Abstract
Synthesis of amylase by Aspergillus niger strain UO-01 under solid-state fermentation with sugarcane bagasse was optimized by using response surface methodology and empirical modelling. The process parameters tested were particle size of sugarcane bagasse, incubation temperature and pH, moisture level of solid support material and the concentrations of inoculum, total sugars, nitrogen and phosphorous. The optimum conditions for high amylase production (457.82 EU/g of dry support) were particle size of bagasse in the range of 6–8 mm, incubation temperature and pH: 30.2°C and 6.0, moisture content of bagasse: 75.3%, inoculum concentration: 1 × 107 spores/g of dry support and concentrations of starch, yeast extract and KH2PO4: 70.5, 11.59 and 9.83 mg/g of dry support, respectively. After optimization, enzyme production was assayed at the optimized conditions. The results obtained corroborate the effectiveness and reliability of the empirical models obtained.
Similar content being viewed by others
Introduction
The Aspergillus species produce a large variety of extracellular enzymes of which amylases are of world-wide interest in fermentation, food, pharmaceutical, textile and paper industries (Bhargav et al. 2008; Uma Maheswar Rao and Satyanarayana 2007). Production of amylases by Aspergillus strains in both submerged liquid fermentation (SLF) and solid-state fermentation (SSF) by using different food wastes or agricultural residues has been thoroughly studied (Ellaiah et al. 2002; Francis et al. 2002, 2003; Murado et al. 1997; Salas et al. 2006). However, comparative studies between SLF and SSF claim higher yields and other advantages for products obtained by SSF, such as low energy requirements, lower availability of water that reduces the possibilities of contamination by bacteria and yeast, small volumes of polluting effluents and low downstream processing cost (Guerra et al. 2003; Raimbault 1998).
The production of amylases in SSF is affected by a variety of physicochemical factors, including the type and nutrient composition of the substrate, incubation temperature, pH, aeration, concentration and type of the carbon, phosphate and nitrogen sources, concentration and age of the inoculum, particle size and moisture level of the substrate (Balkan and Ertan 2007; Ramachandran et al. 2004; Rodríguez and Sanromán 2006). Therefore, after selecting a culture medium for amylase production, the fermentation conditions must be optimized to improve enzyme production at a low production cost (Balkan and Ertan 2007; Spier et al. 2006).
In recent years, there has been an increasing interest towards more efficient utilization of different agro-industrial residues, including sugarcane bagasse, wheat bran, wheat straw, rye straw and corncob leaf and oil cakes, for amylases production (Balkan and Ertan 2007; Bhargav et al. 2008; Ramachandran et al. 2004). Sugarcane bagasse has been used as a solid inert support for the production of value-added products including l-glutamic acid, ergot alkaloids, penicillin, fruity aroma, lactic acid, pigments and pectinases (Pandey et al. 2000). However, to our knowledge, there is not information available on the use of sugarcane bagasse as a support material for the production of amylases by Aspergillus strains.
Considering the substantial availability of sugarcane bagasse at very low prices by local sugar cane factories of Santiago de Cuba, its use as a support material could offer an attractive possibility for a low cost production of amylases by Aspergillus niger strain UO-01 in SSF. Since the amylase production by this strain was studied before in SLF (Salas et al. 2006), the aim of the present study was to optimize enzyme production by SSF conditions. Particle size of sugarcane bagasse was firstly optimized in a single parameter study. Subsequently, other important process parameters were optimized in a sequential order by using response surface methodology (RSM) and empirical modelling. The other process variables optimized were incubation pH and temperature (second experiment), moisture level, inoculum and total sugars concentrations (third experiment) and the concentrations of phosphorous and six nitrogen sources (fourth experiment). Finally amylase production was assayed at the optimized conditions to verify the effectiveness and the accuracy of the empirical models obtained.
Materials and methods
Microorganism
Aspergillus niger strain UO-01, was acquired from the Biotechnology Center of the University of Oriente (Santiago de Cuba, Cuba) and it was maintained on potato dextrose agar slants at 4°C (Ellaiah et al. 2002; Salas et al. 2006).
Inoculum preparation, culture media and fermentation conditions
Inocula were prepared by transferring 2-ml of 60-h old slant culture in 50 ml of medium (250 ml Erlenmeyer) composed by (g l−1): glucose, 20; (NH4)2SO4, 6.6; KH2PO4, 3.5; FeSO4·7H2O, 0.15; MgSO4·7H2O, 0.10; MnCl2·2H2O, 0.45 and mycological peptone, 3.0; at pH 6.8. The culture was incubated at 30°C and 220 rev/min for 48 h (Salas et al. 2006).
