016/j.jenvman.2013.11.006
Tam NFY, Wong YS (2008) Effectiveness of bacterial inoculum and mangrove plants on remediation of sediment contaminated with polycyclic aromatic hydrocarbons. Mar Pollut Bull 57(6–12):716–726. doi:10.1016/j.marpolbul.2008.02.029
Tansel B, Lee M, Tansel DZ (2013) Comparison of fate profiles of PAHs in soil, sediments and mangrove leaves after oil spills by QSAR and QSPR. Mar Pollut Bull 73:258–262. doi:10.1016/j.marpolbul.2013.05.011
Tiehm A (1994) Degradation of polycyclic aromatic hydrocarbons in the presence of synthetic surfactants. Appl Environ Microbiol 60(1):258–263
Trindade PVO, Leite SGF, Sobral LG, Rizzo ACL, Lemos JLS, Millioli VS, Santos R, Soriano AU (2002) Evaluation of biostimulation and bioaugmentation techniques in the bioremediation’s process of petroleum hydrocarbons contaminated soil. In: 9th International Petroleum Environmental Conference. USA, Albuquerque, New Mexico
Viñas M, Sabaté J, Espuny MJ, Solanas AM (2005) Bacterial community dynamics and polycyclic aromatic hydrocarbon degradation during bioremediation of heavily creosote-contaminated soil. Appl Environ Microbiol 71(11):7008–7018. doi:10.1128/AEM.71.11.7008-7018.2005
Wang L, Li F, Zhan Y, Zhu L (2016) Shifts in microbial community structure during in situ surfactant-enhanced bioremediation of polycyclic aromatic hydrocarbon-contaminated soil. Environ Sci Pollut Res Int. doi:10.1007/s11356-016-6630-4
Widdel F, Rouvière PE, Wolfe RS (1988) Classification of secondary alcohol-utilizing methanogens including a new thermophilic isolate. Arch Microbiol 150(4):477–481. doi:10.1007/BF00422290
Yu KSH, Wong AHY, Yau KWY, Wong YS, Tam NFY (2005) Natural attenuation, biostimulation and bioaugmentation on biodegradation of polycyclic aromatic hydrocarbons (PAHs) in mangrove sediments. Mar Pollut Bull 51:1071–1077. doi:10.1016/j.marpolbul.2005.06.006
Zhu X, Venosa AD, Suidan MT, Lee K (2004) Guidelines for the bioremediation of oil-contaminated salt marshes. United States Environmental Protection Agency, EPA 600-R-04-074. http://edocs.dlis.state.fl.us/fldocs/oilspill/federal/LPS68040.pdf Accessed 25 July 2013
Acknowledgments
The authors are grateful for the financial support provided by the following Brazilian agencies: the Fundação de Amparo à Ciência e Tecnologia do Estado de Pernambuco (FACEPE; IBPG-1414-3.07/08, BFM-0008-3.07/11), the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), the Agência Nacional do Petróleo, Gás Natural e Biocombustíveis (ANP), and the Financiadora de Estudos e Projetos (FINEP). The authors would also like to thank PETROBRAS for supporting the RELINE/RECUPETRO network (UFPE, UFC, UFRN, UFCG, UFAL, and UFBA) and LMA-CTG-SENAI/RN for the analyses of hydrocarbons.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Robert Duran
Electronic supplementary material
ESM 1
(DOCX 68 kb)
Rights and permissions
About this article
Cite this article
Nakazawa, M.M., Gavazza, S., Kato, M.T. et al. Evaluation of rhamnolipid addition on the natural attenuation of estuarine sediments contaminated with diesel oil. Environ Sci Pollut Res 24, 25522–25533 (2017). https://doi.org/10.1007/s11356-016-7152-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-016-7152-9