Sugarcane bagasse obtained from a local sugar cane factory of Santiago de Cuba was used as a solid support material. It was treated with NaOH [0.12 g of NaOH/g of dry support (gds)] and autoclaved at 121°C for 20 min (Gutiérrez-Correa and Tengerdy 1997) to remove the core and noncore lignin fractions (Doran et al. 1994). Then, the samples of sugarcane bagasse were thoroughly washed with tap water, subsequently with distilled water until neutrality and dried at 80°C (Doran et al. 1994).
Fermentations were conducted in 250-ml Erlenmeyer flasks containing 5 g of dry bagasse which was moistened with the liquid media (salt solution and distilled water) previously supplemented with soluble potato starch to get desired level of sugar in the solid support material. The salt solution was prepared to provide (%, g/g of dry bagasse), NaNO3, 1; KH2PO4, 1; NaCl, 0.2; MgSO47H2O, 0.2 (Francis et al. 2003).
The contents of the flasks were mixed thoroughly and then autoclaved at 121°C for 15 min. After cooling, the sterile media were inoculated with 1.25 ml of inoculum containing the appropriate cell suspension (spores/gds) and thoroughly mixed. Then, the flasks were incubated at the corresponding temperature in a chamber with temperature and humidity control. Samples as whole flasks in triplicate were withdrawn each 12 h.
Extraction of the enzyme
After fermentations, the whole sample of each flask was extracted by the addition of distilled water containing 0.1% Tween-80, to a total extract volume of 100 ml. Contents were mixed thoroughly (150 rev/min, at room temperature for 1 h) in a rotary shaker and filtered using a Whatman filter paper No. 1. The culture filtrates were used as the crude enzyme extracts (Francis et al. 2003).
Analytical methods
Total amylase activity (TAA) was determined at 40°C by mixing appropriately the diluted crude enzyme extract with 0.15 M citrate-phosphate buffer (pH 5.0) and 4% soluble starch (previously maintained at 40°C/15 min) and determining the reducing sugars (glucose equivalents) after 10 min (Murado et al. 1993). One unit of amylase activity [enzymatic units (EU)/ml] was defined as the amount of enzyme releasing 1 mg/ml of reducing sugars/min under the assay conditions. Then, the amylase activity units were expressed as EU/gds.
Method for determination of total sugars in the supernatants was described or referred to in a previous work (Salas et al. 2006). All determinations were carried out in triplicate.
Optimization of process parameters in SSF
The particle size (PS) was optimized by using the “one variable at a time” method for amylase production. Thus, in a first experiment, the bagasse was milled to different particle sizes (<1, 1–2, 2–4, 4–6, 6–8, 8–10, 10–12, 12–14, 14–16 and 16–20 mm), which were used as support materials to determine the appropriate particle size for maximal enzyme production in SSF. The dried bagasse was impregnated with 2.5 ml of the salt solution buffered at pH 6.0 with potassium hydrogen phthalate-HCl buffer (0.1 M) and 16.25 ml of distilled water previously supplemented with soluble potato starch to get 26 mg of total sugars/gds. After sterilization, the media were inoculated with 1.25 ml of inoculum to give an inoculum concentration of 1 × 107 spores/gds. The final moisture content after inoculation was calculated to be as 80%. The flasks were incubated at 30°C until the total sugars were completely consumed.
Then, a three-step optimization strategy using RSM and empirical modelling (Uma Maheswar Rao and Satyanarayana 2007) was employed to optimize amylase production. In the first step, initial culture pH and temperature (T) were optimized using the optimum particle size of the sugarcane bagasse calculated in the previous experiment. In this assay, the liquid medium was buffered at different pHs with the appropriate buffer (0.1 M potassium hydrogen phthalate-HCl buffer for pHs 3.0, 3.5 and 5.5 or 0.1 M sodium phosphate buffer for pHs 7.5 and 8.0) and the flasks were incubated for 84 h at the corresponding temperature according to the experimental matrix defined by the design used (Table 1).
In the second step, the levels of three factors: moisture content (M), inoculum concentration (IC) and total sugars concentration (TS) were then optimized in the optimal conditions (PS, T and pH) calculated in the two previous experiments. In this assay, sugarcane bagasse was impregnated with 2.5 ml of the salt solution and an appropriate volume of distilled water (both at pH 6.0) containing different amounts of soluble potato starch to give the different moisture contents and total sugars concentrations defined in Table 1. The sterile media were inoculated with 1.25 ml of a suspension containing the appropriate amount of spores to obtain the corresponding inoculum concentrations (Table 1). All the media were then incubated at 30°C for 84 h.
In the third optimization step, the concentrations of the nitrogen (N) and phosphorous (P) sources were optimized by including the appropriate nitrogen source [NH4CH3COO, (NH4)2HPO4, NaNO2, NaNO3, yeast extract or (NH4)2SO4] and KH2PO4 in the salt solution to give the concentrations indicated in Table 1. This last experiments were carried out in the optimal conditions (PS, T, pH, M, IC and TS) previously calculated. After sterilization and inoculation, the media were incubated at 30°C for 84 h.
Results were analyzed by Experimental Design Module of the Statistica software package (Statistica 5.1 for Windows computer program manual; StatSoft Inc. Tulsa, OK, USA). The Student’s t-test was employed to check the statistical significance of the regression coefficients. The Fisher’s F-test for analysis of variance (ANOVA) was performed on experimental data to evaluate the statistical significance of the model.
Results
The first optimization experiment was performed to check the influence of different bagasse particle sizes on amylase production by A. niger UO-01 in SSF. The results obtained are shown in Fig. 1. Maximum enzyme production (109.12 EU/gds) was observed when the particle size of bagasse was in the range of 6–8 mm, decreasing for lower of higher particle sizes. In subsequent experiments, therefore, a particle size between 6–8 mm was used for the production of amylase by A. oryzae FQB-01.
After optimizing the particle size of bagasse, the following experiments were focused on the determination of the optimum levels of seven factors (T, pH, M, IC, TS, N and P) for high amylase production by using second-order orthogonal designs. The corresponding analysis of variance (ANOVA) of each empirical model obtained along with the values of the determination coefficient (R 2) and the adjusted determination coefficient (adj. R 2) are presented in Tables 2, 3 and 4. In each case, the lack-of-fit analysis gave non-significant P-values (>0.050) and F-values lower than the corresponding tabulated F-values, thus ensuring that the models obtained were highly significant. In addition, the high value of R 2 (0.960 or higher) indicated that the fitted models could explain at least 96% of the total variation in the responses. These facts indicate that the quadratic models were appropriate to fit and describe satisfactorily the experimental data.
After eliminating the non-significant terms, the final response equation for amylase production (TAA) in response to pH and temperature (T), in coded units, is given as follows:
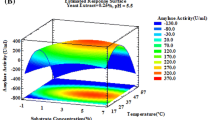
The three-dimensional response surface (Fig. 2) obtained from model (1) shows that the maximum TAA (111.58 EU/gds) was obtained at a pH value of 6.0 and a temperature of 30.2°C. This result is in perfect agreement with the TAA level (109.12 EU/gds) obtained in the previous experiment with the optimal particle size (6–8 mm), which was performed at T = 30°C and pH 6.0.
On the other hand, the polynomial model obtained for amylase production as a function of moisture content (M), inoculum concentration (IC) and total sugars concentration (TS) was:
The significant coefficients for the quadratic terms (M 2 and IC2) in model (2) implied the existence of optimum values for M and IC inside the experimental domain. Since the coefficient for the quadratic term TS2 was found to be non-significant and the linear term TS had a positive coefficient, the optimum value for this variable is located in the maximum level (70.5 mg/gds) assayed.
The response surfaces curves generated according to model (2) show the relative effects of two variables at a time on amylase production, when the coded value of the third variable is maintained at fixed levels of “−1” or “1” (Fig. 3a, b, c). The results obtained showed that an initial moisture content of 75.3%, inoculum concentration of 1 × 107 spores/gds and starch concentration of 70.5 mg/gds were the best conditions to produce the high amounts of enzyme (308.24 EU/gds) with A. niger UO-01 in SSF.
From the comparison between the results obtained in submerged (Salas et al. 2006) and SSF (Fig. 3), it can be noted that amylase production ceased to increase from a starch concentration of 30 g l−1 in the submerged cultures, while in SSF this effect was not observed (Fig. 3b, c). In fact, the empirical model (2) predicted a linear increase in amylase concentration when the TS concentrations increased from 9.5 to 70.5 mg/gds (Fig. 3b, c). The same trend was observed for amylase production by A. oryzae CBS 125-59 by SSF on polyurethane foams (Murado et al. 1997).
On the other hand, the enzyme yield coefficients expressed as amylase concentration on initial total sugars (Y TAA/iTS in EU/g of initial TS) obtained in the submerged cultures with initial starch concentrations of 10, 20, 30 and 40 g l−1 (Salas et al. 2006) were calculated to be as 1,757, 2,067, 2,400 and 4,200 EU/g of initial TS, respectively. These values are lower than the Y TAA/iTS value obtained in the optimal conditions determined in this experiment, which was calculated to be as 4,372 EU/g of initial of TS.
The corresponding empirical equations obtained for TAA production as a function of the concentrations of NH4CH3COO (model 3), (NH4)2HPO4 (model 4), NaNO2 (model 5), NaNO3 (model 6), yeast extract (model 7) or (NH4)2SO4 (model 8) and the phosphorous source (KH2PO4) were:
The response surfaces obtained according to the above six models showed that the enzyme production was highly influenced not only by the phosphorus concentrations but also by the type and concentrations of the nitrogen source (Fig. 4). In general, a linear increase in TAA production was observed when the concentrations of N and P increased up to the optimal values, and thereafter TAA production declined sharply. In addition, the optimal N and P values as well as the maximum TAA levels obtained with the use of the six nitrogen sources were clearly different (Table 5). The highest amylase concentration (457.82 EU/gds) was obtained with the use of yeast extract followed by NH4CH3COO (378.88 EU/gds), NaNO2 (359.79 EU/gds), (NH4)2HPO4 (355.47 EU/gds), (NH4)2SO4 (326.48 EU/gds) and NaNO3 (317.50 EU/gds).
Verification of the predicted results in the optimal conditions
Verification of the predicted results was accomplished by using the optimized conditions in triplicate incubation experiments for each nitrogen source (Fig. 5). Under the calculated optimal culture conditions, the mean values of TAA (in EU/gds) after 84 h of incubation were 380.55 ± 2.06 (in case of NH4CH3COO), 353.32 ± 2.64 (in case of (NH4)2HPO4), 358.12 ± 1.84 (in case of NaNO2), 319.85 ± 2.41 (in case of NaNO3), 455.16 ± 3.89 (in case of yeast extract) and 325.04 ± 1.38 (in case of (NH4)2SO4). These values were found to be very close to the predicted values (Table 5) obtained from the empirical models (3), (4), (5), (6), (7) and (8). Therefore, the models developed can be considered to be accurate and reliable for predicting amylase production by A. niger UO-01 in SSF with sugarcane bagasse as a support material.
Discussion
Particle size, incubation temperature, pH, initial moisture content of solid material, inoculum concentration, as well as the concentrations of sugars, nitrogen and phosphorous sources are important factors that influence amylase production by A. oryzae strains in SSF (Gigras et al. 2002; Murado et al. 1997; Francis et al. 2003). However, the effects of these variables on enzyme production depend on the producer strain and the solid substrate used. For example, optimal particle size, moisture level and inoculum concentration for α-amylase production in SSF by a Penicillium chrysogenum strain were >1 mm, 75 and 20% for corncob leaf, >1 mm, 65 and 20% for wheat straw, 1 mm, 65 and 20% for wheat bran and >1 mm, 55 and 30% for rye straw (Balkan and Ertan 2007). However, initial moisture content of 70%, inoculum concentration of 1 × 107 spores/g dry substrate and an incubation temperature in the range of 25–30°C were necessary to achieve the maximum α-amylase production by A. oryzae NRRL 6270 on spent brewing grains (Francis et al. 2002). In the same way, the optimal moisture levels for glucoamylase production by Aspergillus sp. A3 in wheat bran (Ellaiah et al. 2002) and for α-amylase and glucoamylase production by A. niger LPB 28 in cassava starch and sugarcane bagasse (Spier et al. 2006) were 80 and 90%, respectively.
Taking into account these observations, amylase production by A. oryzae FQB-01 under SSF using sugarcane bagasse as a support material was optimized by determining the optimal values of eight important process variables (PS, T, pH, IC, M, TS, N and P).
A particle size in the range of 6–8 mm gave the highest amylase production in comparison with the enzyme levels obtained with other particle sizes (<1, 1–2, 2–4, 4–6, 8–10, 10–12, 12–14, 14–16 and 16–20 mm). Thus, the optimum particle size (6–8 mm) probably provides the most effective support material for attachment of the fungal strain (Laxmi et al. 2008), or facilitates the mass transfer performance (gas and nutrient diffusion) greatly, resulting in a better respiration/aeration efficiency and an increased availability of nutrients (Balkan and Ertan 2007; Mazutti et al. 2007; Pandey et al. 1999). These facts favour both the growth and enzyme production (Balkan and Ertan 2007; Pandey et al. 1999).
Aspergillus niger UO-01 exhibited its best performance for enzyme production in the mesophilic range (T = 30.2°C), as it was reported before for A. niger LPB 28, which produce the highest amylase level at 30°C (Spier et al. 2006). Temperature values higher than 30°C may lead to enzymatic inactivation (Mazutti et al. 2006) or suppression of cell viability (Francis et al. 2002). In contrast, low temperature values may reduce the metabolism of the microorganism (Mazutti et al. 2006) and consequently, the enzyme synthesis. In this study, it was also verified that A. niger UO-01 has a preference to pH around 6.0 for amylase production, but its production capacity decreased for pH levels higher and lower, probably as a consequence of a reduction in the metabolic activity of the amylase-producing strain (Ellaiah et al. 2002).
The effect of initial moisture content on enzyme production in SSF has usually been related to the interference that this variable produces on the physical properties of the solid particles (Ellaiah et al. 2002). Thus, higher moisture content could reduce the porosity of the solid substrate, thus interfering with the oxygen transfer (Lonsane et al. 1985). On the contrary, lower moisture levels can lead to a reduction in the solubility of nutrients of the substrate, low degree of swelling and high water tension (Zadrazil and Brunnert 1981).
Inoculum concentration is other important factor that influences the production of metabolites under SSF (Balkan and Ertan 2007; Mazutti et al. 2007; Pandey 2003). An inoculum concentration higher than the optimum value may produce a high amount of biomass which rapidly depletes the nutrients necessary for growth and product synthesis (Baysal et al. 2003; Selvakumar and Pandey 1999). On the other hand, lower inoculum levels may give insufficient biomass and allow the growth of undesirable organisms in the production medium. This increases the necessary time to grow to an optimum number to consume the substrate and synthesise the desired product (Balkan and Ertan 2007; Kashyap et al. 2002).
On the other hand, yeast extract was the best nitrogen source for amylase production, probably due to its high content in minerals, vitamins, coenzymes and nitrogen components (Guerra and Pastrana 2002). Although the optimal concentrations of nitrogen and phosphorous were different for each nitrogen source used, at concentrations higher than the optimal values both the nitrogen and phosphorous sources caused inhibition of enzyme synthesis. Since the liquid medium was initially buffered at pH 6.0 in the six experiments, this inhibition could be related with the repression effect that the high concentrations of nitrogen (Abu and Ado 2004; Michelena and Castillo 1984) and phosphorous (Michelena and Castillo 1984) produced on amylase production. This repression effect has been observed before for cellulose production by Aspergillus terreus (Ali et al. 1991).
Some studies on the effect of nutrients (carbon, nitrogen and phosphorous) on amylase production in both SSF and SLF have been carried out using a fixed concentration of both nutrients (Ellaiah et al. 2002; Francis et al. 2002; Kunamneni et al. 2005). Although this approach allows the selection of the best N and P sources, determination of their optimal concentrations is not possible. Other studies based on the optimization of N and P levels for amylase production, used the medium optimization by the traditional “one variable at a time” technique (Kekos et al. 1987; Kunamneni et al. 2005; Michelena and Castillo 1984). However this method is time consuming and could lead to an incomplete interpretation of the behaviour of the system, resulting in a lack of predictive ability, mainly when there are interactions between the independent variables. In contrast, determination of the effects of N and P concentrations and their interaction on amylase production by using central composite designs is an effective way to obtain the best nitrogen and phosphate source as well as their optimal concentrations. With this procedure, the cost of amylase production in SSF could be reduced by selecting the cheapest N and P source. With regard to this, the optimum N and P values obtained in the present work for high enzyme production by A. oryzae UO-01 were different for each nitrogen source used (Table 5). This is an important factor that must be considered for a large scale production, because the amounts of nutrients added to the culture media have a direct relation with the production cost. Thus, in the present study, the estimated costs associated with the use of the optimum amounts of each nitrogen source and KH2PO4 were calculated by taking into account the price of each nutrient. As it can be observed in Table 5, the combined use of yeast extract and KH2PO4 for amylase production gave the highest amylase levels but also the highest cost. On the other hand, the use of NH4CH3COO provided an optimum amylase concentration 1.2 times lower than yeast extract, but gave a cost 1.5 times lower than that of yeast extract. These results suggest that NH4CH3COO could be the most appropriate nitrogen source for a low-cost amylase production by A. niger UO-01 in solid-state cultivation with sugarcane bagasse.
Conclusions
The results obtained in this work showed that sugarcane bagasse is a valuable support material for amylase production and provided useful information and reference for the optimization of medium composition for other SSF processes with the same support material. Statistical optimization method for fermentation process using response surface methodology could overcome the limitations of classic empirical methods and proved to be a powerful tool for the optimization of amylase production by A. niger UO-01 in SSF. On the other hand, validation experiments showed that the predicted values by the empirical models agreed with the experimental values well. These results therefore proved the validity and corroborate the effectiveness and reliability of the empirical models obtained.
Abbreviations
- RSM:
-
Response surface methodology
- SLF:
-
Submerged liquid fermentation
- SSF:
-
Solid-state fermentation
- gds:
-
g of dry support
- TAA:
-
Total amylase activity
- EU:
-
Enzymatic units
- T:
-
Temperature
- IC:
-
Inoculum concentration
- M:
-
Moisture content
- TS:
-
Total sugars concentration
- N:
-
Nitrogen concentration
- P:
-
Phosphorous concentration
References
Abu EA, Ado SA (2004) Comparative studies on the effect of organic and inorganic nitrogen supplementation of millet and sorghum pomace on the production of three industrial enzymes by Aspergillus niger SL.1. Biokemistry 16:64–70
Ali S, Sayed A, Sarker RT, Alam R (1991) Factors affecting cellulose production by Aspergillus terreus using water hyacinth. World J Microbiol Biotechnol 7:62–66
Balkan B, Ertan F (2007) Production of α-amylase from P. chrysogenum. Food Technol Biotechnol 45:439–442
Baysal Z, Uyar F, Aytekin C (2003) Solid state fermentation for production of α-amylase by a thermotolerant Bacillus subtilis from hot-spring water. Process Biochem 38:1665–1668
Bhargav S, Panda BP, Ali M, Javed S (2008) Solid-state fermentation: an overview. Chem Biochem Eng Q 22:49–70
Doran JB, Aldrich HC, Ingram LO (1994) Saccharification and fermentation of sugarcane bagasse. Biotechnol Bioeng 44:240–247
Ellaiah P, Adinarayana K, Bhavani Y, Padmaja P, Srinivasulu B (2002) Optimization of process parameters for glucoamylase production under solid state fermentation by a new isolated Aspergillus species. Process Biochem 38:615–620
Francis F, Sabu A, Nampoothiri KM, Szakacs G, Pandey A (2002) Synthesis of α-amylase by Aspergillus oryzae in solid-state fermentation. J Basic Microbiol 5:320–326
Francis F, Sabu A, Nampoothiri KM, Ramachandran S, Ghosh S, Szakacs G, Pandey A (2003) Use of response surface methodology for optimizing process parameters for the production of α-amylase by Aspergillus oryzae. Biochem Eng J 15:107–115
Gigras P, Sahai V, Grupta R (2002) Statistical media optimization and production of its ITS alpha amylase from Aspergillus oryzae in a biorreactor. Curr Microbiol 45:203–208
Guerra NP, Pastrana L (2002) Nisin and pediocin production on mussel-processing waste supplemented with glucose and five nitrogen sources. Lett Appl Microbiol 34:114–118
Guerra NP, Torrado A, López C, Pastrana L (2003) Main characteristics and applications of solid substrate fermentation. Electron J Environ Agric Food Chem 2: 343–350. Available from: http://ejeafche.uvigo.es
Gutiérrez-Correa M, Tengerdy RP (1997) Production of cellulase on sugarcane bagasse by fungal mixed culture solid substrate fermentation. Biotechnol Lett 19:665–667
Kashyap P, Sabu A, Pandey A, Szakacs G (2002) Extra-cellular l-glutaminase production by Zygosaccharomyces rouxii under solid-state fermentation. Process Biochem 38:307–312
Kekos D, Galiotou-Panayotou M, Macris BJ (1987) Some nutritional factors affecting α-amylase production by Calvatia gigantea. Appl Microbiol Biotechnol 26:527–530
Kunamneni A, Permaul K, Singh S (2005) Amylase production in solid state fermentation by the thermophilic fungus Thermomyces lanuginosus. J Biosci Bioeng 100:168–171
Laxmi GS, Sathish T, Rao CS, Brahmaiah P, Hymavathi M, Prakasham RS (2008) Palm fiber as novel substrate for enhanced xylanase production by isolated Aspergillus sp. RSP-6. Curr Trends Biotechnol Pharm 2:447–455
Lonsane BK, Ghildyal NP, Budiatman S, Ramakrishna SV (1985) Engineering aspects of solid state fermentation. Enzyme Microb Technol 7:258–265
Mazutti M, Bender JP, Treichel H, Di Luccio M (2006) Optimization of inulinase production by solid-state fermentation using sugarcane bagasse as substrate. Enzyme Microb Technol 39:56–59
Mazutti M, Ceni G, Di Luccio M, Treichel H (2007) Production of inulinase by solid-state fermentation: effect of process parameters on production and preliminary characterization of enzyme preparations. Bioprocess Biosyst Eng 30:297–304
Michelena VV, Castillo FJ (1984) Production of amylase by Aspergillus foetidus on rice flour medium and characterization of the enzyme. J Appl Bacteriol 56:395–407
Murado MA, Siso MIG, González MP, Montemayor MI, Pastrana L, Mirón J (1993) Characterization of microbial biomasses and amylolytic preparations obtained from mussel-processing waste treatment. Bioresour Technol 43:117–125
Murado MA, González MP, Torrado A, Pastrana LM (1997) Amylase production by solid state culture of Aspergillus oryzae on polyurethane foams. Some mechanistic approaches from an empirical model. Process Biochem 32:35–42
Pandey A (2003) Solid state fermentation. Biochem Eng J 13:81–84
Pandey A, Selvakumar P, Soccol CR, Nigam P (1999) Solid state fermentation for the production of industrial enzymes. Curr Sci 77:149–162
Pandey A, Soccol CR, Nigam P, Soccol VT (2000) Biotechnological potential of agro-industrial residues. I: sugarcane bagasse. Bioresour Technol 74:69–80
Raimbault M (1998) General and microbiological aspects of solid substrate fermentation. Electron J Biotechnol 1: 1–22. Available from http://www.ejb.org/content/vol1/issue3/full/9/index.html
Ramachandran S, Patel AK, Nampoothiri KM, Francis F, Nagy V, Szakacs G, Pandey A (2004) Coconut oil cake-a potential raw material for the production of α-amylase. Bioresour Technol 93:169–174
Rodríguez S, Sanromán MA (2006) Application of solid-state fermentation to food industry-A review. J Food Eng 76:291–302
Salas M, Rodríguez M, Guerra NP, Pérez R (2006) Amylase production by Aspergillus niger in submerged cultivation on two wastes from food industries. J Food Eng 73:93–100
Selvakumar P, Pandey A (1999) Solid state fermentation for the synthesis of inulinase from Staphylococcus sp. and Kluyveromyces marxianus. Process Biochem 34:851–858
Spier MR, Woiciechowski AL, Vandenberghe LPS, Zoclo CR (2006) Production and characterization of amylases by Aspergillus niger under solid state fermentation using agro industrials products. Int J Food Eng 2. Available from: http://www.bepress.com/ijfe/vol2/iss3/art6/
Uma Maheswar Rao JL, Satyanarayana T (2007) Improving production of hyperthermostable and high maltose-forming α-amylase by an extreme thermophile Geobacillus thermoleovorans using response surface methodology and its applications. Bioresour Technol 98:345–352
Zadrazil F, Brunnert H (1981) Investigation of physical parameters important for the SSF of straw by white rot fungi. Eur J Appl Microbiol Biotechnol 11:183–188
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rosés, R.P., Guerra, N.P. Optimization of amylase production by Aspergillus niger in solid-state fermentation using sugarcane bagasse as solid support material. World J Microbiol Biotechnol 25, 1929–1939 (2009). https://doi.org/10.1007/s11274-009-0091-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-009-0091-6